Vaults are mysterious cellular structures that can be genetically engineered to store information about a cell’s history.

The study, published in Genomic Psychiatry, identified how stress hormones activate specific RNA molecules called long noncoding RNAs, or IncRNAs, that interact with the gene-silencing complex PRC2, turning off genes that are vital to communication between neurons. In essence, these IncRNAs act like “switches,” turning off functionality for more than 3,000 genes, many of which support neurotransmitter signaling and other processes that are essential for healthy brain functioning. The study specifically discovered 79 IncRNAs that were significantly altered under stress conditions.
While scientists have long understood that stress hormones send signals to the brain that affect gene functionality, it was previously unknown as to exactly how these signals create long-lasting changes inside cells. The study, led by Yogesh Dwivedi, Ph.D., Distinguished Professor and Elesabeth Ridgely Shook Endowed Chair in the Department of Psychiatry and Behavioral Neurobiology, and co-director of UAB Depression and Suicide Center, uncovers how lncRNAs associate with a molecule called polycomb repressive complex 2, or PRC2, to modify chromatin following activation of the glucocorticoid receptor, or GR — the cell’s master regulator of stress response. Chromatin is important in relaying messages from the external environment, including stressful conditions, to alter the genetic composition, a process known as epigenetics.
“As chronic stress is a major risk factor for conditions like major depressive disorder, this newly uncovered link between stress hormones and IncRNA gene silencing could potentially lead to more targeted mental health treatments,” Dwivedi said. “In fact, stress-induced changes in chromatin structure have been implicated in a range of psychiatric and neurodegenerative conditions.”
