Losing muscle strength is a natural part of aging. At the core of this decline is a drop in the number of muscle stem cells (MuSCs), the specialized cells responsible for maintaining and regenerating muscle tissue throughout our lives. Loss of muscle strength can severely affect mobility, increasing the risk of falls, fractures and, most importantly, the loss of independence.
Published in Nature Aging, a recent study took a crucial first step toward restoring stem cell function in aging muscles—gaining a clearer understanding of how metabolism changes when stem cells are activated and how these critical processes weaken with age.
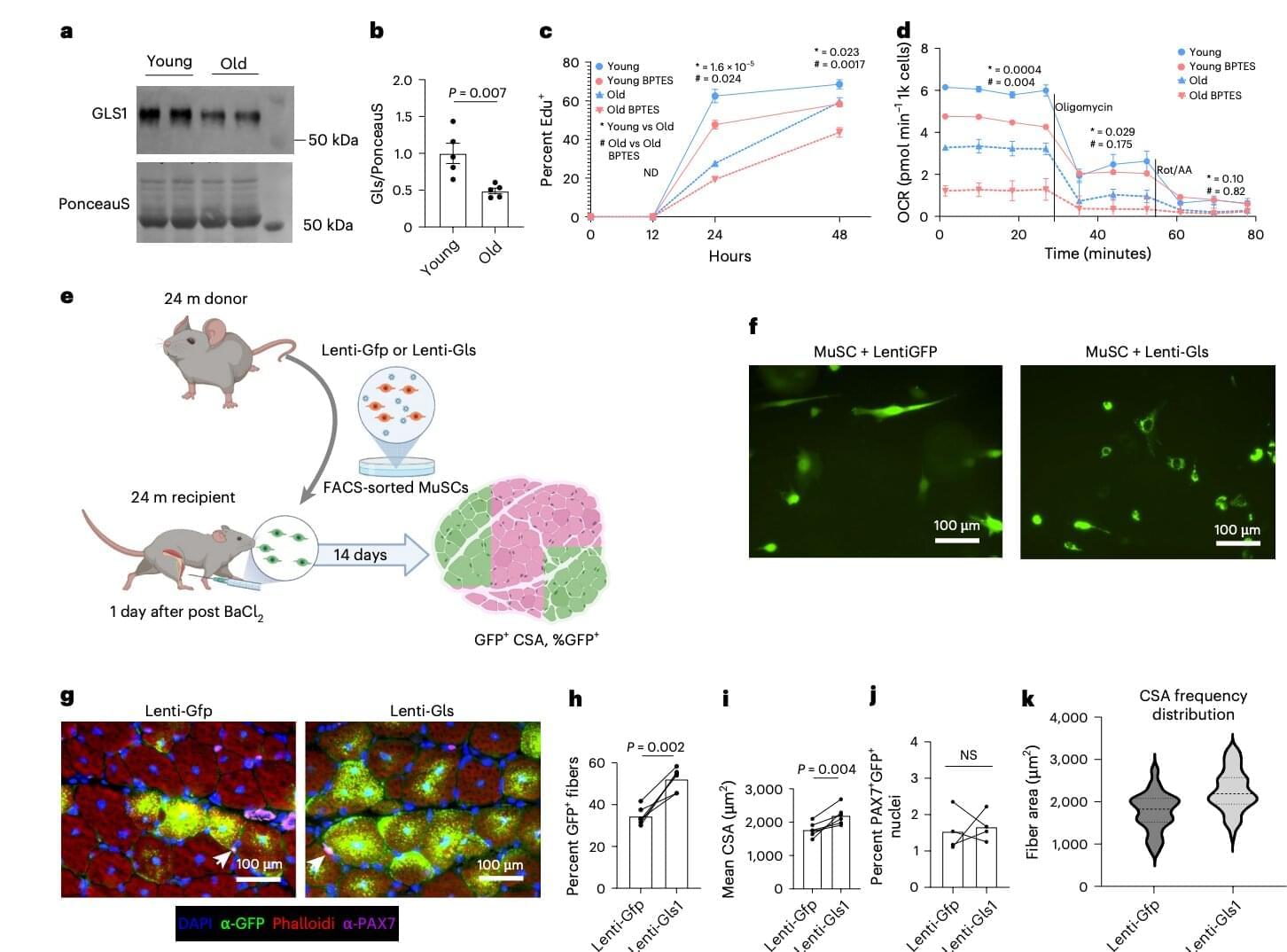
The researchers’ investigation led them to glutamine metabolism, the process by which cells use the amino acid glutamine to support essential functions. They found that for MuSCs, glutamine is more than just a nutrient. It provides the raw material needed to produce fatty acids that help cells grow, divide, and repair damaged muscles.