I believe nanomachines or new advanced rna antivirals that can target one’s own variants of viruses will be game changers to prevent future global pandemics. Also eventually new genetic engineering could allow for the end to all viruses with some sorta Omni vaccine.
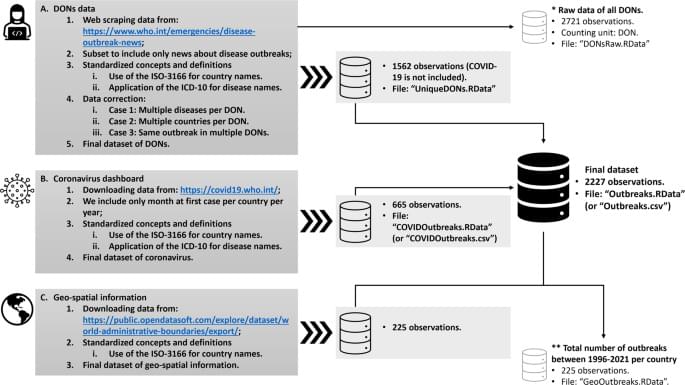
Measurement(s) Pandemic-and epidemic-prone disease outbreaks Technology Type(s) Text mining using R Sample Characteristic — Organism Disease outbreaks Sample Characteristic — Environment spatiotemporal region Sample Characteristic — Location Global.