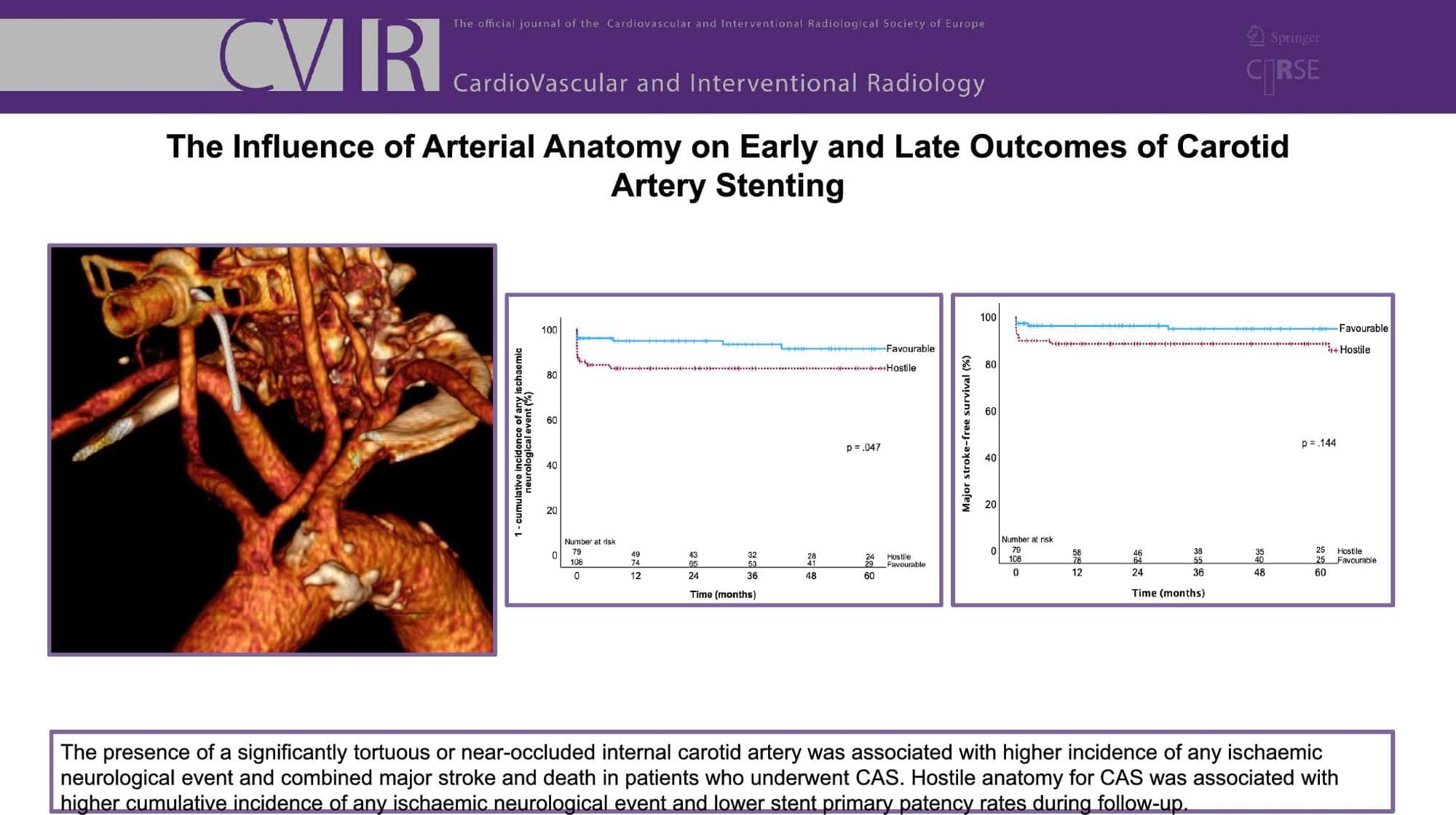
To identify anatomical risk factors for poor perioperative outcomes in patients treated with carotid artery stenting (CAS). To compare long-term results for patients with favourable and hostile anatomy (HA) for CAS.
Single-centre, retrospective study of patients who underwent CAS from 2004 to 2024 at a public hospital in Latin America. Anatomical data were extracted from preoperative imaging regarding the aorta, the supra-aortic trunks, and the internal carotid arteries (ICA). Short-term outcomes included the primary composite outcome of major stroke and death (S/D); any ischaemic neurological event (AINE); major stroke, any stroke. Multivariate analysis (MA) was performed to identify classifying anatomical factors regarding hostility to CAS. Long-term outcomes, defined as AINE; cumulative survival; major stroke-free survival; and stent primary patency (SPP), were analysed using Kaplan–Meier estimates.
MA associated hostile anatomy with complex ICA, defined as either significant tortuosity or near-occlusion. One hundred and eighty-seven CAS procedures were performed in 172 patients, of which 79 (42.3%) had HA. HA correlated with a higher perioperative incidence of AINE (p = .018), any stroke (p = .029), and S/D (p = .049), but not with major stroke (p = .054). At 5 years, the HA group presented higher cumulative incidence of AINE (p = 0,047) and lower SPP (p = 0,011). There were no differences between groups for cumulative survival and major stroke-free survival.