IncRNAs act like “switches,” turning off functionality for more than 3,000 genes that are essential for healthy brain functioning. A recent groundbreaking study from researchers at the University of Alabama at Birmingham highlights the discovery of a molecular link between stress hormones and changes in brain cell communication, which could open the door for new treatments to address depression and other psychiatric conditions.
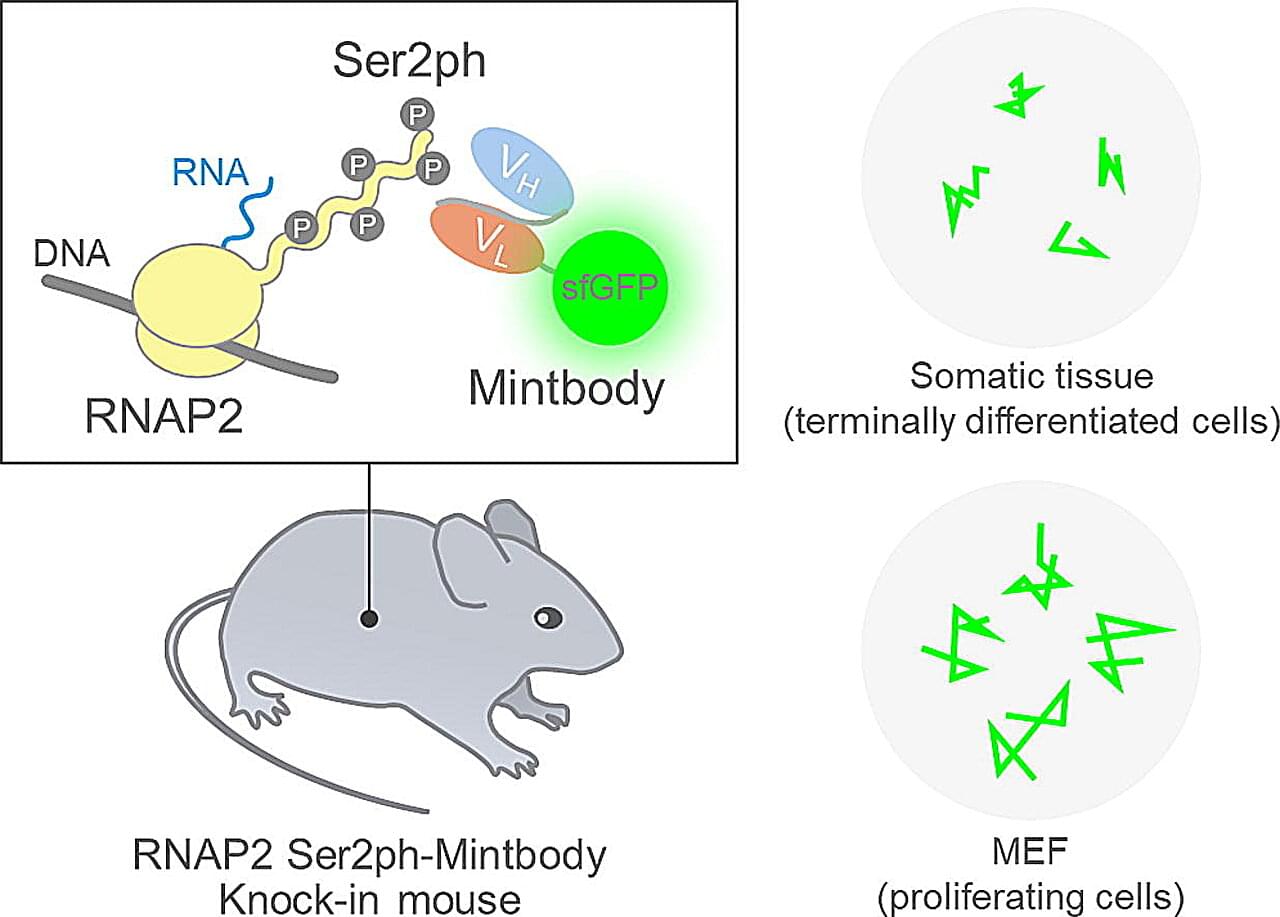
DNA can be thought of as a vast library that stores all genetic information. Cells do not use this information all at once. Instead, they copy only the necessary parts into RNA, which is then used to produce proteins—the essential building blocks of life. This copying process is called transcription, and it is carried out by a molecule known as RNA polymerase II.
When RNA polymerase II begins actively transcribing DNA, a specific site called Ser2 on its tail region is marked with a small chemical group known as a phosphate. This phosphate acts as a sign that transcription is in progress. Until now, observing this sign required stopping cellular activity and chemically treating the cells to visualize the phosphate. As a result, it was impossible to see how transcription changes dynamically in living cells.
To overcome this limitation, a research team led by Professor Hiroshi Kimura at Institute of Science Tokyo (Science Tokyo) chose a different approach. Instead of freezing cells at a single moment, they aimed to track transcription continuously without stopping cellular activity.
The metabolites associated with type 2 diabetes were also found to be genetically linked to clinical traits and tissue types that are relevant to the disease. Furthermore, the team developed a unique signature of 44 metabolites that improved prediction of future risk of type 2 diabetes. ScienceMission sciencenewshighlights.
