Scientists have taken a significant step toward neutralizing Epstein-Barr virus.


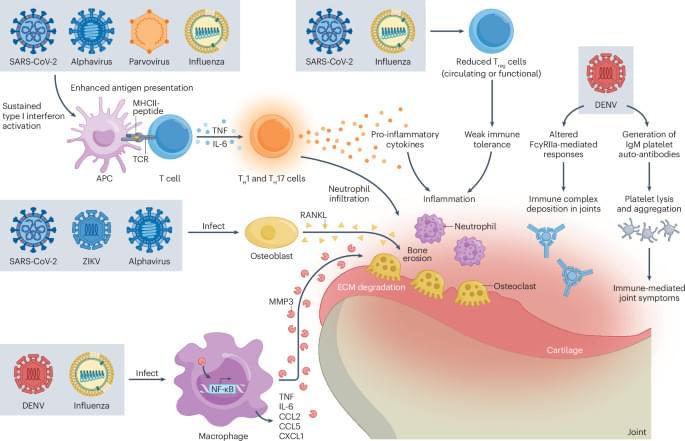
In this Review, the authors summarize the potential role of emerging viruses in autoimmune rheumatic diseases (AIRDs). They describe the association between viruses and AIRD flare ups, the putative mechanisms linking AIRD to viral infections and hormone modulation of viral pathogenesis and autoimmune diseases.

Plant-derived THC extracts significantly reduced fibromyalgia pain in patients resistant to standard treatments. [ https://www.labroots.com/trending/cannabis-sciences/30219/pl…gia-pain-2](https://www.labroots.com/trending/cannabis-sciences/30219/pl…gia-pain-2)
How can cannabis oil help alleviate fibromyalgia pain? This is what a recent study published in the Journal of Anesthesia, Analgesia, and Critical Care hopes to address as a team of researchers from Italy investigated the benefits of using tetrahydrocannabinol (THC)-based medicine options, specifically cannabis oil, for treating fibromyalgia pain. This study has the potential to help researchers, medical professionals, legislators, and the public better understand the benefits of using cannabis products for treating chronic diseases over prescriptions medications, and the steps that can be taken to implement them.
For the study, the researchers analyzed data obtained from 65 adult patients comprised of 59 women and 6 men diagnosed with fibromyalgia for a minimum of 7 years and were treated with cannabis oil therapy with a concentration of 15 percent from 2021 to 2023. The goal of the study was to ascertain the effectiveness of cannabis oil on mitigating fibromyalgia pain compared to traditional methods, specifically prescription medication. In the end, the researchers found that not only did the patients report decreased fibromyalgia pain during cannabis oil therapy, but this pain reduction was greater with younger patients.
The study notes, “While these observations should not be interpreted as evidence of effectiveness, they contribute real-world insights into tolerability and adherence in routine care. We hope they may help inform future prospective research aimed at better defining the role of cannabis oil in fibromyalgia management.”
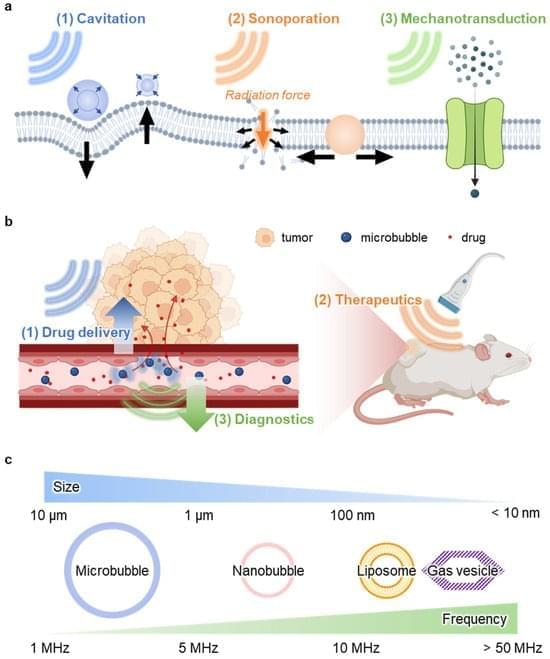
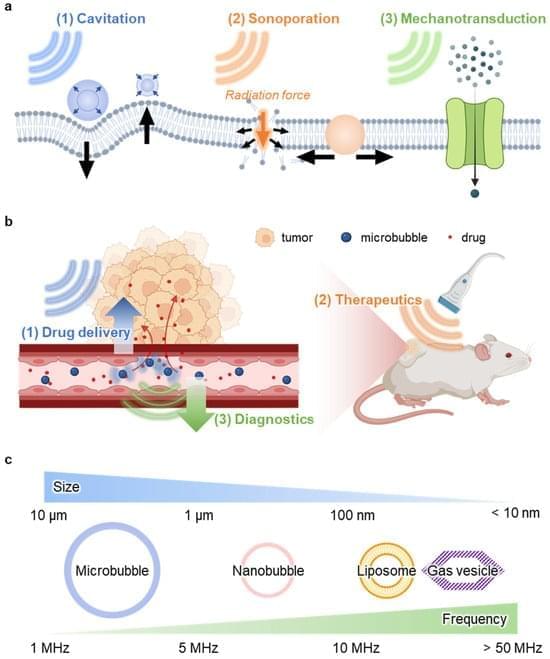
Deep tissue drug delivery is restricted by biological barriers. Ultrasound modulation offers a non-invasive solution using nanobubbles.
