From the article:
To put this in quantitative terms: consider an individual at the 5th percentile of genetic vitality. Even with an impeccable lifestyle, such a person might only reach the 25th percentile of vitality (energy levels, mood, motivation). Now consider someone at the 95th percentile of genetic vitality. Even with a mediocre or actively harmful lifestyle, this person might still operate at the 75th percentile or above. The gap between these two individuals, after both have optimized (or neglected) every modifiable factor, is entirely genetic.
The single most effective thing one can do to guarantee great energy, mood, motivation, metabolism, cognition, physique, and longevity is to pick the right parents.
This is not to say that lifestyle, hormones, and pharmaceuticals are unimportant. They clearly matter, often enormously. Rather, the point is that these interventions operate within a window whose size, position, and ceiling are defined by inherited genetic variation.
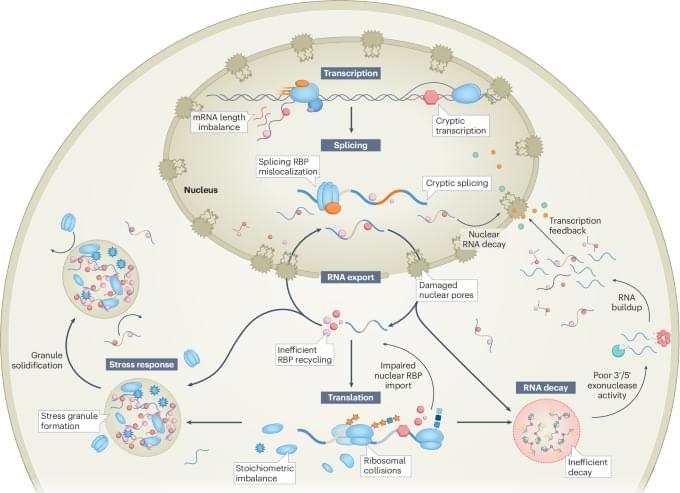
Vitality is affected by many things which I extensively discuss on my blog. These include metabolic health, hormones, inflammation, diet, exercise, and sleep, among other things. Each of these domains is important, and each is modifiable to varying degrees through lifestyle choices, pharmaceutical interventions, or behavioral change.
However, every one of these discussions has implicitly assumed a background variable that I have largely unaddressed: genetics.