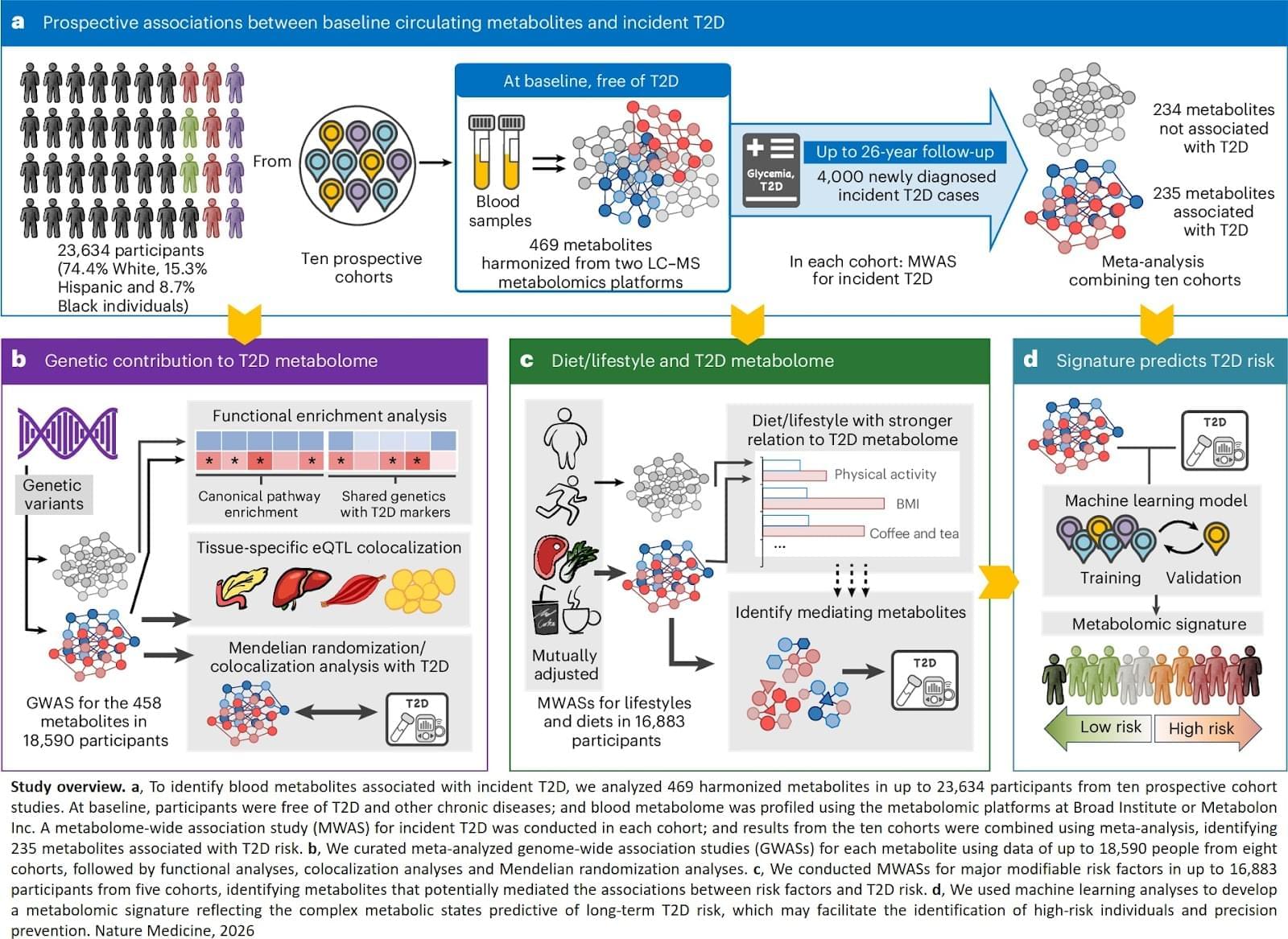
Diabetes, a metabolic disease, is on the rise worldwide, and over 90 percent of cases are type 2 diabetes, where the body does not effectively respond to insulin. Researchers identified metabolites (small molecules found in blood generated through metabolism associated with risk of developing type 2 diabetes in the future and revealed genetic and lifestyle factors that may influence these metabolites. They also developed a metabolomic signature that predicts future risk of type 2 diabetes beyond traditional risk factors. Their results are published in Nature Medicine.
In this study, researchers tracked 23,634 individuals with diverse ethnic backgrounds across 10 prospective cohorts with up to 26 years of follow-up. These individuals were initially free of type 2 diabetes. The team analyzed 469 metabolites in blood samples, as well as genetic, diet, and lifestyle data, to see how they relate to risk of developing type 2 diabetes. Of the metabolites examined, 235 were found to be associated with a higher or lower risk of developing type 2 diabetes, 67 of which were new discoveries.
