The FDA had promised to render a decision on the approval of aducanumab by March 7. The process is now being extended to June 7.




For centuries, scientists have tried to work on formulas to extend human life. While scientific and medical advancements have pushed the average human life expectancy to 71 years worldwide, researchers are still trying to find out if it can be extended further. Now, a group of scientists is leaning on artificial intelligence (AI) to combine conventional medicine to treat diseases and prevent them to extend longevity.
Conventional medicine has been the go-to tool for scientists to treat diseases and effectively extend life expectancy. But it has limitations too. While it can treat diseases, it’s not very effective in detecting early signs of an ailment that can reduce life expectancy. But combining AI, fundamental research and medicine could bring about a change in the research of extending human life. Dubbed as Longevity medicine, the scientists — Alex Zhavoronkov, Evelyne Yehudit Bischof and Kai-Fu Lee — proposed a framework. Their article was published in the journal Nature Aging.
Dr Zhavoronkov is a computer scientist with a doctorate in biophysics. Bischof is a practicing doctor who is researching on aging and gerooncology at the University Hospital Basel in Switzerland. Lee is known for his expertise in AI.
Scientists have proposed an artificial intelligence-based framework that can analyze millions of parameters and help fight aging.
This is the first part of the interview with Harold Katcher in Modern Healthspan YouTube channel.
Dr. Harold Katcher is a professor of Biology at the University of Maryland. He has been a pioneer in the field of cancer research, in the development of modern aspects of gene hunting and sequencing. He carries expertise in bioinformatics, chronobiology, and biotechnology. Dr. Katcher is currently working in the capacity of Chief Technical Officer at Nugenics Research exploring rejuvenation treatments in mammals.
In May 2020 there was a paper published on biorxiv about the rejuvenation of rats by over 50%. We did a review of the paper which you can find linked to above. In this interview series we talk with Dr. Harold Katcher, one of the main authors of the paper about the experiment, the steps to get validation, commercialization and how the results fit into his theories of aging.
In this video we talk about the actual experiment and some of the results that Dr. Katcher and his team saw.
The paper can be found here https://www.biorxiv.org/content/10.1101/2020.05.07.082917v1.full.
If you would like to support our channel, we’d love a coffee…thank you! https://www.buymeacoffee.com/mhealthspan.

An international team of researchers has used modeling techniques borrowed from chemistry applications to create a new kind of city simulator. In their paper published in the journal Proceedings of the Royal Society A, the group describes using their models to create simulations of of COVID-19 spread for two real-world cities: Birmingham England and Bogota Columbia.
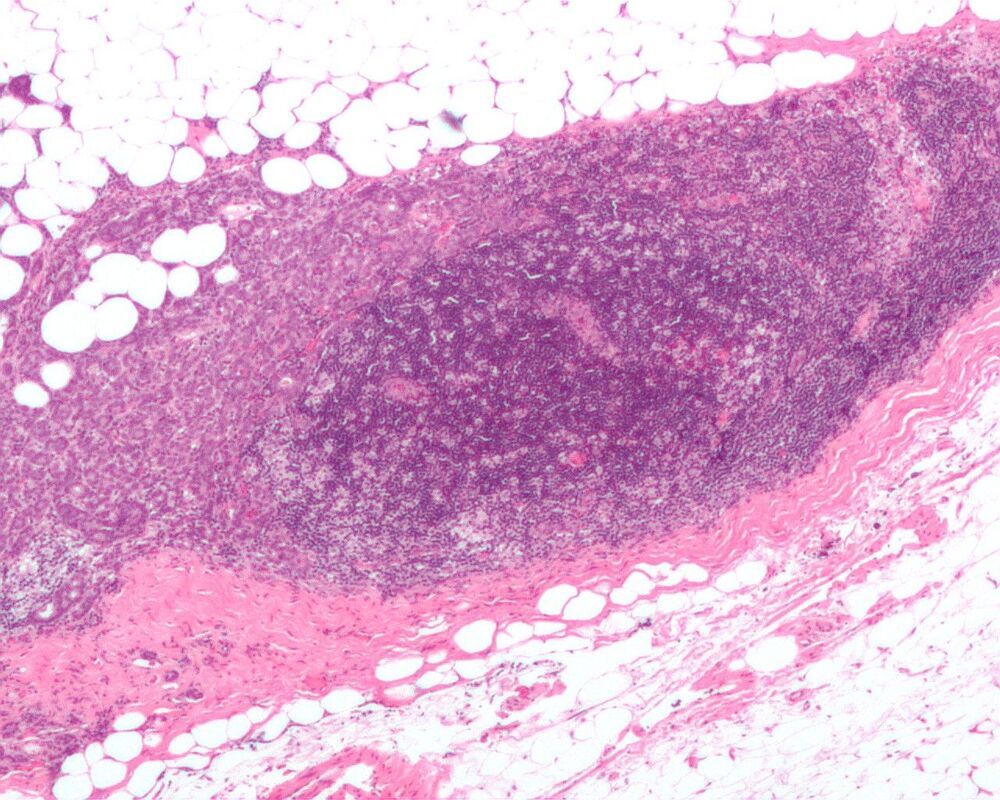
A team of researchers with members from institutions in the U.S., Sweden and Taiwan has developed an artificial intelligence system for predicting breast cancer years before tumors appear. In their paper published in the journal Science Translational Medicine, the group describes how they developed and trained their system and how well it worked when tested.

