While cannabis is known to have immunomodulatory properties, the clinical consequences of its use on outcomes in COVID-19 have not been extensively evaluated. We aimed to assess whether cannabis users hospitalized for COVID-19 had improved outcomes compared to non-users.
We conducted a retrospective analysis of 1831 patients admitted to two medical centers in Southern California with a diagnosis of COVID-19. We evaluated outcomes including NIH COVID-19 Severity Score, need for supplemental oxygen, ICU (intensive care unit) admission, mechanical ventilation, length of hospitalization, and in-hospital death for cannabis users and non-users. Cannabis use was reported in the patient’s social history. Propensity matching was used to account for differences in age, body-mass index, sex, race, tobacco smoking history, and comorbidities known to be risk factors for COVID-19 mortality between cannabis users and non-users.
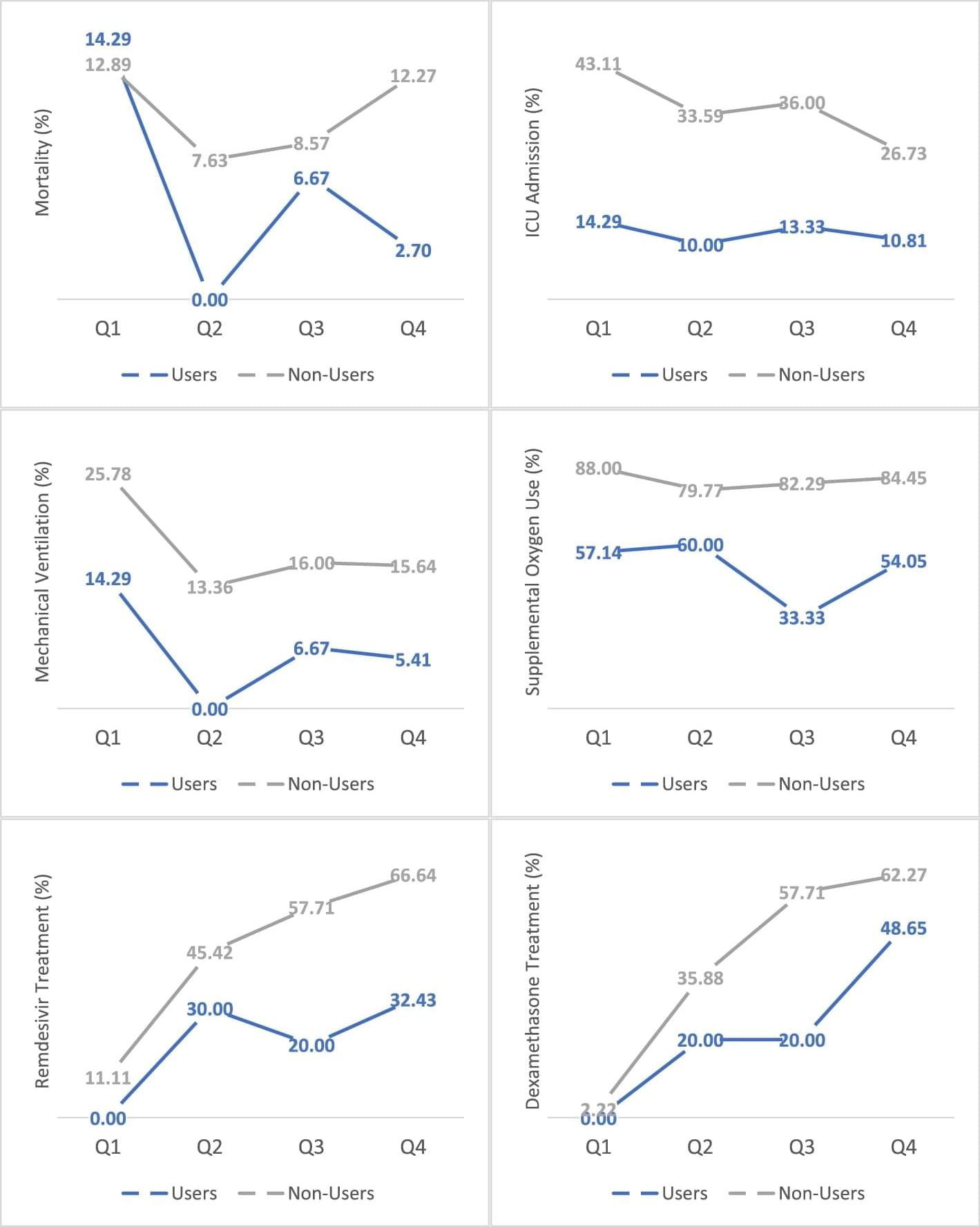
Of 1831 patients admitted with COVID-19, 69 patients reported active cannabis use (4% of the cohort). Active users were younger (44 years vs. 62 years, p < 0.001), less often diabetic (23.2% vs 37.2%, p < 0.021), and more frequently active tobacco smokers (20.3% vs. 4.1%, p < 0.001) compared to non-users. Notably, active users had lower levels of inflammatory markers upon admission than non-users—CRP (C-reactive protein) (3.7 mg/L vs 7.6 mg/L, p < 0.001), ferritin (282 μg/L vs 622 μg/L, p < 0.001), D-dimer (468 ng/mL vs 1,140 ng/mL, p = 0.017), and procalcitonin (0.10 ng/mL vs 0.15 ng/mL, p = 0.001). Based on univariate analysis, cannabis users had significantly better outcomes compared to non-users as reflected in lower NIH scores (5.1 vs 6.0, p < 0.001), shorter hospitalization (4 days vs 6 days, p < 0.001), lower ICU admission rates (12% vs 31%, p < 0.001), and less need for mechanical ventilation (6% vs 17%, p = 0.027).