Anthropic cofounder and president Daniela Amodei sits down with Stripe head of corporate strategy Alex Komoroske at AI Day.
Category: robotics/AI – Page 1,099
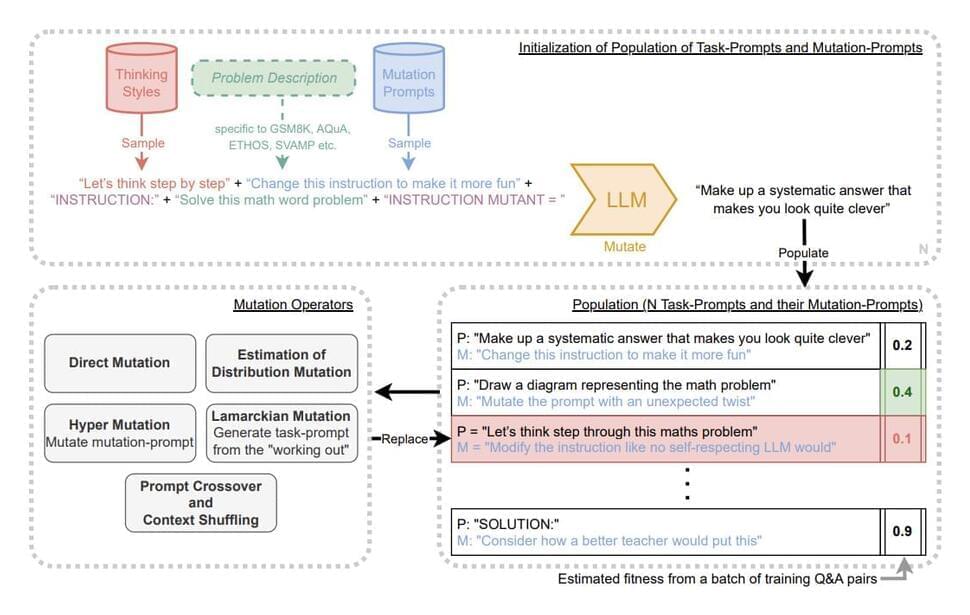
Google DeepMind Researchers Introduce Promptbreeder: A Self-Referential and Self-Improving AI System that can Automatically Evolve Effective Domain-Specific Prompts in a Given Domain
Large Language Models (LLMs) have gained a lot of attention for their human-imitating properties. These models are capable of answering questions, generating content, summarizing long textual paragraphs, and whatnot. Prompts are essential for improving the performance of LLMs like GPT-3.5 and GPT-4. The way that prompts are created can have a big impact on an LLM’s abilities in a variety of areas, including reasoning, multimodal processing, tool use, and more. These techniques, which researchers designed, have shown promise in tasks like model distillation and agent behavior simulation.
The manual engineering of prompt approaches raises the question of whether this procedure can be automated. By producing a set of prompts based on input-output instances from a dataset, Automatic Prompt Engineer (APE) made an attempt to address this, but APE had diminishing returns in terms of prompt quality. Researchers have suggested a method based on a diversity-maintaining evolutionary algorithm for self-referential self-improvement of prompts for LLMs to overcome decreasing returns in prompt creation.
LLMs can alter their prompts to improve their capabilities, just as a neural network can change its weight matrix to improve performance. According to this comparison, LLMs may be created to enhance both their own capabilities and the processes by which they enhance them, thereby enabling Artificial Intelligence to continue improving indefinitely. In response to these ideas, a team of researchers from Google DeepMind has introduced PromptBreeder (PB) in recent research, which is a technique for LLMs to better themselves in a self-referential manner.
Mo Gawdat: AI Today, Tomorrow and How You Can Save Our World (Nordic Business Forum 2023)
Mo Gawdat openly discusses the current rate of advancement of AI and the expected technological innovation that will follow at the Nordic Business Forum 2023 in Helsinki on September 27, 2023.
Learning points:
Where AI might be heading as a technology.
Ethical questions to consider as a business leader.
What is the role each of us and our businesses have to play to ensure that AI will be a driver for positive change.

Transformers — Intuitively and Exhaustively Explained
In this post you will learn about the transformer architecture, which is at the core of the architecture of nearly all cutting-edge large language models. We’ll start with a brief chronology of some relevant natural language processing concepts, then we’ll go through the transformer step by step and uncover how it works.
Who is this useful for? Anyone interested in natural language processing (NLP).
How advanced is this post? This is not a complex post, but there are a lot of concepts, so it might be daunting to less experienced data scientists.
Artificial Intelligence Shaping the Future of Medicine
SUBSCRIBE TO DOCTORLY UNHINGED PODCAST!
https://doctorly.podlink.to/unhinged.
*Shop Favorite Skincare Products*
Shop My Shelf: https://shopmyshelf.us/shops/dermdoctor.
Amazon Favorites: https://www.amazon.com/shop/doctorly?tag=lifeboatfound-20.
20% Off Paula’s Choice: https://www.tkqlhce.com/click-100667849-15267262
Shop SkinBetter Science: https://skinbetter.pro/doctorly.
*Affiliate Links Generate Revenue for the Channel*
We take a look at how the latest advancements in artificial intelligence are impacting modern medicine and theorize about what the future may hold.
00:00 Intro.
Disclaimer: This video is not intended to provide diagnosis, treatment, or medical advice. Content provided in this video is for educational purposes only. Please consult with a physician regarding any health-related diagnosis or treatment.

Using Artificial Intelligence to Help Students With Down Syndrome Succeed
The Center for Artificial Intelligence at King Khalid University embraces AI-driven innovation by using and creating advanced digital technologies to help fulfill Vision 2030 objectives. With that as the main driver, Nada Saeed Al-Qahtani, student and inventor at the College of Computer Science at King Khalid University, developed a robot called “Eve”. Eve is tailored to students with Down Syndrome, being equipped with machine learning curriculum adaptations that make completing tasks easier.
Technology helps paralysed man to walk
A cyclist who was paralysed in an accident is able to walk again, thanks to a breakthrough in neurosurgery and artificial intelligence in Switzerland.
Al Jazeera’s Andrew Simmons reports.
- Subscribe to our channel: http://aje.io/AJSubscribe.
- Follow us on Twitter: https://twitter.com/AJEnglish.
- Find us on Facebook: https://www.facebook.com/aljazeera.
- Check our website: https://www.aljazeera.com/
@AljazeeraEnglish.
#Aljazeeraenglish.
#News #AI #Technology



Artists across industries are strategizing together around AI concerns
As creative industries grapple with AI’s explosion into every artistic medium at once, separate calls from artists warning the world to take action before it’s too late are starting to converge. From fake Drake songs to stylized Instagram profile pictures, art conjured with newly sophisticated AI tools is suddenly ubiquitous — and so are conversations about how to rein in the technology before it does irrevocable harm to creative communities.
This week, digital rights organization Fight for the Future partnered with music industry labor group United Musicians and Allied Workers to launch #AIdayofaction, a campaign that calls on Congress to block corporations from obtaining copyrights on music and other art made with AI.
The idea is that by preventing industry behemoths like major record labels, for example, from copyrighting music made with the assistance of AI, those companies will be forced to keep looping humans into the creative process. But those same concerns — and the same potential strategies for pushing back against the onslaught of AI — exist across creative industries.