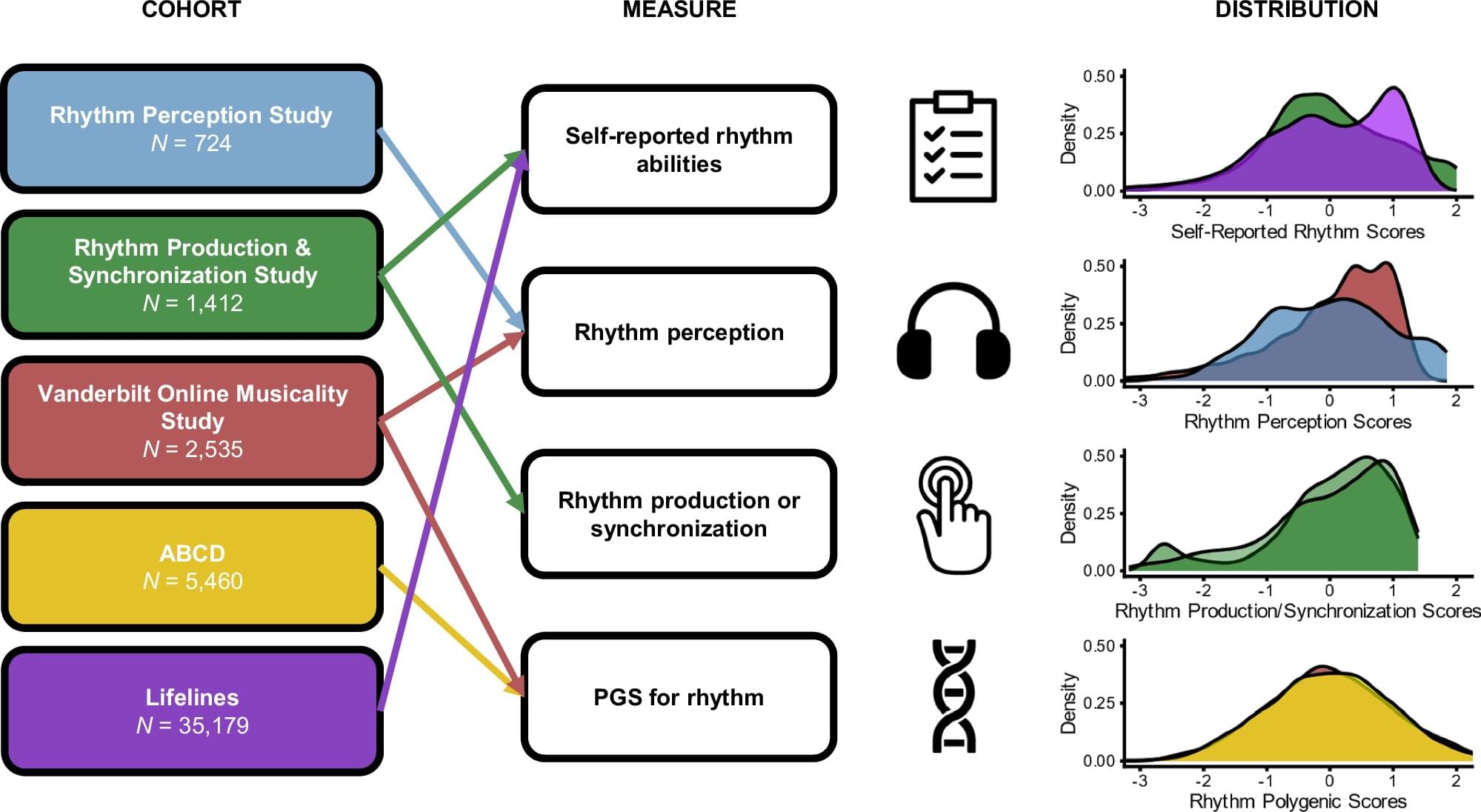
In a paper published in Nature Communications, researchers at Vanderbilt University Medical Center’s Department of Otolaryngology–Head and Neck Surgery leveraged two main studies—one focused on behavior and one focused on genetics—to highlight the correlation between participants’ musical rhythm abilities and developmental speech-language disorders.
These disorders include developmental language disorder, dyslexia and stuttering, among others.
Evidence showed that deficiency in musical rhythm perception is a “modest but consistent risk factor for developmental speech, language and reading disorders,” according to the study’s lead author, Srishti Nayak, Ph.D., assistant professor of Otolaryngology-Head and Neck Surgery.