The ubiquitous Epstein-Barr virus is increasingly being linked to conditions like multiple sclerosis and lupus. But why do only some people who catch it develop these complications? The answer may lie in our genetics

The COVID-19 pandemic gave us tremendous perspective on how wildly symptoms and outcomes can vary between patients experiencing the same infection. How can two people infected by the same pathogen have such different responses? It largely comes down to variability in genetics (the genes you inherit) and life experience (your environmental, infection, and vaccination history).
These two influences are imprinted on our cells through small molecular alterations called epigenetic changes, which shape cell identity and function by controlling whether genes are turned “on” or “off.”
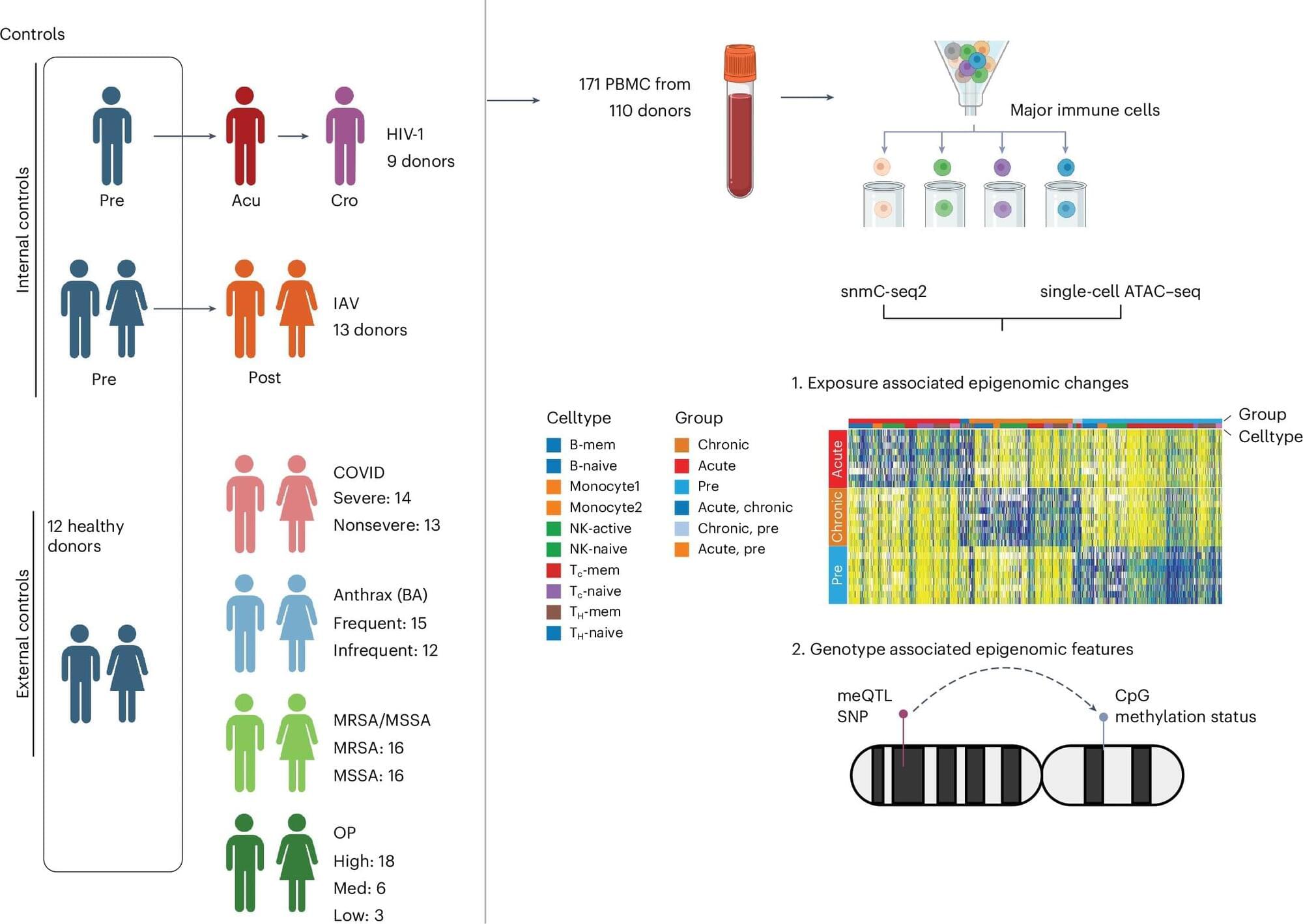
Salk Institute researchers are debuting a new epigenetic catalog that reveals the distinct effects of genetic inheritance and life experience on various types of immune cells. The new cell type-specific database, published in Nature Genetics, helps explain individual differences in immune responses and may serve as the foundation for more effective and personalized therapeutics.
