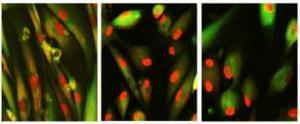
On July 12, 2023, a new research paper was published in Aging, titled, “Chemically induced reprogramming to reverse cellular aging.”
BUFFALO, NY– July 12, 2023 – In a groundbreaking study, researchers have unlocked a new frontier in the fight against aging and age-related diseases. The study, conducted by a team of scientists at Harvard Medical School, has published the first chemical approach to reprogram cells to a younger state. Previously, this was only achievable using a powerful gene therapy.
On July 12, 2023, researchers Jae-Hyun Yang, Christopher A. Petty, Thomas Dixon-McDougall, Maria Vina Lopez, Alexander Tyshkovskiy, Sun Maybury-Lewis, Xiao Tian, Nabilah Ibrahim, Zhili Chen, Patrick T. Griffin, Matthew Arnold, Jien Li, Oswaldo A. Martinez, Alexander Behn, Ryan Rogers-Hammond, Suzanne Angeli, Vadim N. Gladyshev, and David A. Sinclair from Harvard Medical School, University of Maine and Massachusetts Institute of Technology (MIT) published a new research paper in Aging, titled, “Chemically induced reprogramming to reverse cellular aging.”