Laser cutting techniques are usually powered by high energy beams, so hot that they melt most materials. Now scientists from McGill University have developed a gentler, more precise technique using low-power visible light.


Support our work via Patreon: https://www.patreon.com/AsteronX
AsteronX.com.
Tweets by AsteronX
https://www.facebook.com/asteronx/
Did you know that the best kind of armour is the one that is already vaporised?
Hello, I’m Todd and this is AsteronX.
Today we’re going to be discussing real energy shields, and even propose one, maybe two, of our own concepts.
Armour, regardless of whether its protecting against weaponry or the hazards of space travel in general, is limited by its melting and vaporisation temperatures.
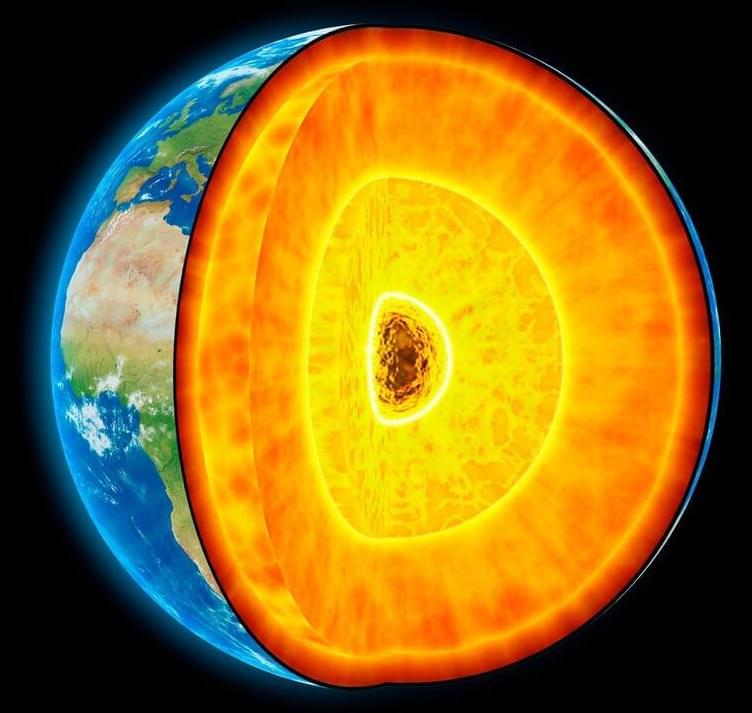
Theory underpins our understanding of convection in the Earth’s outer core and its function in controlling the planet’s magnetic field. Convective flows or how they may be changing have never been directly observed by scientists. Virginia Tech geoscientist Ying Zhou puts proof forward for the first time.
A large earthquake shook the Kermadec Islands region in the South Pacific Ocean in May 1997. A little over 20 years later, in September 2018, a second big earthquake hit the same location, with its waves of seismic energy emanating from the same region.
Although two decades separated the earthquakes, because they occurred in the same region, they’d be expected to send seismic waves through the Earth’s layers at the same speed, said Ying Zhou, a geoscientist with the Department of Geosciences at the Virginia Tech College of Science.