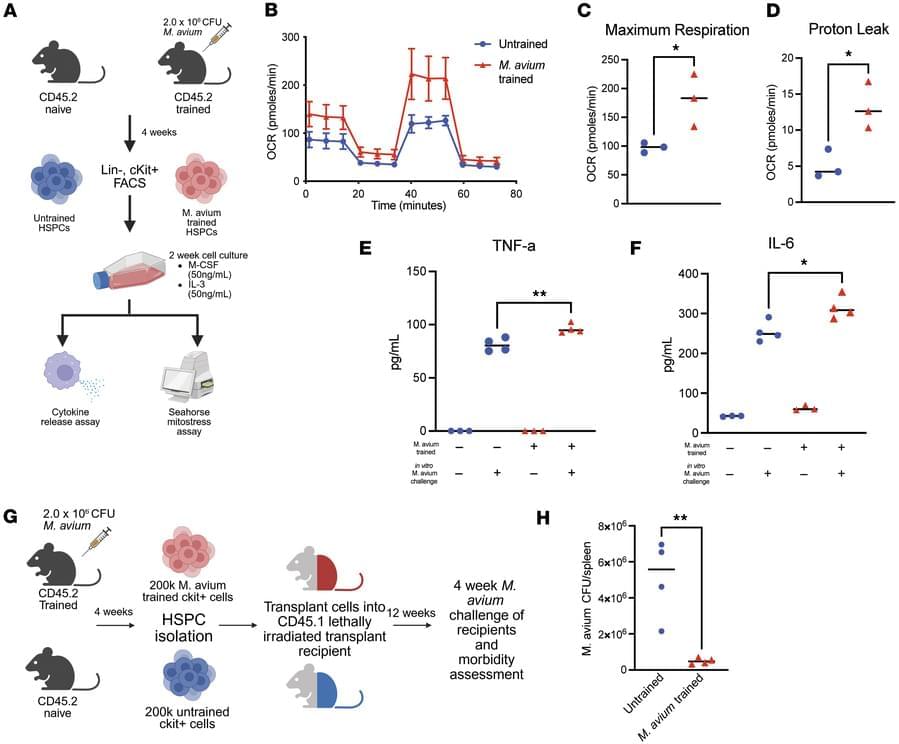
Katherine Y. King & team now identify epigenetic changes in hematopoietic stem and progenitor cells in a mycobacterial infection model that are retained in downstream macrophages, providing mechanistic mediators of innate immune memory and explaining persistence of central trained immunity.
1Graduate Program in Cancer and Cell Biology.
2Department of Pediatrics, Division of Infectious Disease, Texas Children’s Hospital and Baylor College of Medicine.
3Stem Cells and Regenerative Medicine Center.
4Department of Molecular and Human Genetics.
Dr. Natalie Yivgi-Ohana, Ph.D. — CEO, Minovia Therapeutics — Harnessing The Therapeutic Power Of Mitochondria
Is Co-Founder and CEO of Minovia Therapeutics (https://minoviatx.com/), a biotech company dedicated to rapidly advance life-changing therapies that address the unmet need of serious and complex mitochondrial diseases, and are the first clinical-stage company to develop a mitochondrial transplantation approach to treat a broad range of indications generated by a mitochondrial dysfunction which lead to rare-genetic or age-related diseases.