“Interestingly, we found that diet and lifestyle factors may have a stronger influence on metabolites linked to type 2 diabetes than on metabolites not associated with the disease,” said first and co-corresponding author. “This is especially true for obesity, physical activity, and intake of certain foods and beverages such as red meat, vegetables, sugary drinks, and coffee or tea. Increasing evidence suggests that these dietary and lifestyle factors are associated with greater or lower risk of type 2 diabetes. Our study revealed that specific metabolites may act as potential mediators, linking these factors with type 2 diabetes risk.”
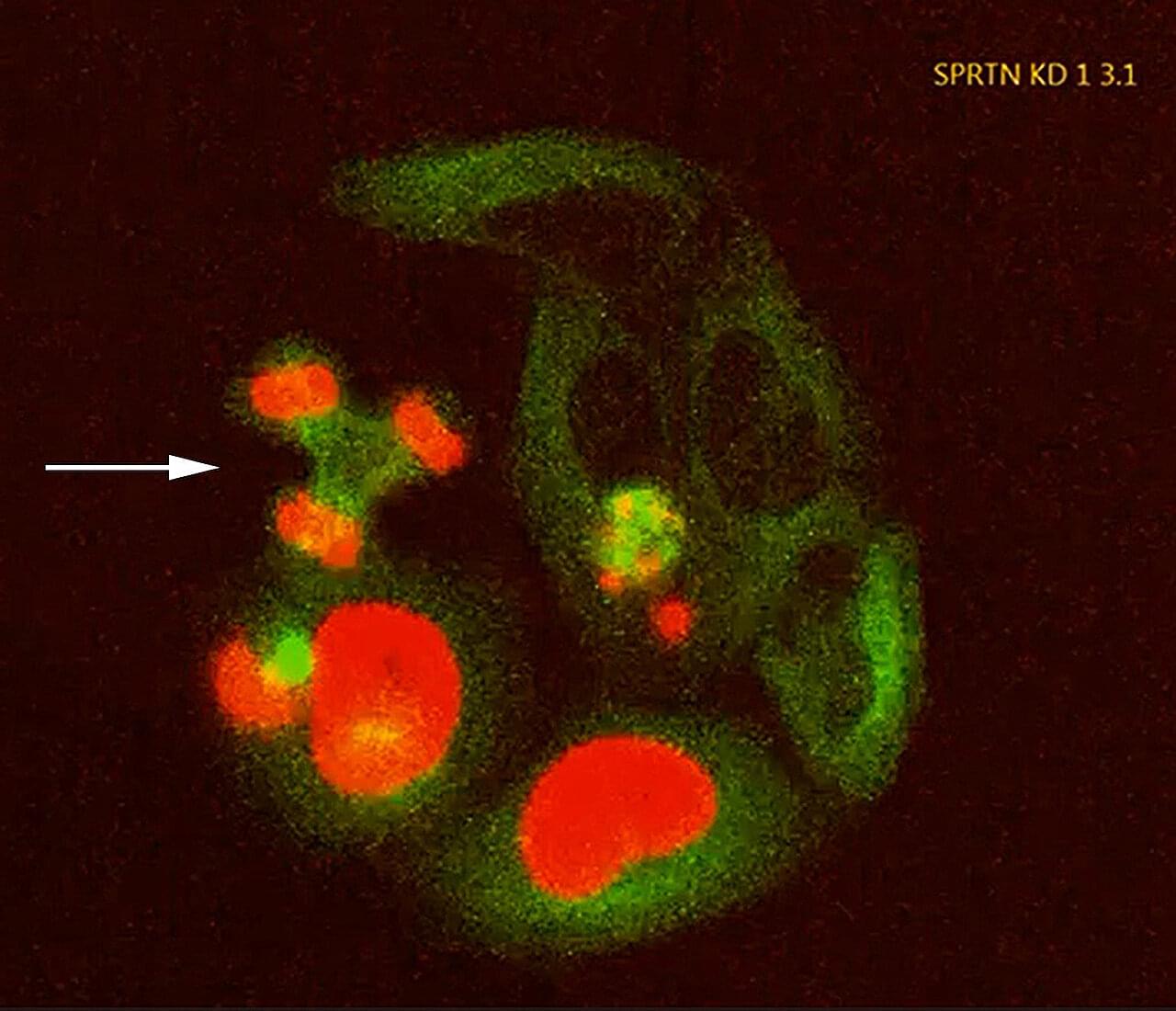
Although DNA is tightly packed and protected within the cell nucleus, it is constantly threatened by damage from normal metabolic processes or external stressors such as radiation or chemical substances. To counteract this, cells rely on an elaborate network of repair mechanisms. When these systems fail, DNA damage can accumulate, impair cellular function, and contribute to cancer, aging, and degenerative diseases.
