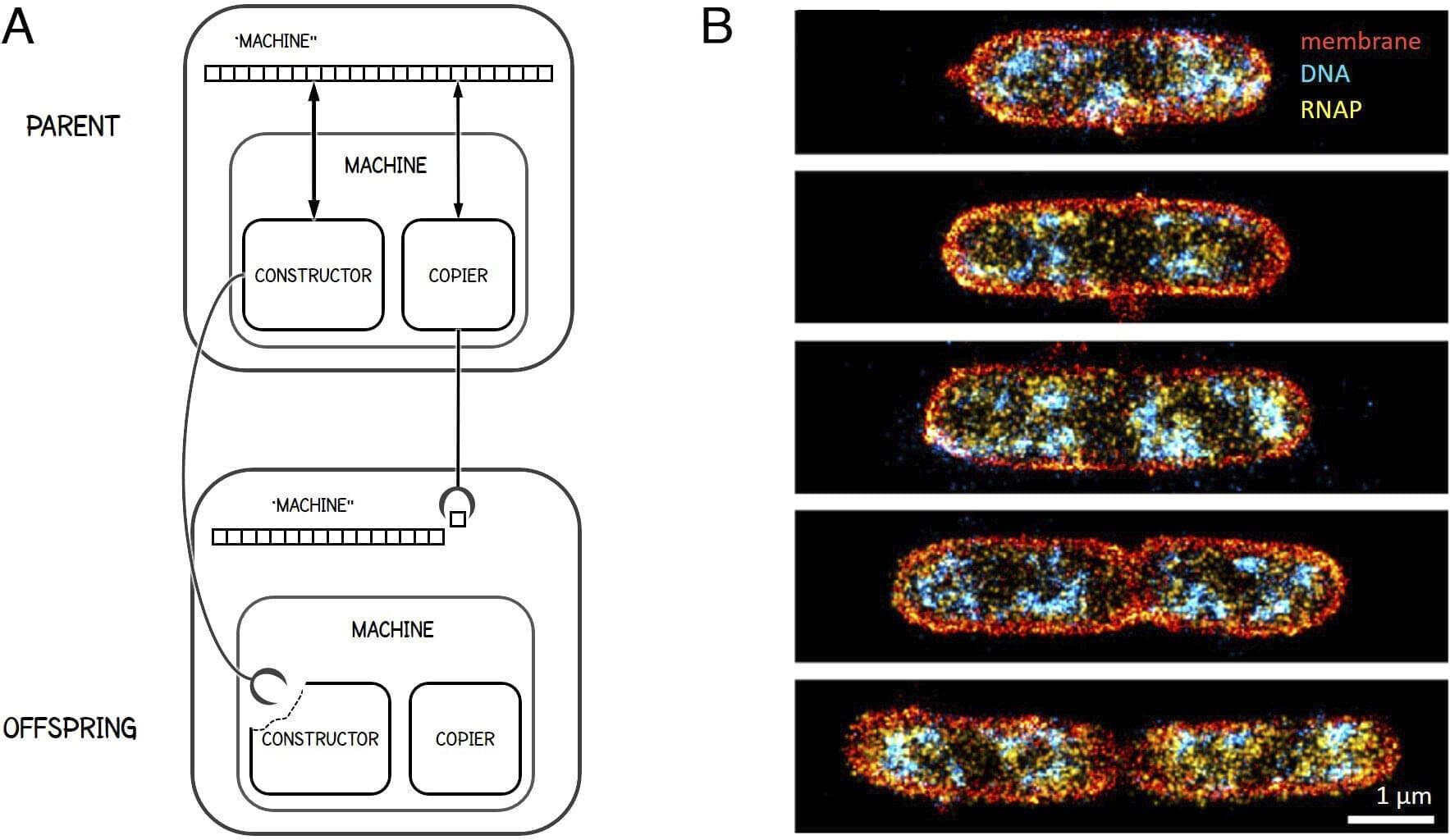
Living matter remains the quintessential puzzle of biological sciences, a question that embodies the intricate complexity and stunning diversity of life forms. A new study suggests that one viable approach to address this extreme complexity is to conceptualize living matter as a cascade of machines producing machines.
This cascade illustrates how cells are composed of smaller submachines, reaching down to the atomic level where molecular machines, such as ion pumps and enzymes, operate. In the other direction, it explains how cells self-organize into larger systems, such as tissues, organs, and populations, cumulating into the biosphere.
This new conceptual framework is a fruit of collaboration between Professors Tsvi Tlusty from the Department of Physics at Ulsan National Institute of Science and Technology (UNIST), South Korea, and Albert Libchaber from the Center for Physics and Biology at Rockefeller University, New York. The study was inspired by the seventeenth-century polymath Gottfried Leibniz, who noted that “the machines of nature, that is living bodies, are still machines in their smallest parts, to infinity.”