Dr. Yivgi-Ohana has twenty years of experience in mitochondrial research and received her Ph.D. in Biochemistry at The Hebrew University, after which she completed her postdoctoral fellowship at the Weizmann Institute of Science.
Dr. Yivgi-Ohana also has her B.Sc., Medical Sciences Ben-Gurion University of the Negev and her Master’s Degree, Human Reproduction Bar-Ilan University.
Dr. Yivgi-Ohana founded Minovia with a passion to help children and adults with mitochondrial diseases worldwide.
Earlier work linked the experimental drug ‘IC7Fc’ to improvements in type 2 diabetes, and new research now points to a possible role in cardiovascular health as well. Scientists report that the compound may lower the risk of heart disease by reducing harmful cholesterol in the bloodstream and calming inflammatory activity that damages blood vessels over time.
The findings come from a preclinical study published in Science Advances, led by researchers at Leiden University Medical Centre in the Netherlands in collaboration with Monash University and other international partners.
In experiments involving mice genetically predisposed to heart disease, treatment with IC7Fc led to clear reductions in blood fat (triglycerides) and cholesterol, markers closely linked to the development of cardiovascular complications.
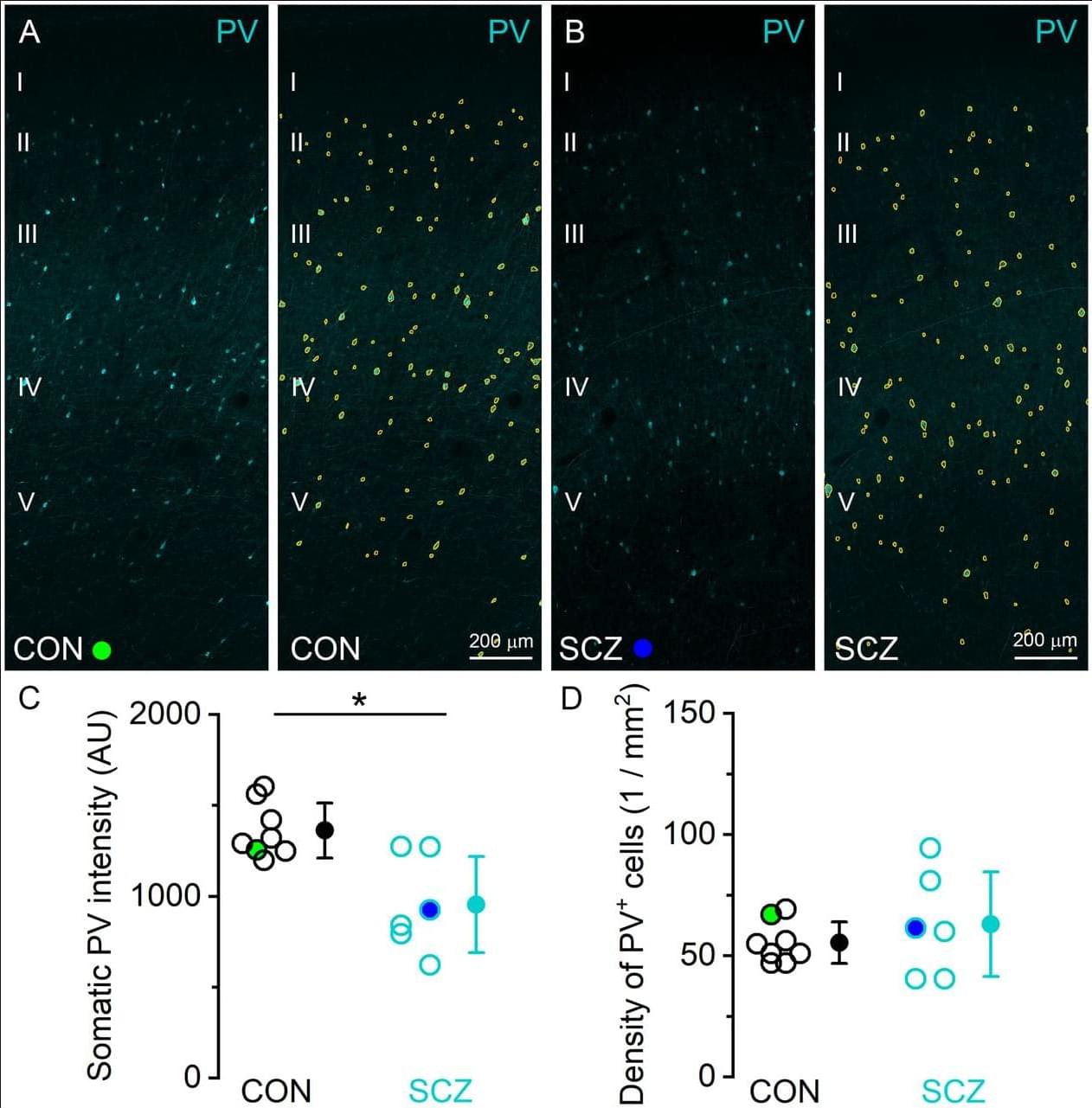
JNeurosci: Lorincz et al. provide novel insights into the molecular mechanisms underlying excitatory synaptic dysfunction in schizophrenia.