One particularly severe form of DNA damage are the so-called DNA–protein crosslinks (DPCs), in which proteins become attached to DNA. DPCs can arise from alcohol consumption, exposure to substances such as formaldehyde or other aldehydes, or from errors made by enzymes involved in DNA replication and repair. Because DPCs can cause serious errors during cell division by stalling DNA replication, DNA–protein crosslinks pose a serious threat to genome integrity.
The enzyme SPRTN removes DPCs by cleaving the DNA-protein crosslinks. SPRTN malfunctions, for example as a result of mutations, may predispose individuals to developing bone deformities and liver cancer in their teenage years. This rare genetic disorder is known as Ruijs-Aalfs syndrome. Its underlying mechanism remains poorly understood, and there are no specific therapies.

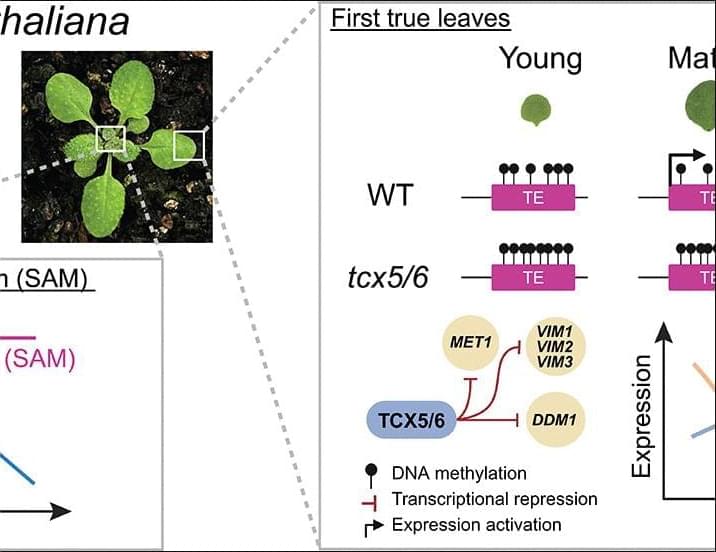
Plants display a wide range of life spans and aging rates. Although dynamic changes to DNA methylation are a hallmark of aging in mammals, it is unclear whether similar molecular signatures reflect rates of aging and organism life span in plants. In this work, we show that the short-lived model plant Arabidopsis thaliana exhibits a loss of epigenetic integrity during aging, which causes DNA methylation decay and the expression of transposable elements. We show that the rate of epigenetic aging can be manipulated by extending or curtailing life span and that shoot apical meristems are protected from these epigenetic changes. We demonstrate that a program of transcriptional repression suppresses DNA methylation maintenance pathways during aging and that mutants of this program display a complete absence of epigenetic decay while physical aging remains unaffected.