Spotted: Although relatively expensive to produce at present, and with storage often cited as a concern, green hydrogen fuel production is increasing. A naturally occurring and superabundant element, hydrogen is popular for several reasons, including the ability to produce it using renewable energy sources. And now, Element 1’s modular, grid-independent hydrogen generation technology is making the fuel even more accessible.
Designed to efficiently convert methanol to hydrogen to electricity, the technology supports both hydrogen fuel cell vehicles and electric vehicles. The company’s catalytic reactor heats a methanol and water feedstock mix before sending it through a membrane purifier for almost 100 per cent fuel cell grade hydrogen.
Because the modular system produces the fuel as needed, the risk of combustion is nearly eliminated, and specialty storage facilities are redundant. This is because the only material that needs to be stored and transported is the methanol and water feedstock. The hydrogen is then produced on-site. Element 1 provides both small and large-scale solutions, as well as a mobile version specifically for refuelling electric vehicles on the go.
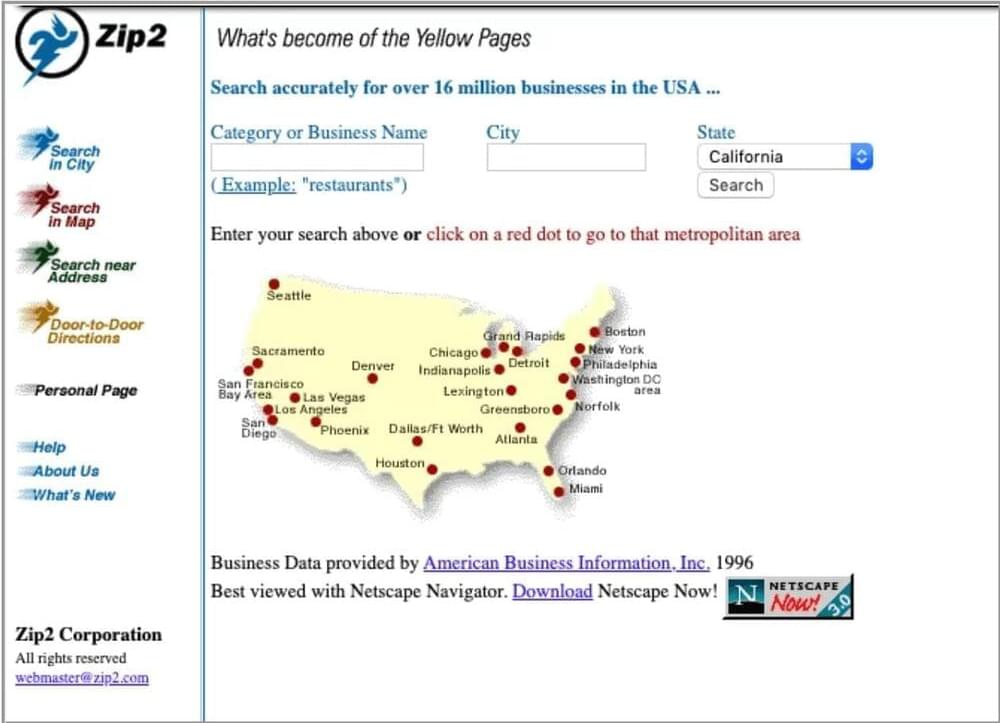
Global Link Information Network was founded in 1995 by Elon Musk, his brother Kimbal Musk, and Greg Kouri who provided $6,000 in funding. Elon famously dropped out of pursuing a PhD at Stanford in energy physics/material science two days into the program to pursue the opportunity. Global Link Information Network was later renamed as Zip2 and served as a directory for local businesses. In 1996, Zip2 starts pulling data from American Business Information Inc. (ABI) and starts a self-comparison to yellow pages that will continue throughout its lifespan as a brand.
A timeline showing Elon Musk’s first website (Zip2) and its evolutions over the years, starting from 1996 and ending in 2004.
Space is a deep, dark, vast abyss that exists between the cosmos, separating them from each other. But is it truly as empty as we think it is? Or is the vacuum that spans everywhere hiding something from us? Something mysterious, and perhaps the most powerful source of energy?
Zero-point energy, also known as vacuum energy, has been touted as a potentially limitless and ubiquitous source of energy, if one could only find the means to harness it.
Welcome to Factnomenal and today we’re looking at the facts and demystifying the most mysterious energy in the universe.
Buy us a coffee to show your support!
https://www.buymeacoffee.com/Factnomenal.
DON’T CLICK THIS LINK: https://tinyurl.com/357shs3j.
Thanks for watching Factnomenal!
Aircrafts transport people, ship goods, and perform military operations, but the petroleum-based fuels that power them are in short supply. In research publishing on June 30 in the journal Joule, researchers at the Lawrence Berkeley Lab have found a way to generate an alternative jet fuel by harvesting an unusual carbon molecule produced by the metabolic process of bacteria commonly found in soil.
“In chemistry, everything that requires energy to make will release energy when it’s broken,” says lead author Pablo Cruz-Morales, a microbiologist at DTU Biosustain, part of the Technical University of Denmark. When petroleum jet fuel is ignited, it releases a tremendous amount of energy, and the scientists at the Keasling Lab at the Lawrence Berkeley Laboratory thought there must be a way to replicate this without waiting millions of years for new fossil fuels to form.
Jay Keasling, a chemical engineer at University of California, Berkeley, approached Cruz-Morales, who was a postdoc in his lab at the time, to see if he could synthesize a tricky molecule that has the potential to produce a lot of energy. “Keasling told me: it’s gonna be an explosive idea,” says Cruz-Morales.

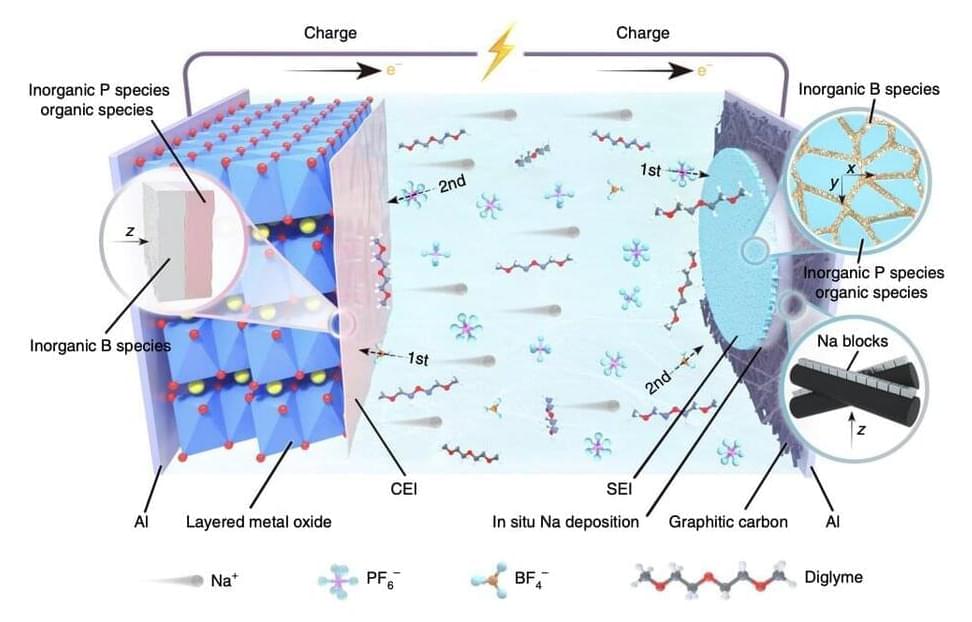
In recent years, engineers worldwide have been trying to devise new battery and energy storage technologies that are more sustainable and cost-effective. One of the solutions attracting particular interest is sodium-based battery technology.
Sodium-ion batteries could have numerous advantages over conventional and widely used lithium-based batteries. Most notably, as sodium is abundant on our planet and can be easily sourced, they could be affordable and easy to produce on a large-scale.
Despite their possible advantages, most sodium-ion batteries developed so far exhibited low energy densities, due to the relatively large atomic size of sodium and its considerable weight. Typically, these batteries exhibit energy densities below 160 Wh kg-1, which is significantly lower than that of lithium-ion batteries.