Focusing on sound production instead of word choice makes for a flexible system.




This week, biologists discovered a new cellular organelle that’s like “a new recycling center within the cell.” Wild-growing tomatoes in the Galápagos are de-evolving. And geologists at the University of Southampton detected deep Earth pulses beneath Africa. Plus: Brain network functionality differs in people with psychopathic personality; sharks have a surprising vulnerability; and people with the highest measured IQ make better probabilistic predictions than people with the lowest IQ.
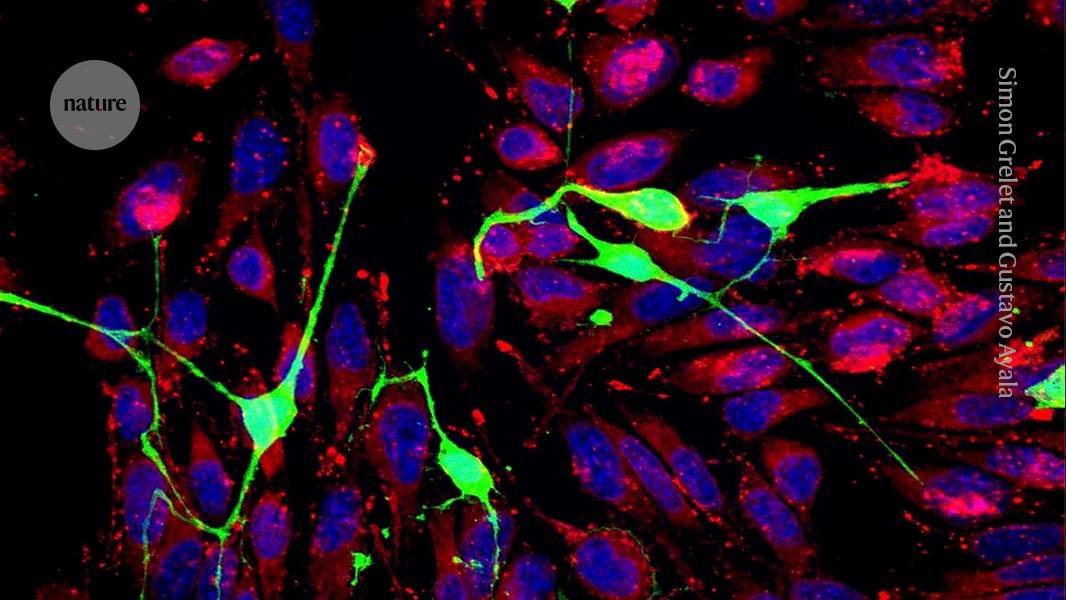
Cancer cells turbocharge themselves by stealing the energy-producing units from neurons in tumours, scientists report today in Nature 1. This act of thievery seems to give cancer cells a boost to help them survive when they metastasize, or spread to distant organs.
The findings show that cancer cells siphon off neurons’ mitochondria — organelles that generate most of a cell’s energy — through ultrathin tubes that grow between the two types of cell. The purloined mitochondria increase cancer cells’ ability to withstand the stress of shooting through blood vessels during metastasis.
“Now we have a new culprit for metastasis, which means we have a new target to block metastasis,” says study co-author Simon Grelet, a cancer neurobiologist at the University of South Alabama in Mobile. “And metastasis is what make cancers so deadly.”
The theft probably helps the cells to spread around the body, and preventing it could provide a path to treatment, researchers say.
Mark Hersam is a nanotechnologist who believes that understanding materials at the shortest of length scales can provide solutions to the world’s largest problems. Using an interdisciplinary approach at the intersection of neuroscience and nanoelectronics, Hersam presents a solution to the greatest societal threat posed by AI.
Dr. Mark C. Hersam, the Walter P. Murphy Professor of Materials Science and Engineering, Director of the Materials Research Center, and Chair of the Materials Science and Engineering Department at Northwestern University, has made major breakthroughs in the field of nanotechnology. His research interests include nanomaterials, additive manufacturing, nanoelectronics, scanning probe microscopy, renewable energy, and quantum information science. Dr. Hersam has received several honors including the Marshall Scholarship, Presidential Early Career Award for Scientists and Engineers, American Vacuum Society Medard Welch Award, U.S. Science Envoy, and MacArthur Fellowship. In addition, he is an elected member of the American Academy of Arts and Sciences, National Academy of Engineering, and National Academy of Inventors and has founded two companies, NanoIntegris and Volexion, which are suppliers of nanoelectronic and battery materials, respectively.