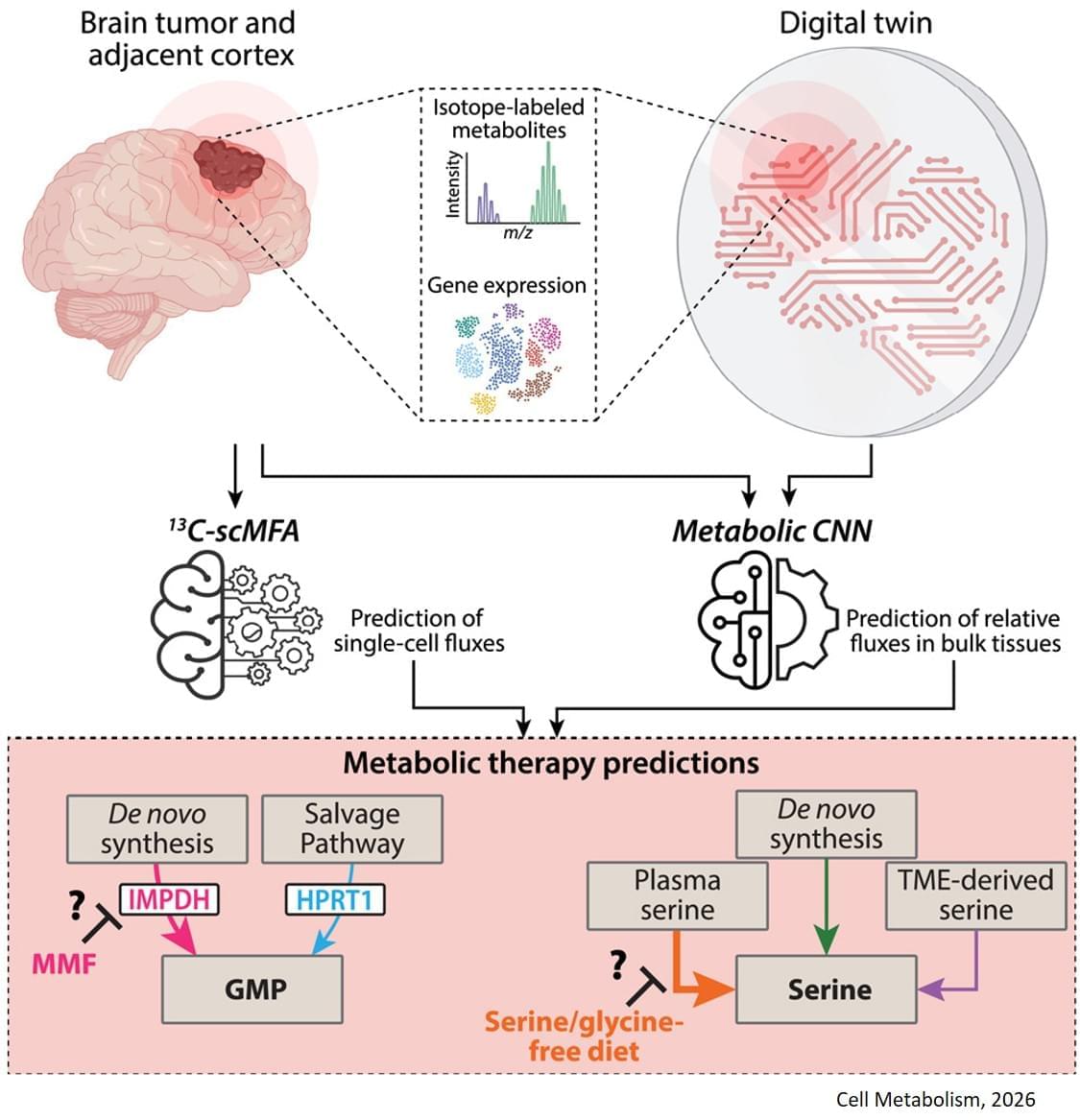
The study, published in Cell Metabolism, builds on previous research showing that some gliomas can be slowed down through the patient’s diet. If a patient isn’t consuming certain protein building blocks, called amino acids, then some tumors are unable to grow. However, other tumors can produce these amino acids for themselves, and can continue growing anyway. Until now, there was no easy way to tell which patients would benefit from dietary restrictions.
The digital twin’s ability to map metabolic activity in tumors also helped determine whether a drug that prevents tumors from producing a building block for replicating and repairing DNA would work, as some cells can obtain that molecule from their environments.
To overcome challenges in mapping tumor metabolism inside the brain, the team developed a computer-based “digital twin” that can predict how an individual patient’s brain tumor will react to each treatment.