Read the Editor’s Choice article: mdpi.com/3348542
The cell membrane plays a critical role in regulating substance exchange, signal transduction, and energy conversion, making it essential for maintaining homeostasis and responding to environmental stimuli.
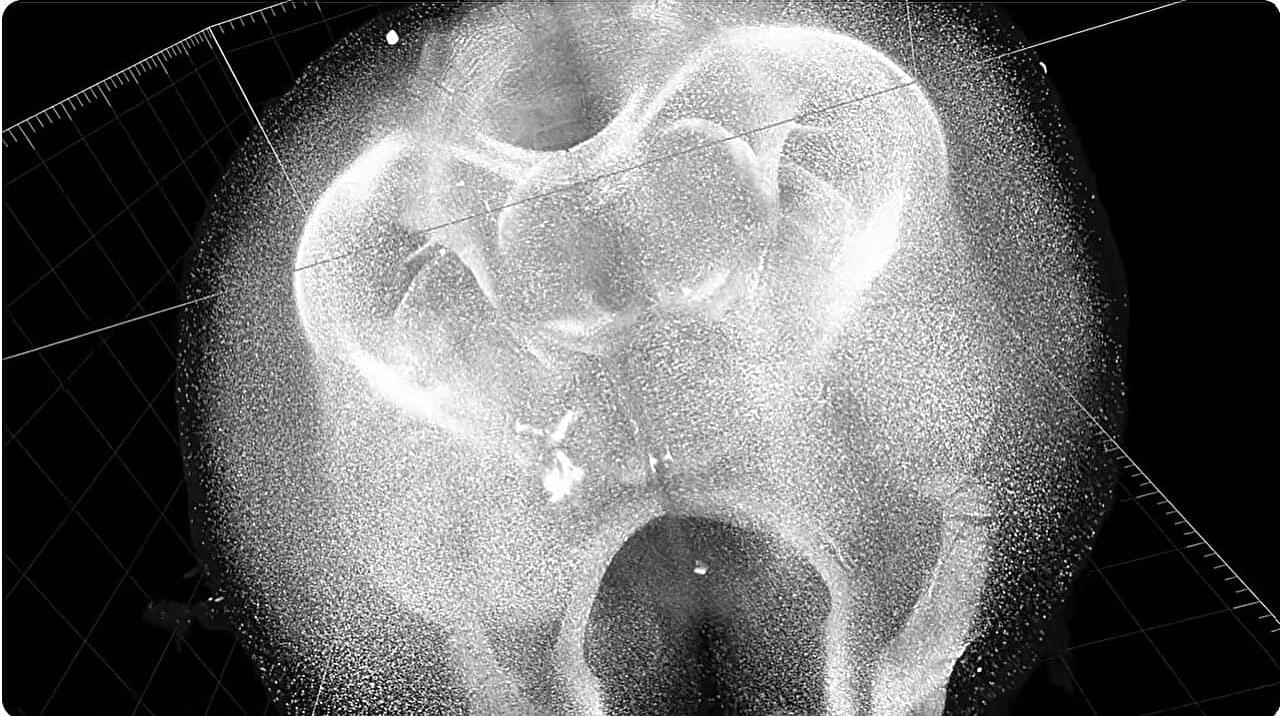
Johns Hopkins scientists say they have used 3D imaging, special microscopes and artificial intelligence (AI) programs to construct new maps of mouse brains showing a precise location of more than 10 million cells called oligodendrocytes. These cells form myelin, a protective sleeve around nerve cell axons, which speeds transmission of electrical signals and support brain health.
Published online Feb. 18 in Cell and funded by the National Institutes of Health, the maps not only paint a whole-brain picture of how myelin content varies between brain circuits, but also provide insights into how the loss of such cells impacts human diseases such as multiple sclerosis, Alzheimer’s disease and other disorders that affect learning, memory, sensory ability and movement, say the researchers. Although mouse and human brains are not the same, they share many characteristics and most biological processes.