▶️
Abnormal excitatory synaptic transmission in the human prefrontal cortex has been implicated in the pathophysiology of schizophrenia based primarily on genetic evidence. However, changes in synaptic function cannot be predicted from altered gene expressions, but determining the amount, density, and subsynaptic distribution of synaptic proteins is the only reliable indirect readout of function. Detecting proteins in individual synapses of human postmortem tissues has been severely constrained by technical limitations. Here we overcome this limitation by optimizing a high-resolution, quantitative localization method to facilitate antigen recognition at excitatory synapses in postmortem brains of both sexes.
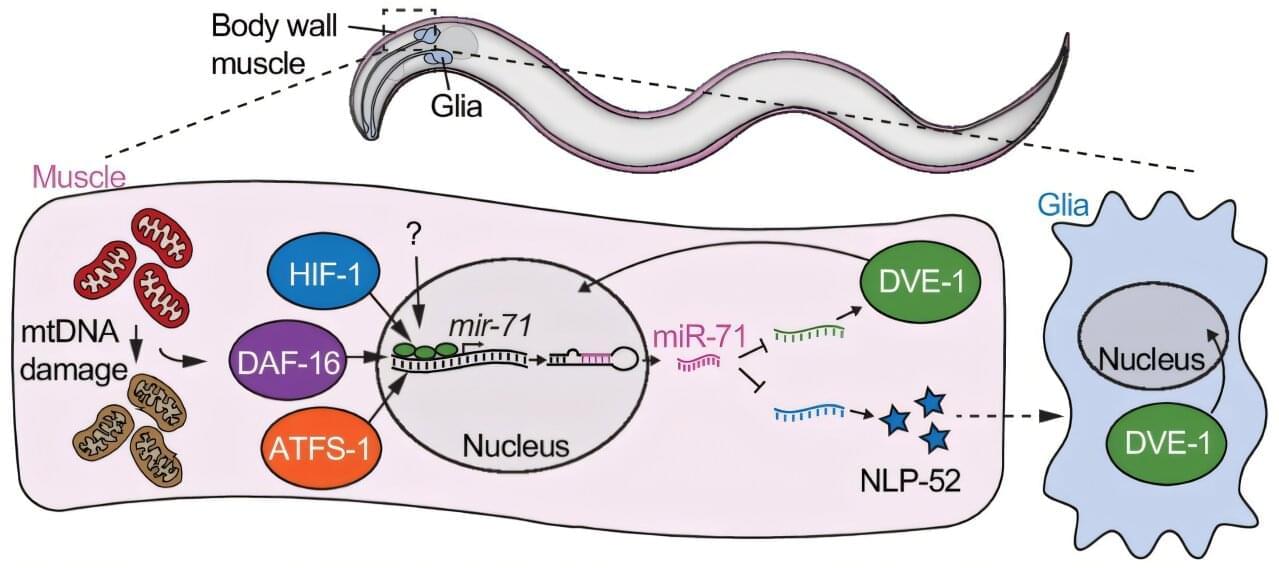
University of Queensland researchers say the discovery of a new stress reduction role for a naturally occurring molecule in the body could lead to new drugs and treatment for metabolic disorders and aging.
