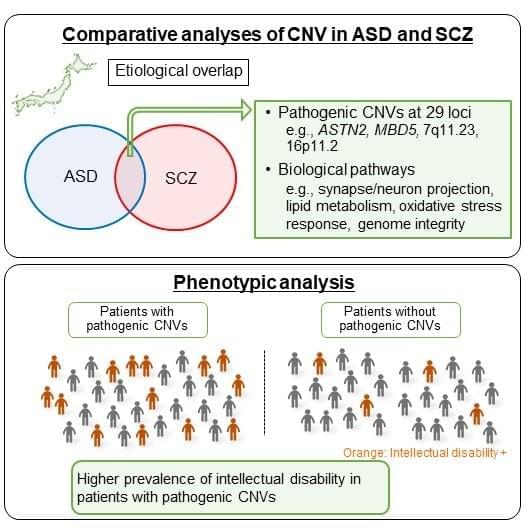
Tune Therapeutics, a Durham biotechnology startup co-founded by a Duke professor, announced the completion of its Series B fundraising round on Jan. 12, in which it raised $175 million to support clinical trials for its epigenome editor.
The company will use the funding to advance clinical trials for Tune-401, the epigenetic silencing drug for treating chronic Hepatitis B — a viral infection that damages the liver and affects millions globally. The investment will also support the development of various other therapies, including additional gene, cell and regenerative therapy programs.
“The goal is to epigenetically repress the virus to prevent it from being able to replicate and make the viral proteins that it would normally produce,” said Charles Gersbach, John W. Strohbehn distinguished professor of biomedical engineering and cofounder of Tune Therapeutics.