The more we understand how cells produce shape and form, the more inadequate the idea of a genomic blueprint looks by Philip Ball + BIO.
Category: genetics – Page 335

An Alzheimer’s-Proof Brain: Ground-Breaking Case Provides Clues to Treatment and Prevention of Dementia
Due to a rare genetic mutation, Aliria Rosa Piedrahita de Villegas should have had Alzheimer’s.
Alzheimer’s disease is a disease that attacks the brain, causing a decline in mental ability that worsens over time. It is the most common form of dementia and accounts for 60 to 80 percent of dementia cases. There is no current cure for Alzheimer’s disease, but there are medications that can help ease the symptoms.
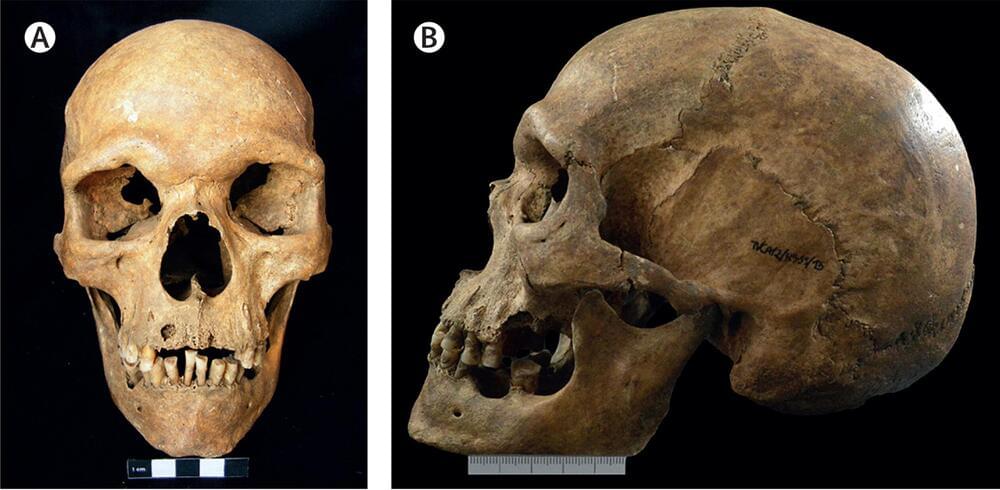
Oldest case of a rare genetic condition discovered
A group of international researchers has uncovered evidence of a super rare genetic condition that gives men an extra X chromosome, reporting the oldest clinical case of Klinefelter syndrome to date.
The evidence comes from a 1,000-year-old skeleton from Portugal.
Klinefelter syndrome is a rare genetic condition where individuals are born with an extra copy of the X chromosome, occurring in approximately one in 1,000 genetic-male births.
Epigenetic Tests #1 and 2: Horvath, Hannum, DunedinPACE
Join us on Patreon!
https://www.patreon.com/MichaelLustgartenPhD
TruDiagnostic Discount Link (Epigenetic Testing)
CONQUERAGING!
https://bit.ly/3Rken0n.
Bristle Discount Link (Oral microbiome quantification):
ConquerAging15
https://www.bmq30trk.com/4FL3LK/GTSC3/
Cronometer Discount Link (Daily diet tracking):
https://shareasale.com/r.cfm?b=1390137&u=3266601&m=61121&urllink=&afftrack=
Support the channel with Buy Me A Coffee!
https://www.buymeacoffee.com/mlhnrca.
Papers referenced in the video:
Hippocampal Egr1-Dependent Neuronal Ensembles Negatively Regulate Motor Learning
Motor skills learning is classically associated with brain regions including cerebral and cerebellar cortices and basal ganglia nuclei. Less is known about the role of the hippocampus in the acquisition and storage of motor skills. Here, we show that mice receiving a long-term training in the accelerating rotarod display marked hippocampal transcriptional changes and reduced pyramidal neurons activity in the CA1 region when compared with naive mice. Then, we use mice in which neural ensembles are permanently labeled in an Egr1 activity-dependent fashion. Using these mice, we identify a subpopulation of Egr1-expressing pyramidal neurons in CA1 activated in short-term (STT) and long-term (LTT) trained mice in the rotarod task. When Egr1 is downregulated in the CA1 or these neuronal ensembles are depleted, motor learning is improved whereas their chemogenetic stimulation impairs motor learning performance. Thus, Egr1 organizes specific CA1 neuronal ensembles during the accelerating rotarod task that limit motor learning. These evidences highlight the role of the hippocampus in the control of this type of learning and we provide a possible underlying mechanism.
SIGNIFICANCE STATEMENT It is a major topic in neurosciences the deciphering of the specific circuits underlying memory systems during the encoding of new information. However, the potential role of the hippocampus in the control of motor learning and the underlying mechanisms has been poorly addressed. In the present work we show how the hippocampus responds to motor learning and how the Egr1 molecule is one of the major responsible for such phenomenon controlling the rate of motor coordination performances.

Scientists Just Genetically Edited a Million Years of Evolution Into Mouse DNA
O.o!!!!!
Changing the number of chromosomes an animal has can take millions of generations to happen in nature through the course of evolution – and now, scientists have been able to make these same changes in lab mice in a relative blink of an eye.
The new technique using stem cells and gene editing is a major accomplishment, and one that the team is hoping will reveal more about how the rearrangement of chromosomes can influence the way that animals evolve over time.
It’s in chromosomes – those strings of protein and DNA inside cells – that we find our genes, inherited from our parents and blended together to make us who we are.
Shrouded in Mystery: Scientists Finally Discover the Origin of Chromatin
Analysis of the genome and proteome shows that eukaryotic evolution gave rise to the regulatory function of chromatin.
Two meters of DNA
DNA, or deoxyribonucleic acid, is a molecule composed of two long strands of nucleotides that coil around each other to form a double helix. It is the hereditary material in humans and almost all other organisms that carries genetic instructions for development, functioning, growth, and reproduction. Nearly every cell in a person’s body has the same DNA. Most DNA is located in the cell nucleus (where it is called nuclear DNA), but a small amount of DNA can also be found in the mitochondria (where it is called mitochondrial DNA or mtDNA).