Researchers at San Diego State University and Michigan State University are shedding new light on how viruses meticulously pack their genetic material — a breakthrough that could help researchers engineer antivirals and gene therapies.



A paper coauthored by geneticist George Church has been retracted following an internal review at a university where several coauthors are based.
The article appeared in the Proceedings of the National Academy of Sciences in 2022. The work supports an anti-aging gene therapy developed by BioViva, a company for which Church serves as an adviser. The paper’s authors claim cytomegalovirus (CMV) can be a gene therapy vector for a treatment for “aging-associated decline” that can be inhaled or injected monthly.
The work has been cited 41 times, two of which are citations from corrections to the article, according to Clarivate’s Web of Science.

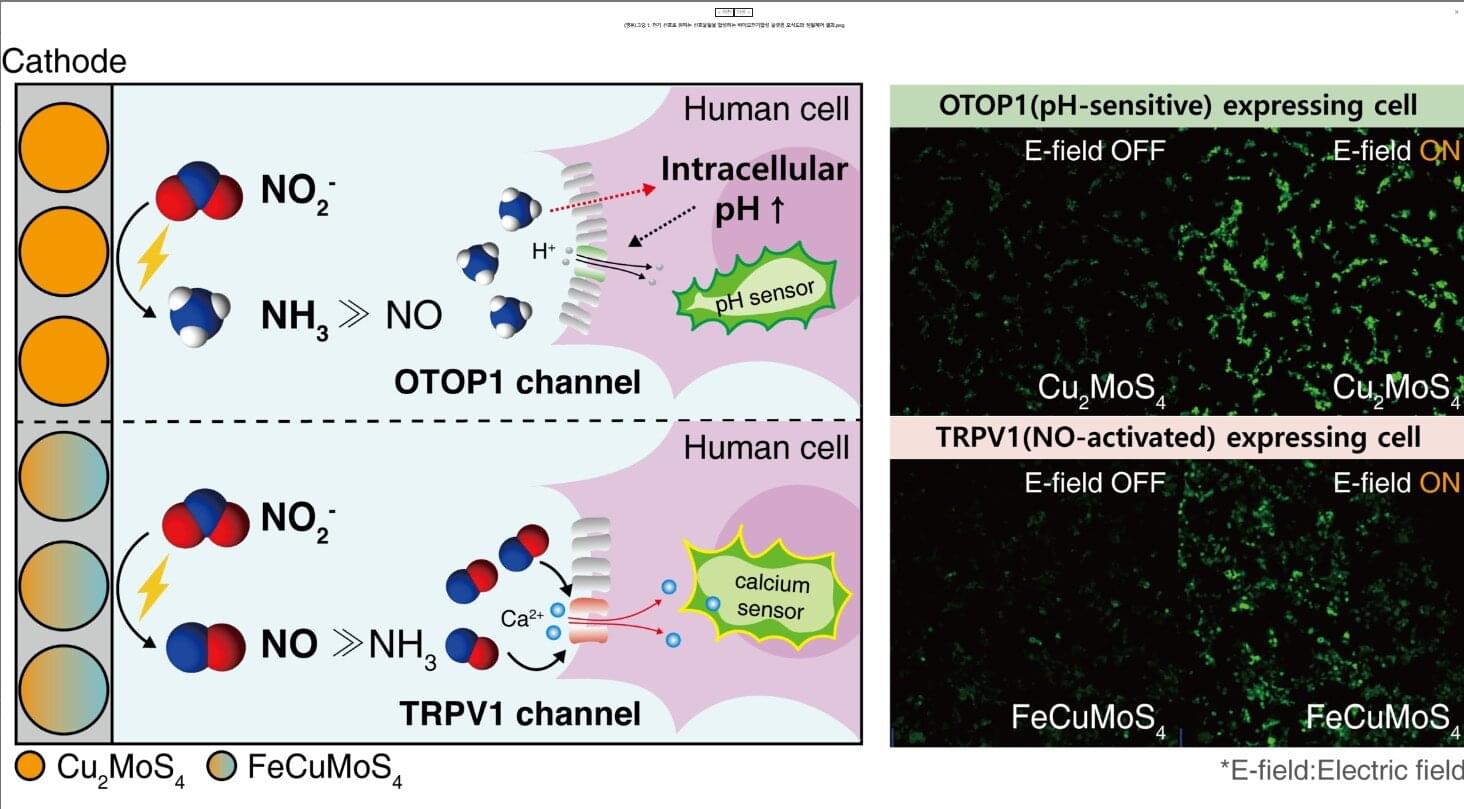
Cells use various signaling molecules to regulate the nervous, immune, and vascular systems. Among these, nitric oxide (NO) and ammonia (NH₃) play important roles, but their chemical instability and gaseous nature make them difficult to generate or control externally.
