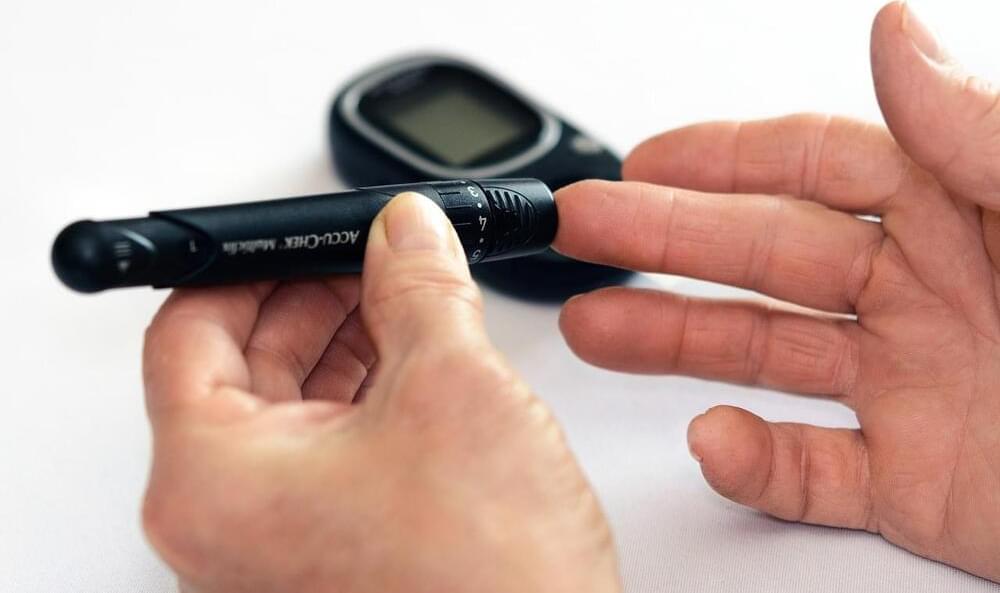
In America, roughly 40 million Americans have diabetes and about 95% of them have type 2 diabetes. Type 2 diabetes occurs when the body cannot correctly process sugar and fuel cells. More specifically, the body does not produce enough insulin to break down sugar into glucose for the cells to use. In this case, treatment includes insulin shots or a pump in addition to a strict diet excluding sweets or high fat meals. Treatment limitations disrupt patient quality of life. Some researchers have been working on better detection for diabetic retinopathy with artificial intelligence (AI), but research is limited on how to better detect diabetes itself. Thus, many researchers are working to detect diabetes early on and discover better treatments.
Klick labs, located in multiple cities across the world, is trying to detect type 2 diabetes by having a patient speak into a microphone for 10 seconds. Klick labs believes this technology can better detect diabetes and help patients get treatment earlier. The study was published in Mayo Clinic Proceedings: Digital Health, which details how patients spoke for 10 seconds and combined with health data, including age, sex, height, and weight, created an AI model that discerns whether a person has type 2 diabetes or not. After further tests, scientists determined it has 89% and 86% accuracy for women and men, respectively.
In the study, Klick Labs collected voice recordings of 267 people, either non-diabetic or diabetic. The participants were asked to record a phrase into their smartphones six times a day for a total of 2-weeks. Over 18,000 recordings were taken and analyzed to distinguish 14 acoustic features that helped distinguish non-diabetic to type 2 diabetic individuals. The research highlights specific vocal variations in pitch and intensity that could lead to how the medical community screens for early-onset diabetes. A major barrier to early detection includes time, travel, and cost, which many people do not have. Voice diagnosis can help eliminate those barriers and improve detection and treatment in diabetic patients.