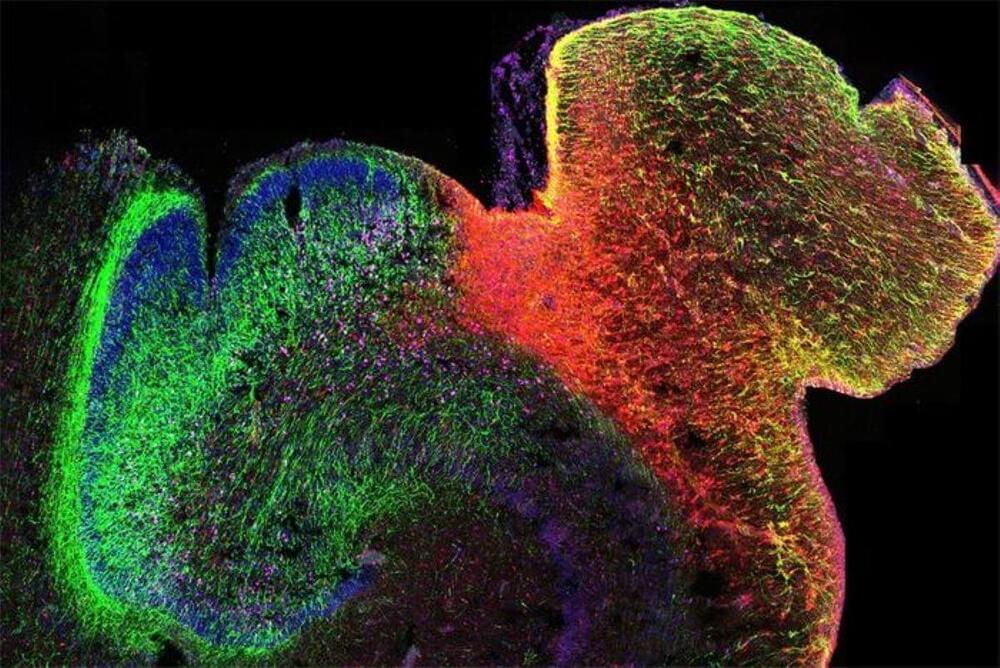
Summary: Researchers have developed the first 3D map of gene regulation in the human brain, offering insights into how early brain development influences lifelong mental health. This map, focusing on regions tied to memory and emotional regulation, reveals how chromatin structure controls gene activity, especially during key developmental stages.
These findings may help identify when and where genetic variants linked to autism and schizophrenia disrupt normal development. By understanding these early influences, scientists hope to improve neurodevelopmental disorder research and stem-cell models, potentially paving the way for earlier intervention strategies.