A KAIST research team has developed a platform that generates specific signaling molecules in situ from a single precursor under an applied electrical signal, enabling switch-like, precise spatiotemporal control of cellular responses. This approach could provide a foundation for future medical technologies such as electroceuticals, electrogenetics, and personalized cell therapies.
The research team led by Professor Jimin Park from the Department of Chemical and Biomolecular Engineering, in collaboration with Professor Jihan Kim’s group, has developed a bioelectrosynthesis platform capable of producing either nitric oxide or ammonia on demand using only an electrical signal. The platform allows control over the timing, spatial range, and duration of cell responses.
Ovarian cancer is the leading cause of death among women with gynecological cancers. The current medical playbook—surgery followed by chemotherapy—initially shows promise. Tumors shrink, sometimes disappearing entirely. But in more than 80% of patients, the cancer not only comes back, but returns more aggressive and increasingly resistant to the very treatments that once seemed effective.
But now, there could be new hope. In a study published in the journal Med, UCLA researchers have detailed their development of a new type of immune cell therapy, called CAR-NKT cell therapy, that could transform ovarian cancer care by delivering superior cancer-fighting power.
“This is the culmination of over a decade of work in my lab and represents over six years of collaboration with gynecologic oncologist Dr. Sanaz Memarzadeh,” said co-senior author Lili Yang, a professor of microbiology, immunology and molecular genetics and a member of the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA.

In studies with genetically engineered mice, Johns Hopkins Medicine researchers say they have identified a potentially new biological target involving Aplp1, a cell surface protein that drives the spread of Parkinson’s disease-causing alpha-synuclein.
The findings, published May 31 2024 in Nature Communications, reveal how Aplp1 connects with Lag3, another cell surface receptor, in a key part of a process that helps spread harmful alpha-synuclein proteins to brain cells. Those protein buildups are hallmarks of Parkinson’s disease.
Notably, the researchers say, Lag3 is already the target of a combination cancer drug approved by the U.S. Food and Drug Administration (FDA) that uses antibodies to “teach” the human immune system what to seek and destroy.
Basically I believe that the flatworm could give the genetic code for essentially brain immortality other just nad plus. But it would have to be made in the brain and controlled possibly with nanotransfection which would scan the body and modify the human brain cells to have its characteristics that may already exist in the human brain also.