This talk was given at a TEDx event using the TED conference format but independently organized by a local community.
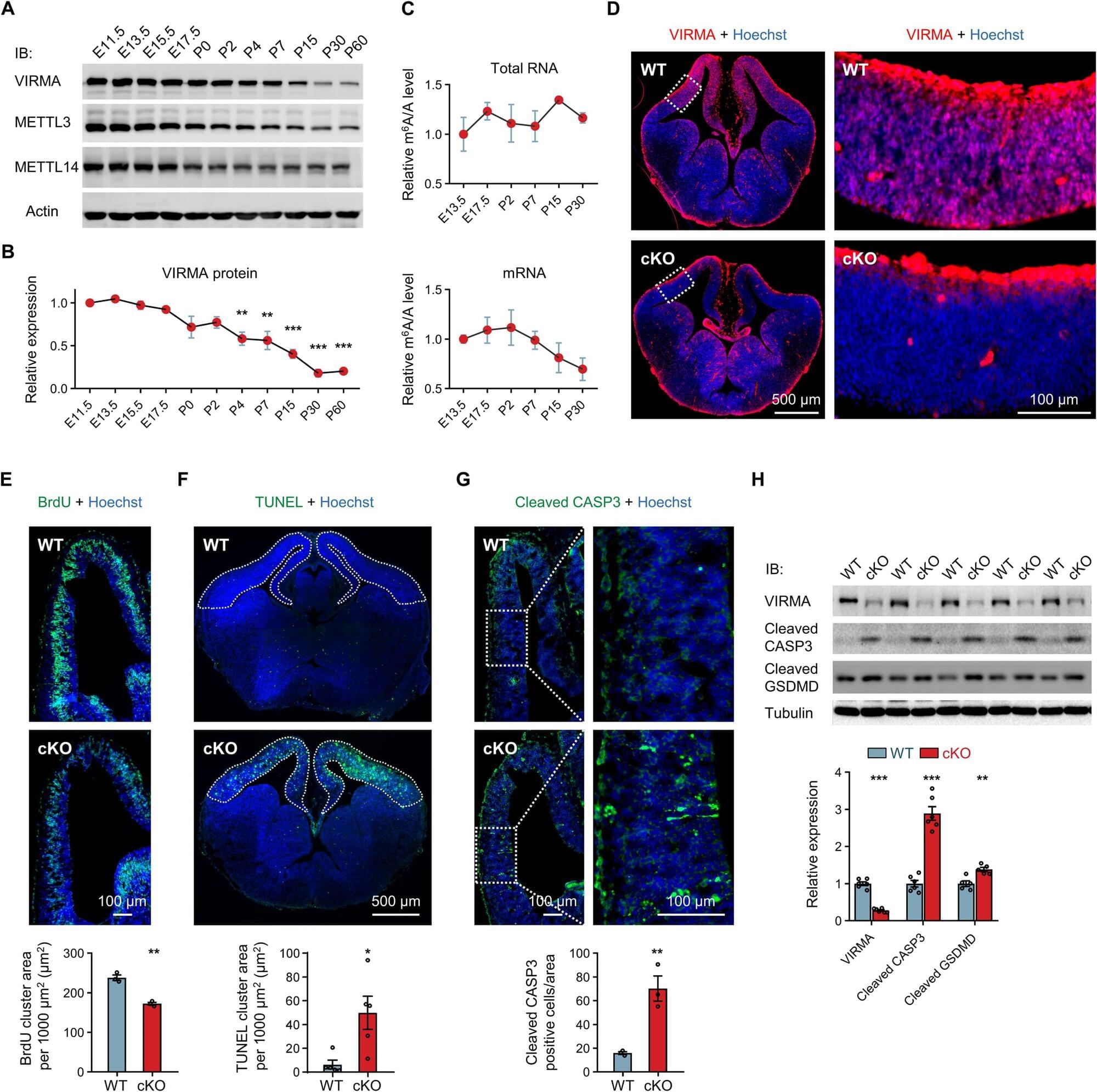
Ribosomes are tiny molecular machines inside all living cells that build proteins, and ribosome biogenesis is the complex, multi-step process by which they are made. During brain development, neural stem cell proliferation relies on active ribosome biogenesis to meet high protein demand. This process involves the concerted action of numerous ribosomal RNA processing factors and assembly proteins. Studies have shown that precise regulation of ribosome biogenesis is essential for normal brain development and tumor prevention.