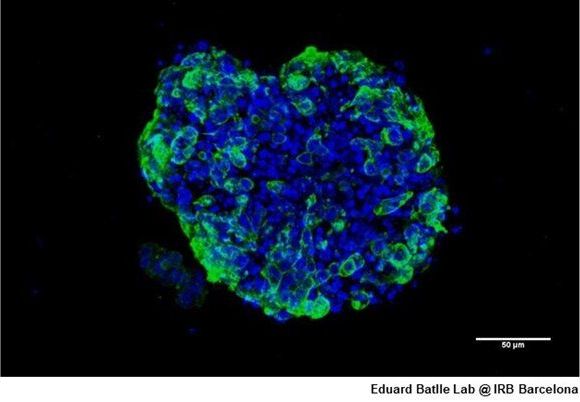
First, we found that every cancer organoid retains the properties of the tissue of origin, so this shows that if the samples were obtained from the surgery of a colon or pancreatic cancer, the organoid closely resembles the original primary tumor. Second, we discovered that there is no contamination of normal cells, thus, the malignant pure transformed cells can be analyzed without interferences. And finally, the 3D organoid cancers are closer to the patient tumors than the commonly used 2-D cell lines.
Scientists have used 3D models to break down the DNA behavior of cancer cells, in a breakthrough new study which could revolutionize treatment for the disease.
In what is a first for science, a research team led by Dr. Manel Esteller, Director of the Josep Carreras Leukaemia Research Institute (IJC), demonstrated how 3D models (known as organoids) can now be used to develop a characterization of the DNA make-up—or the epigenetic fingerprint—of human cancer.
Pubished in Epigenetics, the research validates the use of these 3D samples for cancer research that could deliver new oncology treatments.