Professor Steven Zuryn, a molecular geneticist from UQ’s Queensland Brain Institute, was part of a team that found that very small RNA molecules, called microRNAs, bind to genes and prevent them from being over-activated.
MicroRNAs were discovered in C. elegans about 30 years ago and have since been shown to be important in human health and disease. This initial discovery led to the 2024 Nobel Prize in Physiology or Medicine.
Join us on Patreon! https://www.patreon.com/MichaelLustgartenPhD
Discount Links/Affiliates:
Blood testing (where I get the majority of my labs): https://www.ultalabtests.com/partners/michaellustgarten.
Blood testing with LifeExtension.com: https://www.anrdoezrs.net/click-101614996-15750394
At-Home Metabolomics: https://www.iollo.com?ref=michael-lustgarten.
Use Code: CONQUERAGING At Checkout.
Clearly Filtered Water Filter: https://get.aspr.app/SHoPY
Epigenetic, Telomere Testing: https://trudiagnostic.com/?irclickid=U-s3Ii2r7xyIU-LSYLyQdQ6…M0&irgwc=1
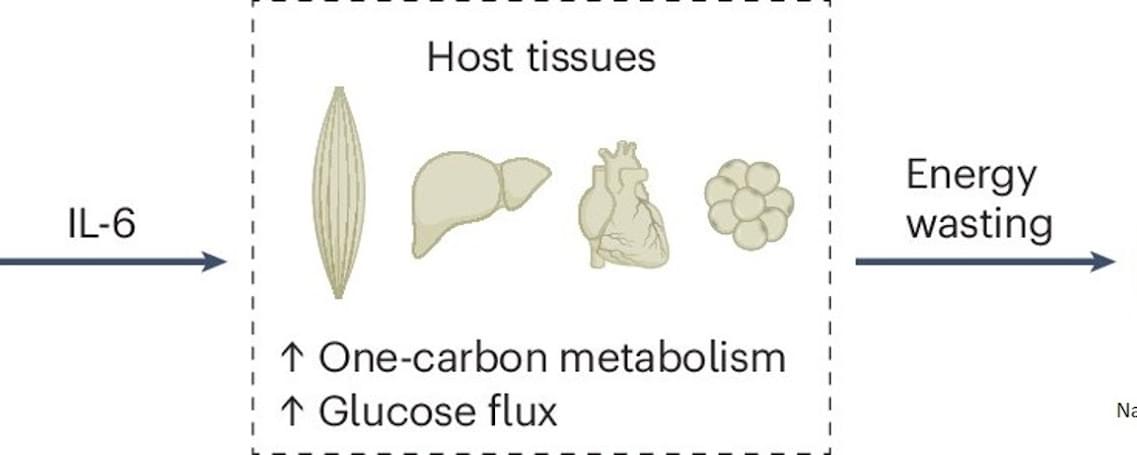
Combined metabolomics and transcriptomics analysis in eight different organs of tumor-bearing mice with and without cachexia allowed researchers to create metabolic signatures typical of cancer-associated weight loss. High-throughput analyses identified a cachexia-specific metabolic and genetic signature that provides insight into the progression of these metabolic changes.
The researchers found that all organs showed increased activation of the so-called “one carbon cycle”, a biochemical process essential for the synthesis of nucleotides, amino acids, and cell regeneration. Products of this cycle, such as sarcosine or dimethylglycine, could potentially serve as biomarkers for cachexia in the future.
The study also revealed that hyperactivation of the one carbon cycle in muscle is associated with increased glucose metabolism (glucose hypermetabolism) and muscle atrophy. Early experiments suggest that inhibiting this process could prevent muscle loss. Comparative analyses across eight different mouse tumor models (lung, colon, and pancreatic cancer) confirmed that the one carbon signature represents a universal cachexia signature, independent of cancer type.
Currently, there is no approved drug for cancer cachexia in Germany. New approaches are being explored to address cancer-related appetite loss. This study provides the first evidence of how metabolism itself could potentially be normalized. Early experiments in cell cultures show that interventions targeting the one carbon cycle can have positive effects. sciencenewshighlights ScienceMission.
