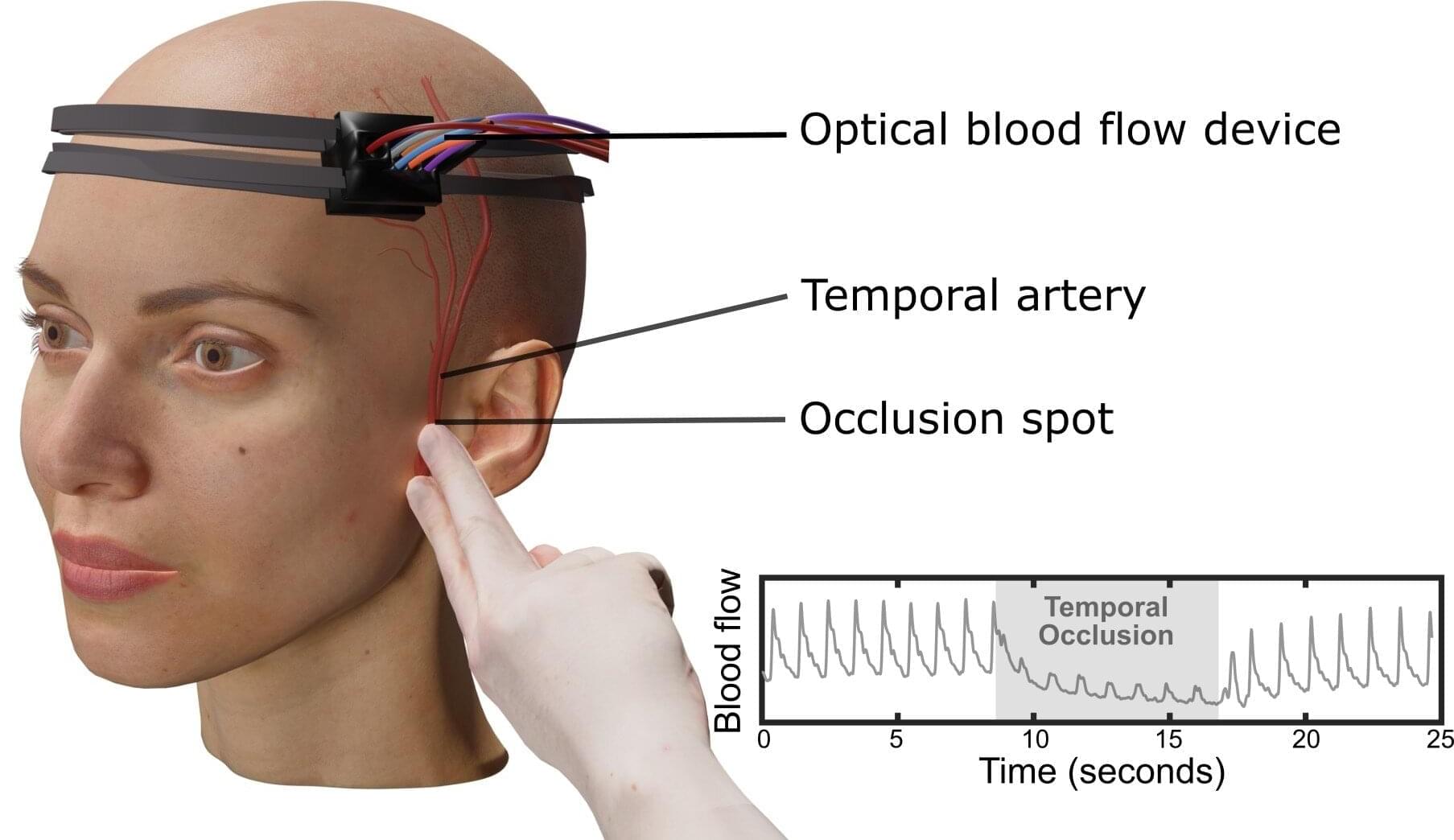
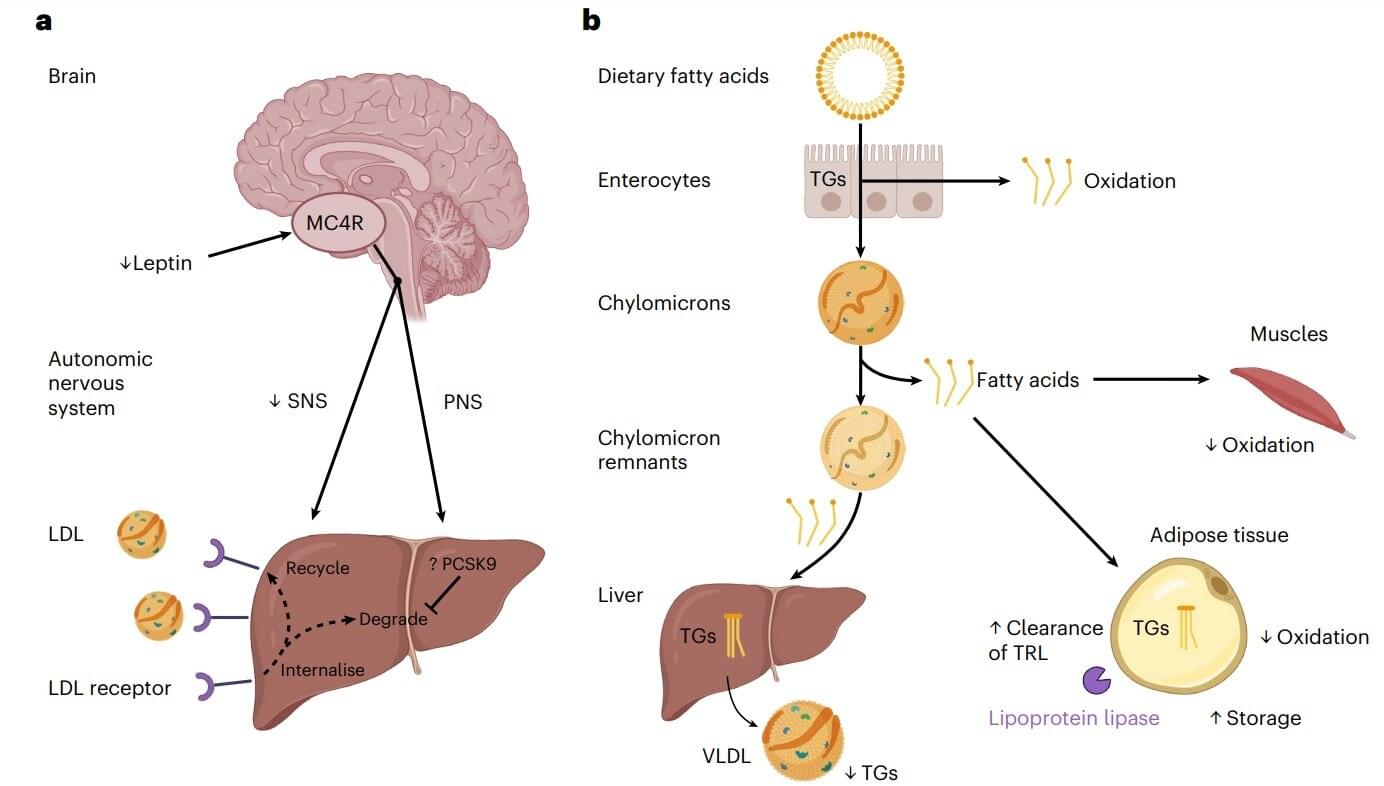
Measuring blood flow in the brain is critical for responding to a range of neurological problems, including stroke, traumatic brain injury (TBI) and vascular dementia. But existing techniques, including magnetic resonance imaging and computed tomography, are expensive and therefore not widely available.
Researchers from the USC Neurorestoration Center and the California Institute of Technology (Caltech) have built a simple, noninvasive alternative. The device takes a technique currently used in animal studies known as speckle contrast optical spectroscopy (SCOS) and adapts it for potential clinical use in humans. It works by capturing images of scattered laser light with an affordable, high-resolution camera.
“It’s really that simple. Tiny blood cells pass through a laser beam, and the way the light scatters allows us to measure blood flow and volume in the brain,” said Charles Liu, MD, Ph.D., professor of clinical neurological surgery, urology and surgery at the Keck School of Medicine of USC, director of the USC Neurorestoration Center and co-senior author of the new research.