“Our study identifies not only the location of oligodendrocytes in the brain, but also integrates information about gene expression and the structural features of neurons,” says Dwight Bergles, Ph.D., the Diana Sylvestre and Charles Homcy Professor in the Department of Neuroscience at the Johns Hopkins University School of Medicine. “It’s like mapping the location of all the trees in a forest, but also adding information about soil quality, weather and geology to understand the forest ecosystem.”
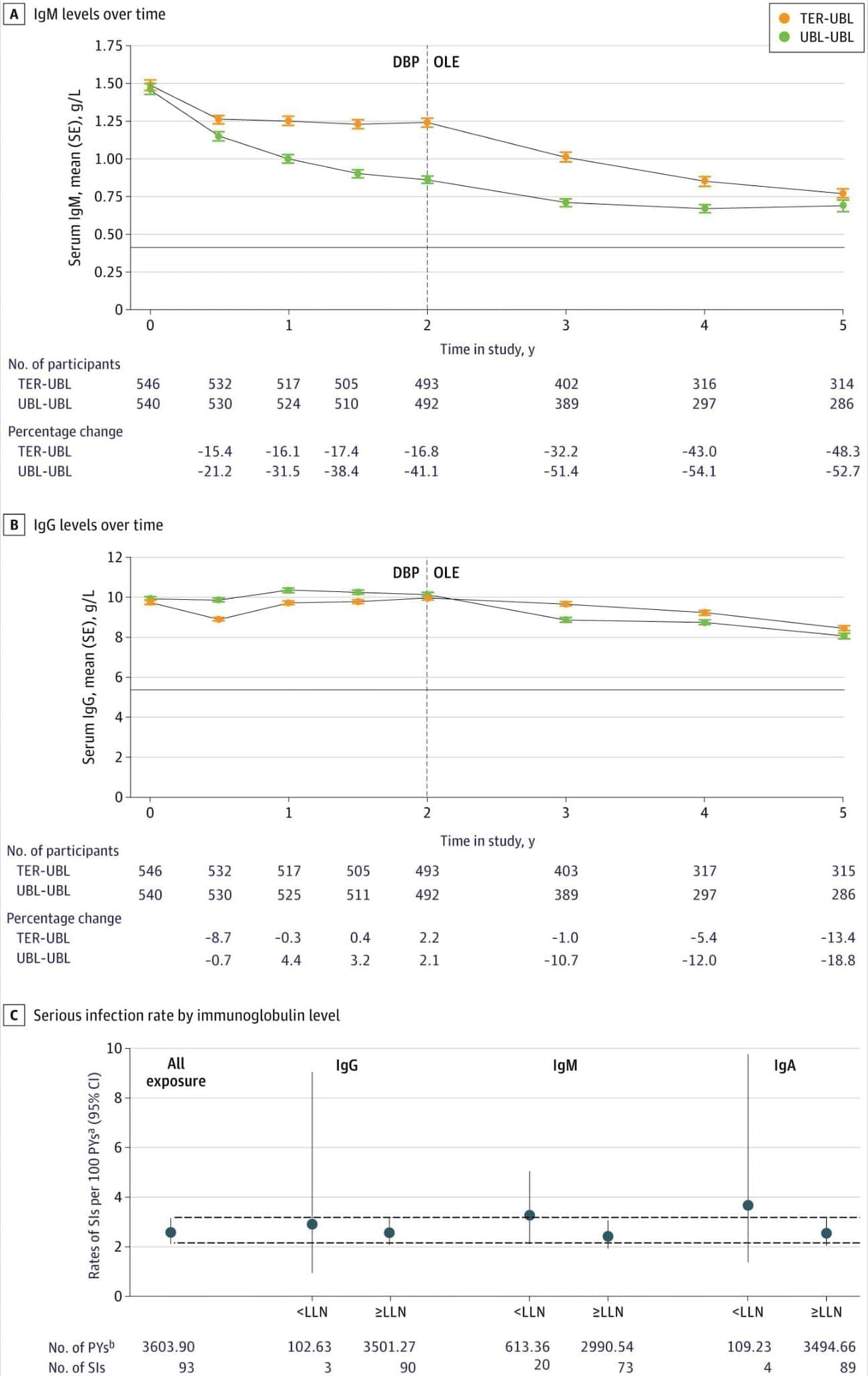
Five years of ublituximab treatment in MultipleSclerosis demonstrated sustained reduction in relapse rates and confirmed disability progression, with safety profile consistent over time.
Question What is the long-term clinical efficacy and safety of ublituximab in people with relapsing multiple sclerosis (RMS)?
Findings In this trial including 985 adults, participants treated with continuous ublituximab for up to 5 years in the open-label extension study after completion of the randomized Study to Assess the Efficacy and Safety of Ublituximab in Participants With Relapsing Forms of Multiple Sclerosis (ULTIMATE) had significantly lower annualized relapse rate and confirmed disability progression than those initially treated with teriflunomide. The overall safety profile of ublituximab remained consistent with no new safety signals emerging with prolonged treatment.
Meaning Results suggest that early initiation of ublituximab and continued treatment over a period of 5 years provided sustained clinical benefits in participants with RMS.

🧠 AI in pathology cannot succeed without pathologists. As computational pathology advances, clinical expertise remains the critical link between algorithms and real-world impact.
In this discussion, Diana Montezuma, Pathologist and Head of R&D at IMP Diagnostics, explains why pathologist involvement is essential to building AI tools that are usable, clinically relevant, and truly valuable in practice.
👉 Read the discussion:
Pathologists play a key role in AI development for pathology – providing the expertise needed to bridge data and clinical application. To discuss this role and its importance in the development of computational pathology tools, we connected with Diana Montezuma, Pathologist and Head of the R&D Unit at IMP Diagnostics.
From your perspective, what is the most important contribution that diagnosticians bring to AI and algorithm development?
Pathologists bring essential clinical expertise and practical insight to any computational pathology project. Without their involvement, such initiatives risk becoming disconnected from real-world practice and ultimately failing to deliver meaningful clinical value.
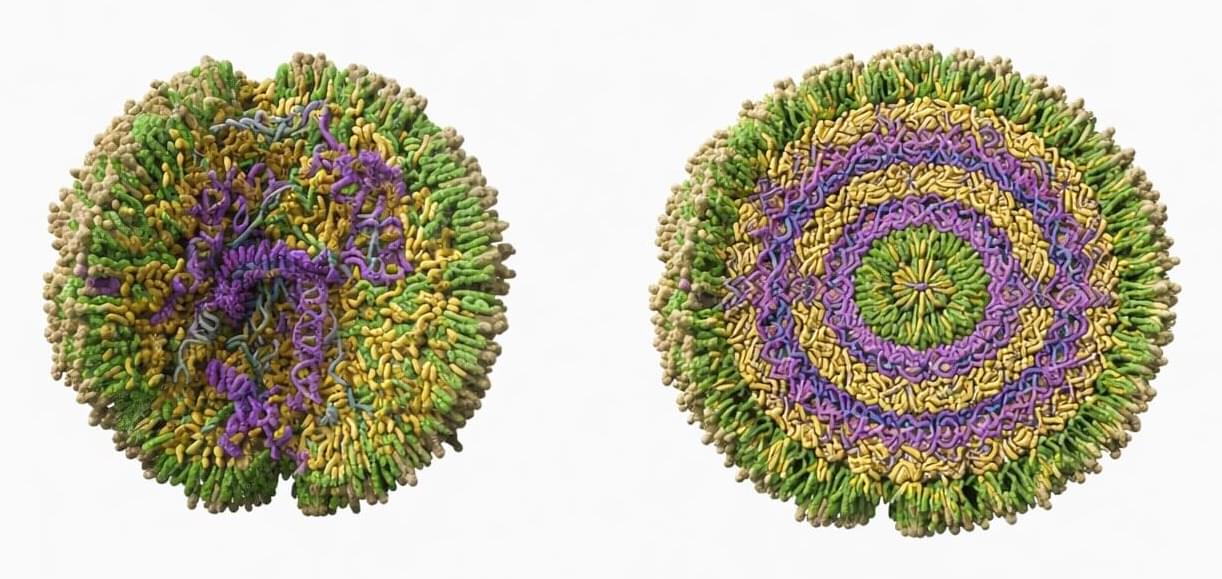
The tiny fatty capsules that deliver COVID-19 mRNA vaccines into billions of arms may work better when they’re a little disorganized. That’s the surprising finding from researchers who developed a new way to examine these drug-delivery vehicles one particle at a time—revealing that cramming in more medicine doesn’t always mean better results.
The research was presented at the 70th Biophysical Society Annual Meeting, held in San Francisco from February 21–25, 2026.
Lipid nanoparticles, or LNPs, are microscopic bubbles of fat that can ferry fragile RNA molecules into cells. They were crucial to the success of mRNA vaccines, and scientists are now working to use them to deliver treatments for cancer, genetic diseases, and other conditions. But there’s a problem: only about 1% to 5% of the cargo inside LNPs actually gets released inside cells.