“Typically, metabolic measurements during surgeries to remove tumors can’t provide a clear picture of tumor metabolism—surgeons can’t observe how metabolism varies with time, and labs are limited to studying tissues after surgery. By integrating limited patient data into a model based on fundamental biology, chemistry and physics, we overcame these obstacles,” said a co-corresponding author of the study.
The digital twin uses patient data obtained through blood draws, metabolic measurements of the tumor tissue and the tumor’s genetic profile. The digital twin then calculates the speed at which the cancer cells consume and process nutrients, known as metabolic flux.
“This is the first time a machine learning and AI-based approach has been used to measure metabolic flux directly in patient tumors,” said a co-first author of the study.
The researchers built a type of deep learning model called a convolutional neural network and trained it on synthetic patient data, generated based on known biology and chemistry and constrained by measurements from eight patients with glioma who were infused with labeled glucose during surgery. By comparing their computer models with different data from six of those patients, they found the digital twins could predict metabolic activity with high accuracy. In experiments conducted on mice, the team confirmed that the diet only slowed tumor growth in mice that the digital twin had identified as good candidates for the treatment.
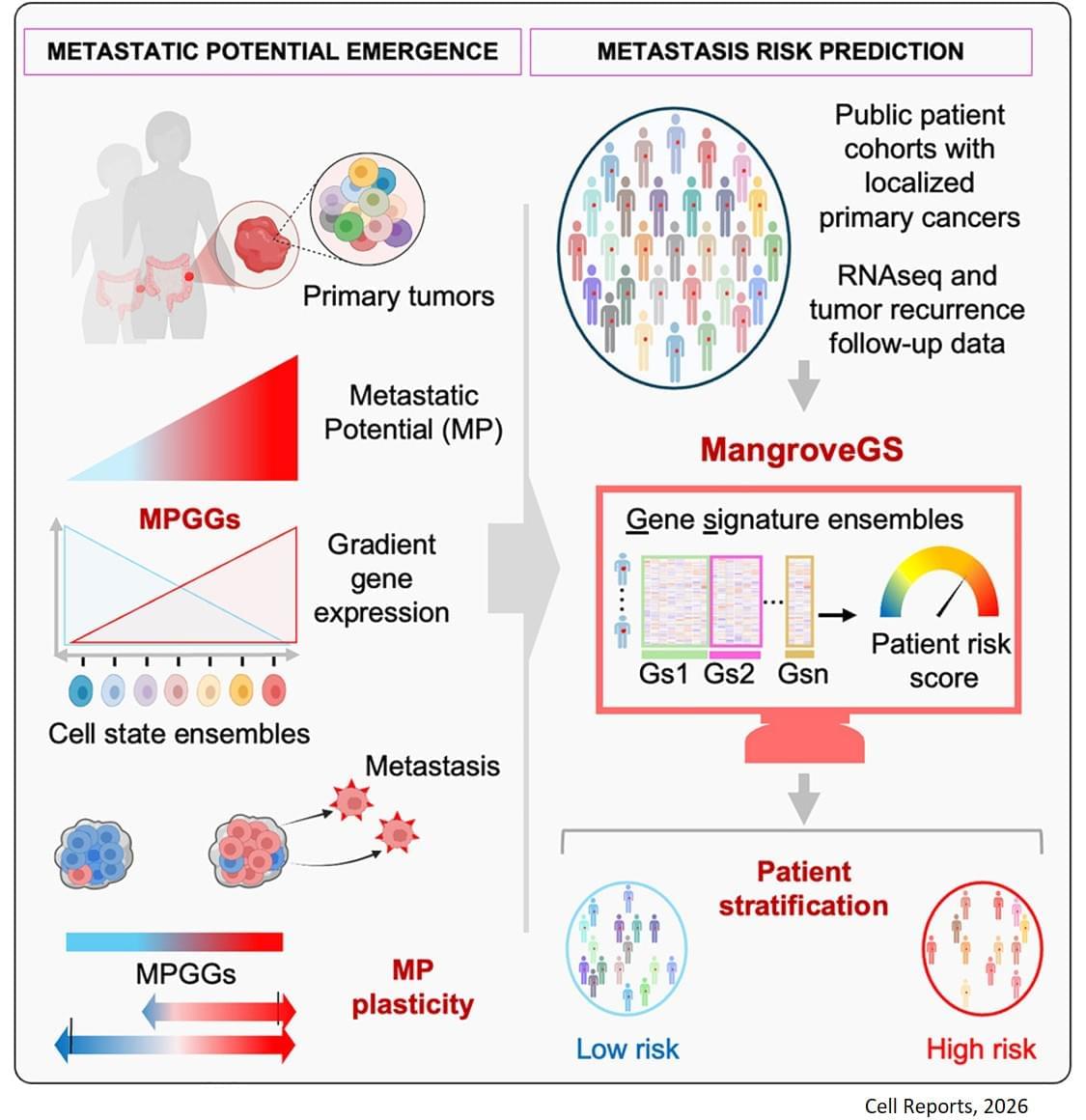
Metastasis remains the leading cause of death in most cancers, particularly colon, breast and lung cancer. Currently, the first detectable sign of the metastatic process is the presence of circulating tumor cells in the blood or in the lymphatic system. By then, it is already too late to prevent their spread. Furthermore, while the mutations that lead to the formation of the original tumors are well understood, no single genetic alteration can explain why, in general, some cells migrate and others do not.
“The difficulty lies in being able to determine the complete molecular identity of a cell – an analysis that destroys it – while observing its function, which requires it to remain alive,” explains the senior author. “To this end, we isolated, cloned and cultured tumor cells,” adds a co-first author of the study. “These clones were then evaluated in vitro and in a mouse model to observe their ability to migrate through a real biological filter and generate metastases.”
