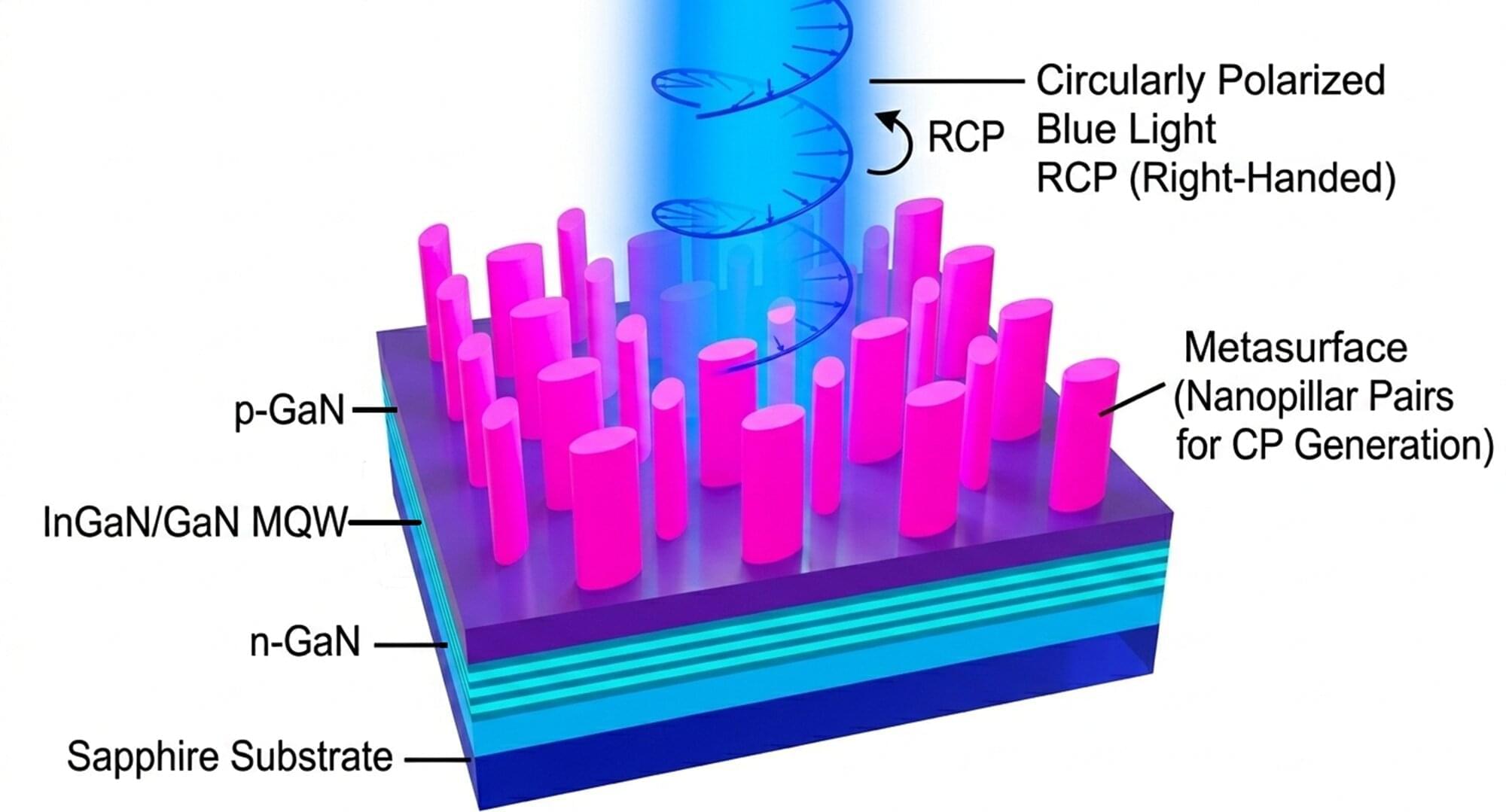
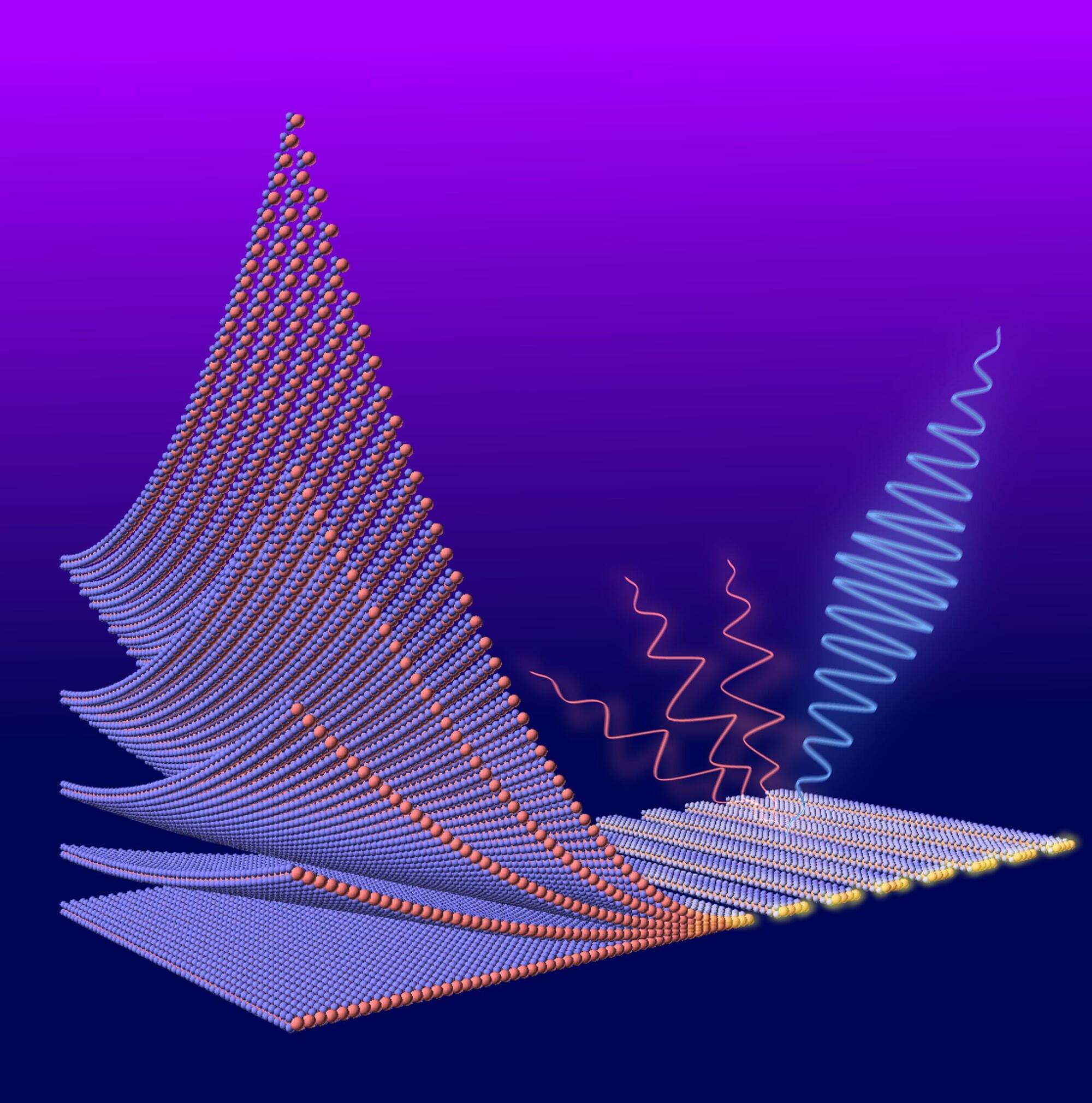
From 3D movie screens to augmented-reality devices, many modern technologies rely on our ability to manipulate light. Doing so in a cost-effective and efficient way, however, is often a formidable task. In an article published in Optics Letters, researchers from the University of Osaka announced a new light-emitting diode (LED) design that may help shrink complex optical systems into much smaller devices. The LED produces circularly polarized light using a built-in nanostructured surface, eliminating the need for bulky external optical components.
Circularly polarized light, whose electric field rotates like a corkscrew as it travels, is essential for technologies such as 3D displays, advanced imaging systems, and quantum communication tools. Traditionally, generating this kind of light requires optical components such as polarizers and special plates that modify the light’s phase. However, these components make devices larger, more complex, and harder to integrate.
“Our goal is to simplify the way circularly polarized light is produced,” says corresponding author Shuhei Ichikawa. “By integrating polarization control directly into the LED with a specially designed metasurface, we remove the need for additional optical components.”