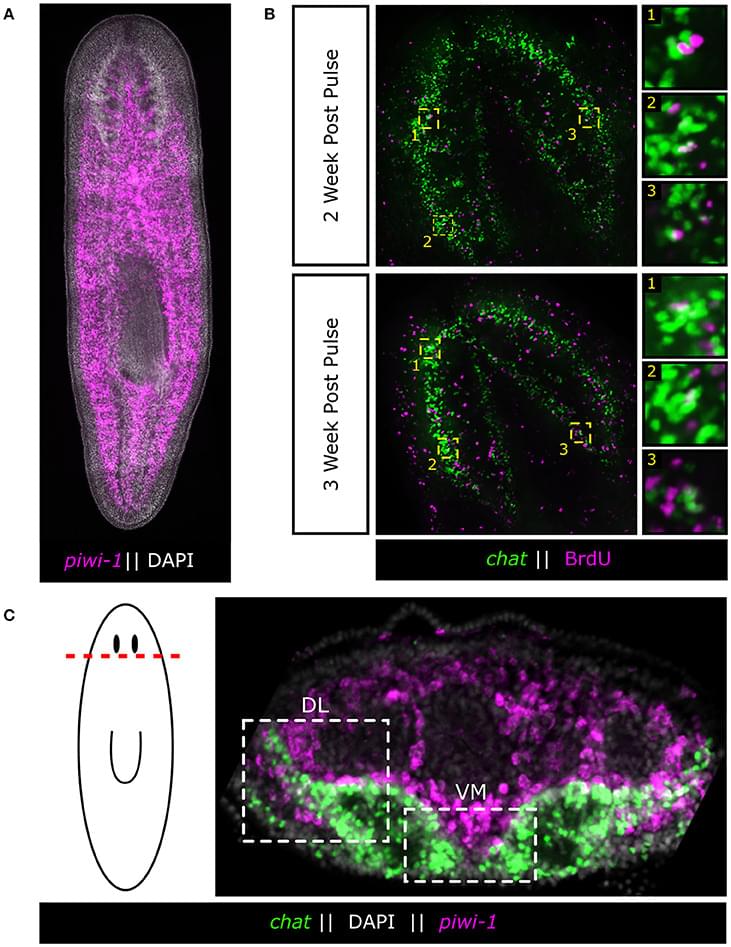
Powerful genetic tools in classical laboratory models have been fundamental to our understanding of how stem cells give rise to complex neural tissues during embryonic development. In contrast, adult neurogenesis in our model systems, if present, is typically constrained to one or a few zones of the adult brain to produce a limited subset of neurons leading to the dogma that the brain is primarily fixed post-development. The freshwater planarian (flatworm) is an invertebrate model system that challenges this dogma. The planarian possesses a brain containing several thousand neurons with very high rates of cell turnover (homeostasis), which can also be fully regenerated de novo from injury in just 7 days. Both homeostasis and regeneration depend on the activity of a large population of adult stem cells, called neoblasts, throughout the planarian body. Thus, much effort has been put forth to understand how the flatworm can continually give rise to the diversity of cell types found in the adult brain. Here we focus on work using single-cell genomics and functional analyses to unravel the cellular hierarchies from stem cell to neuron. In addition, we will review what is known about how planarians utilize developmental signaling to maintain proper tissue patterning, homeostasis, and cell-type diversity in their brains. Together, planarians are a powerful emerging model system to study the dynamics of adult neurogenesis and regeneration.
The adult brain has long been thought to be a fixed structure due to its immense complexity as is illustrated succinctly in the following quote from prominent nineteenth century neuroscientist and Nobel laureate Santiago Ramón y Cajal:
“Once the development was ended, the founts of growth and regeneration of the axons and dendrites dried up irrevocably. In the adult centers, the nerve paths are something fixed, ended, and immutable. Everything may die, nothing may be regenerated. It is for the science of the future to change, if possible, this harsh decree.”
Dr. Esra Çavuşoğlu, PhD’s 71st Ayık Kafa podcast guest is Liz Parrish explores the transformative potential of gene therapy in extending human longevity and enhancing healthspan.
Liz Parrish MBA, is the Founder and CEO of BioViva Sciences USA Inc. BioViva is committed to extending healthy lifespans using gene therapy and works on combinatorial gene therapies with its proprietary CMV gene therapy delivery platform.
Liz is a humanitarian, entrepreneur, author, and innovator. In addition, she is a proponent of the Best Choice Medicine plan (BCM), a more efficient and streamlined regulatory model around the use of genetic therapies.
She shares her personal journey, the scientific basis for gene therapy, and the economic implications of aging-related diseases. The podcast also takes a closer look at the four different gene therapies that Liz Parrish administered to herself: Klotho, Follistatin, PGC-1α, and Telomerase Reverse Transcriptase. The discussion covers the various gene therapies being developed, their safety, ethical considerations, and the importance of early intervention. Liz emphasizes the need for public awareness and investment in longevity research to make these therapies accessible to all.
#EsraÇavuşoğlu #AyıkKafa #ElevatingLifeEvolvingHealth #Longevilab #LizParrish #Longevity #genetherapy.
Liz Parrish: