Discover bioregulator peptides, the Russian anti-aging breakthrough backed by 35 years of research. Learn how Epitalon and Thymalin work at the DNA level.







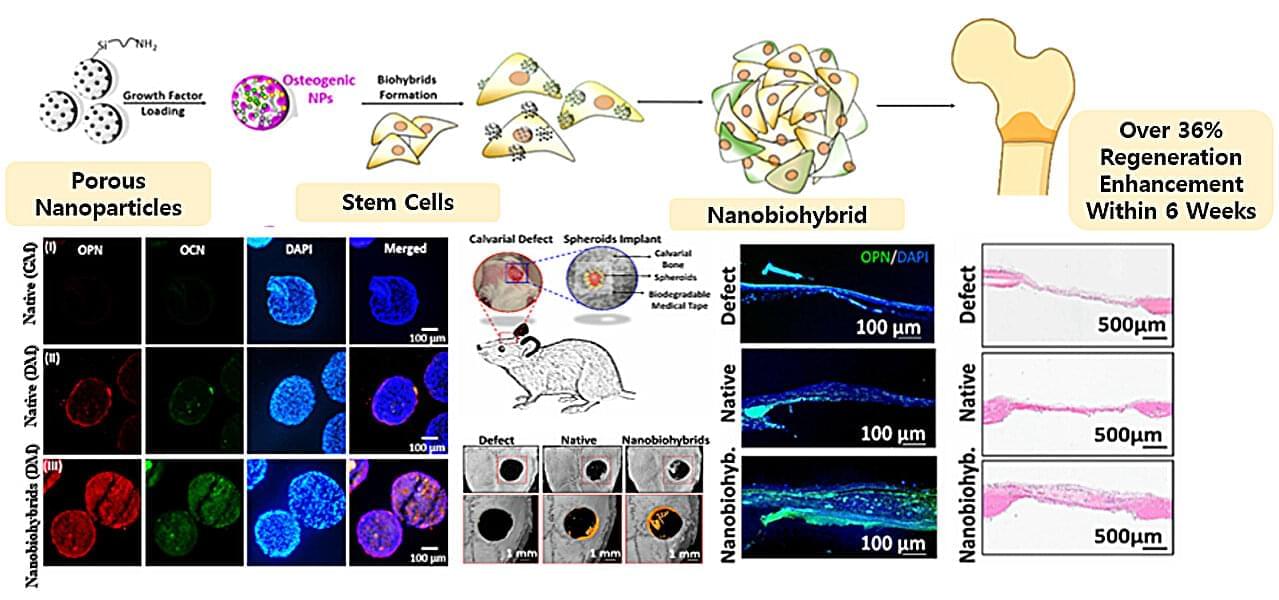
A research team in South Korea has successfully developed a novel technology that combines nanoparticles with stem cells to significantly improve 3D bone tissue regeneration. This advancement marks a step forward in the treatment of bone fractures and injuries, as well as in next-generation regenerative medicine.
The research is published in the journal ACS Biomaterials Science & Engineering.
Dr. Ki Young Kim and her team at the Korea Research Institute of Chemical Technology (KRICT), in collaboration with Professor Laura Ha at Sunmoon University, have engineered a nanoparticle-stem cell hybrid, termed a nanobiohybrid by integrating mesoporous silica nanoparticles (mSiO₂ NPs) with human adipose-derived mesenchymal stem cells (hADMSCs). The resulting hybrid cells demonstrated markedly enhanced osteogenic (bone-forming) capability.
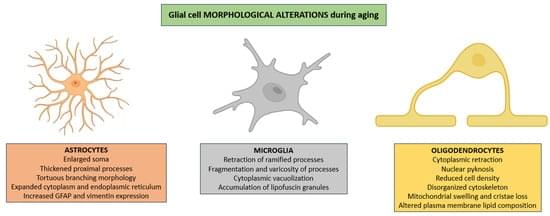
Aging is accompanied by complex cellular and molecular changes that compromise CNS function. Among these, glial cells (astrocytes, microglia, and oligodendrocytes) play a central role in maintaining neural homeostasis, modulating synaptic activity, and supporting metabolic demands. Emerging evidence indicates that aging disrupts glial cell physiology through processes including mitochondrial dysfunction, impaired proteostasis, chronic low-grade inflammation, and altered intercellular signaling. These alterations contribute to synaptic decline, myelin degeneration, and persistent, low-grade inflammation of the CNS. This review synthesizes current knowledge on the bidirectional relationship between aging and glial cell dysfunction, highlighting how age-related systemic and CNS-specific factors exacerbate glial impairments and, in turn, accelerate neural deterioration.