Gearing up for the interview with Harold Katcher!
Epigenetic age reversed by 54%. Scientific trial by Horvath Clock.
In this video we will discuss a paper entitled “Reversing age: dual species measurement of epigenetic age with a single clock”.
The paper can be found here https://www.biorxiv.org/content/10.1101/2020.05.07.082917v1.full.
The paper is a preprint available on bioXriv on a study showing the reversal of age in rats through exchange of blood plasma. Blood plasma of older rats was exchanged for that of younger rats which lead to the older rats having a reduced epigenetic age and many improved biomarkers, including reduced inflammation.
The age of the rats’ tissue was assessed by Dr Steve Horvath using 6 separate clocks which covered individual tissue as well as a pan tissue clocks.
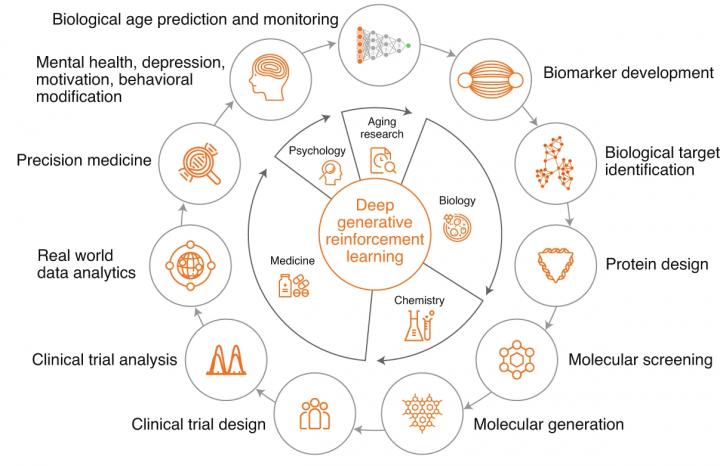
The international team of artificial intelligence experts and medical doctors propose a framework for the application of next-generation AI to extend human longevity.
Deep Longevity Ltd.
An anthropologist dives into the world of genetic engineering to explore whether gene-editing tools such as CRISPR fulfill the hope of redesigning our species for the better.
The Mutant Project: Inside the Global Race to Genetically Modify Humans by Eben Kirksey. St. Martin’s Press, November 2020. Excerpt previously published by Black Inc.
Surreal artwork in the hotel lobby—a gorilla peeking out of a peeled orange, smoking a cigarette; an astronaut riding a cyborg giraffe—was the backdrop for bombshell news rocking the world. In November 2018, Hong Kong’s Le Méridien Cyberport hotel became the epicenter of controversy about Jiankui He, a Chinese researcher who was staying there when a journalist revealed he had created the world’s first “edited” babies. Select experts were gathering in the hotel for the Second International Summit on Human Genome Editing—a meeting that had been called to deliberate about the future of the human species.