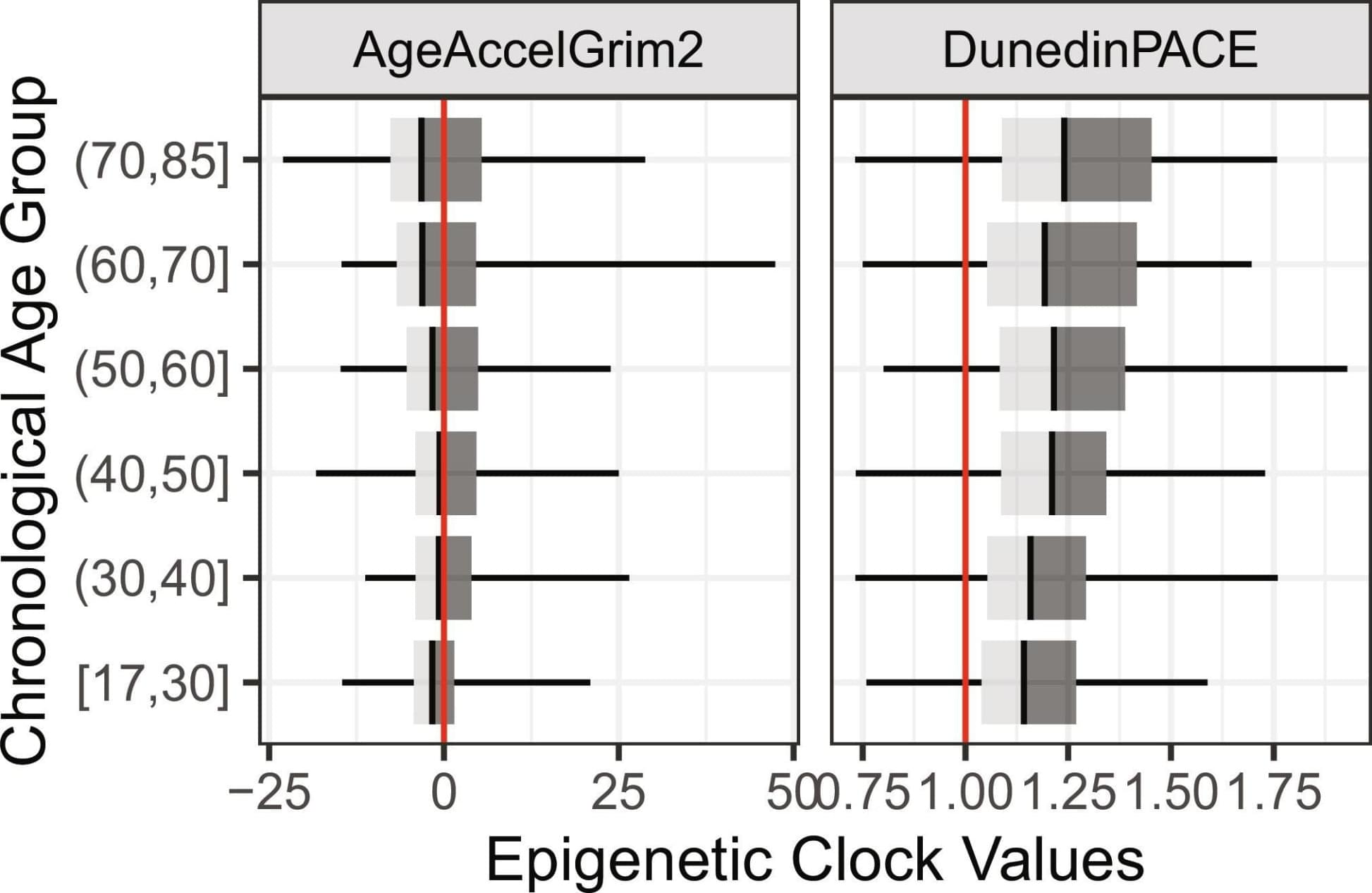
Negative social ties, or “hasslers,” are pervasive yet understudied components of social networks that may accelerate biological aging and morbidity. Using ego-centric network data and DNA methylation-based biological aging clocks (i.e., DunedinPACE and age-accelerated GrimAge2) from saliva from a state representative probability sample in Indiana, we examine how negative social ties are associated with accelerated biological aging and a broad range of health outcomes, including inflammation and multimorbidity. Negative relationships are not rare within close relationships, as nearly 30% of individuals report having at least one hassler in their network. These hasslers tend to occupy peripheral network positions and are more likely to be connected through weak, uniplex ties. Importantly, exposure to negative social ties follows patterns of social and health vulnerability, with women, daily smokers, people in poorer health, and those with adverse childhood experiences more likely to report having hasslers in their networks. Having more hasslers is associated with accelerated biological aging in both rate and cumulative burden: Each additional hassler corresponds to approximately 1.5% faster pace of aging and roughly 9 mo older biological age. Moreover, not all hasslers exert the same influence; kin and nonkin hasslers show detrimental associations, whereas spouse hasslers do not. Finally, a greater number of hasslers is associated with multiple adverse health outcomes beyond epigenetic aging. These findings together highlight the critical role of negative social ties in biological aging as chronic stressors and the need for interventions that reduce harmful social exposures to promote healthier aging trajectories.