The analysis of the expression of several hundred genes, carried out on about thirty clones from two primary colon tumors, identified gene expression gradients closely linked to their migratory potential. In this context, accurate assessment of metastatic potential does not depend on the profile of a single cell, but on the sum of interactions between related cancer cells that form a group.
The gene expression signatures obtained were integrated into an artificial intelligence model developed by the team. “The great novelty of our tool, called ‘Mangrove Gene Signatures (MangroveGS)’, is that it exploits dozens, even hundreds, of gene signatures. This makes it particularly resistant to individual variations,” explains another co-first author of the study. After training, the model achieved an accuracy of nearly 80% in predicting the occurrence of metastases and recurrence of colon cancer, a result far superior to existing tools. In addition, signatures derived from colon cancer can also predict the metastatic potential of other cancers, such as stomach, lung and breast cancer.
After training, the model achieved an accuracy of nearly 80% in predicting the occurrence of metastases and recurrence of colon cancer, a result far superior to existing tools. In addition, signatures derived from colon cancer can also predict the metastatic potential of other cancers, such as stomach, lung and breast cancer.
Thanks to MangroveGS, tumor samples are sufficient: cells can be analysed and their RNA sequenced at the hospital, then the metastatic risk score quickly transmitted to oncologists and patients via an encrypted Mangrove portal that has analysed the anonymised data.
“This information will prevent the overtreatment of low-risk patients, thereby limiting side effects and unnecessary costs, while intensifying the monitoring and treatment of those at high risk,” adds the senior author. “It also offers the possibility of optimising the selection of participants in clinical trials, reducing the number of volunteers required, increasing the statistical power of studies, and providing therapeutic benefits to the patients who need it most.” ScienceMission sciencenewshighlights.

In Ruijs-Aalfs progeria syndrome, patients experience accelerated aging and liver cancer.
Now, scientists showed that mutations in a certain gene prevent cells from repairing DNA damage during mitosis, triggering inflammatory immune responses that may fuel premature aging.
Read more.
Researchers have shown that harmful bonds between protein and DNA fuel immune attack in progeria. Pumping up a protein that cuts these bonds could prevent symptoms.