Cachexia is a metabolic disorder that causes uncontrolled weight loss and muscle wasting in chronic diseases and cancer. A new study shows that cachexia affects more than just muscles. Numerous organs respond in a coordinated manner, ultimately contributing to muscle loss. Analysis of metabolome and transcriptome data, along with glucose tracing in tumor-bearing mouse models, identified a novel mechanism that plays a key role in cancer-associated weight loss.
A loss of 10% of body weight within six months – what may sound desirable in some contexts – often causes uncertainty and frustration in cancer patients with cachexia, as they are unable to maintain or gain body weight despite wanting to. Cachexia (from the Greek kakós, “bad,” and héxis, “condition”) affects 50–80% of all cancer patients, reduces quality of life, diminishes the effectiveness of cancer therapies, and increases mortality.
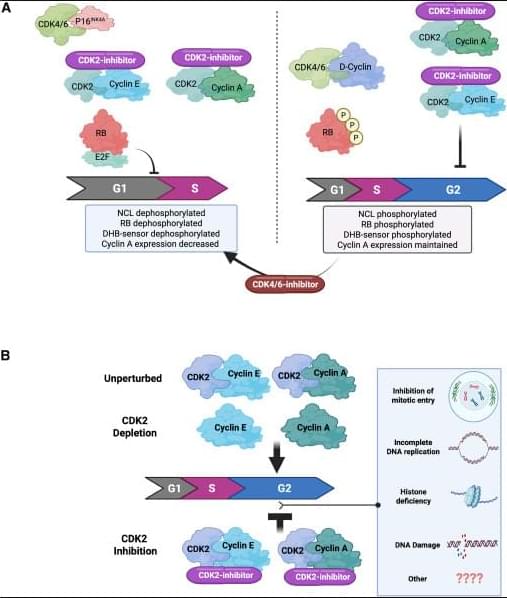
How CDK2 inhibitors halt cancer cell division👇
✅G1 arrest in p16INK4A-high, cyclin E–high tumors (A, left). In tumors with high p16INK4A and cyclin E, catalytic CDK2 inhibitors block phosphorylation of key CDK2 substrates, including RB and nucleolin, and reduce signals from CDK activity sensors. This suppresses E2F transcriptional activity, leading to reduced cyclin A expression and failure to enter S phase. The net result is a G1 cell-cycle arrest, driven by effective shutdown of the RB–E2F axis.
✅4N accumulation when p16INK4A is absent (A, right). In tumors lacking p16INK4A, CDK2 catalytic inhibition alone does not efficiently block RB phosphorylation or early G1 molecular events. Instead, cells continue through S phase and accumulate with 4N DNA content, indicating arrest later in the cycle (post-replication). In this context, adding a CDK4/6 inhibitor can mimic the p16INK4A state, restore RB dephosphorylation, repress E2F, and shift cells toward a G1 arrest, highlighting the importance of dual CDK control of RB.
✅Catalytic inhibition vs genetic depletion of CDK2 (B). Genetic loss of CDK2 is often tolerated because cells can compensate using cyclin A–CDK1 to complete G2/M. In contrast, catalytic CDK2 inhibitors trap CDK2 in inactive complexes with cyclins, which may interfere with normal handoff to CDK1 and other cell-cycle processes. This leads to accumulation of cells with 4N DNA content, reflecting a block after DNA replication.
✅Why the outcomes differ. These findings suggest that CDK2 has roles beyond simple kinase activity—its inactive, cyclin-bound state under catalytic inhibition may disrupt network dynamics differently than complete protein loss. The precise mechanisms of the 4N arrest are still being investigated and may involve defects in S/G2 transitions, replication stress responses, or mitotic entry control.
✅Therapeutic implication. Tumor response to CDK2 inhibitors depends strongly on p16INK4A status, cyclin E levels, and RB pathway integrity. This supports combination strategies (CDK2 + CDK4/6 inhibition) in selected cancers and emphasizes the need for biomarker-guided patient stratification.