Some people who live near the derailment site in East Palestine, Ohio, fear they and their animals may be exposed to chemicals through the air, water and soil.



Amount of toxin present in wheat, which is carcinogenic when heated, can be reduced and grown, new field study confirms Toast could soon be healthier after scientists grew a field of wheat genetically-edited to remove a cancer-causing chemical. Bread, when baked, produces a dangerous toxin called acrylamide, which is believed to be carcinogenic and when toasted is even more lethal.
In 2021, Carbon Robotics unveiled the third-generation of its Autonomous Weeder, a smart farming robot that identifies weeds and then destroys them with high-power lasers. The company now has taken the technology from that robot and built a pull-behind LaserWeeder — and it kills twice as many weeds.
The weedkiller challenge: Weeds compete with plants for space, sunlight, and soil nutrients. They can also make it easier for insect pests to harm crops, so weed control is a top concern for farmers.
Chemical herbicides can kill the pesky plants, but they can also contaminate water and affect soil health. Weeds can be pulled out by hand, but it’s unpleasant work, and labor shortages are already a huge problem in the agriculture industry.

Researchers at the University of Göttingen have created a new approach to generate colored X-ray images. Previously, the only way to determine the chemical composition and arrangement of components in a sample using X-ray fluorescence analysis was to focus X-rays on the entire sample and scan it, which was both time-consuming and costly. The new method allows for the creation of an image of a large area with just one exposure, eliminating the need for focusing and scanning. The findings were published in the journal Optica.
In contrast to visible light, there are no comparably powerful lenses for “invisible” radiation, such as X-ray, neutron, or gamma radiation. However, these types of radiation are essential, for example, in nuclear medicine and radiology, as well as in industrial testing and material analysis.
Uses for X-ray fluorescence include analyzing the composition of chemicals in paintings and cultural artifacts to determine authenticity, origin, or production technique, or the analysis of soil samples or plants in environmental protection. The quality and purity of semiconductor components and computer chips can also be checked using X-ray fluorescence analysis.


The first signs of life emerged on Earth in the form of microbes about four billion years ago. While scientists are still determining exactly when and how these microbes appeared, it’s clear that the emergence of life is intricately intertwined with the chemical and physical characteristics of early Earth.
“It is reasonable to suspect that life could have started differently—or not at all—if the early chemical characteristics of our planet were different,” says Dustin Trail, an associate professor of earth and environmental sciences at the University of Rochester.
But what was Earth like billions of years ago, and what characteristics may have helped life to form? In a paper published in Science, Trail and Thomas McCollom, a research associate at the University of Colorado Boulder, reveal key information in the quest to find out. The research has important implications not only for discovering the origins of life but also in the search for life on other planets.

To make long-term presence on the Moon viable, we need abundant electrical power. We can make power systems on the Moon directly from materials that exist everywhere on the surface, without special substances brought from Earth. We have pioneered the technology and demonstrated all the steps. Our approach, Blue Alchemist, can scale indefinitely, eliminating power as a constraint anywhere on the Moon.
We start by making regolith simulants that are chemically and mineralogically equivalent to lunar regolith, accounting for representative lunar variability in grain size and bulk chemistry. This ensures our starting material is as realistic as possible, and not just a mixture of lunar-relevant oxides. We have developed and qualified an efficient, scalable, and contactless process for melting and moving molten regolith that is robust to natural variations in regolith properties on the Moon.
Using regolith simulants, our reactor produces iron, silicon, and aluminum through molten regolith electrolysis, in which an electrical current separates those elements from the oxygen to which they are bound. Oxygen for propulsion and life support is a byproduct.
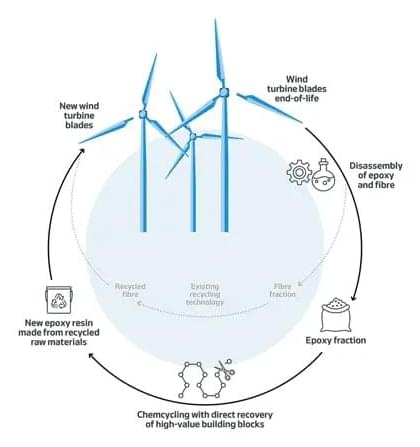
A new chemical process developed by Danish company Vestas can ensure that wind turbine blades are recycled at the end of their life, instead of being abandoned or going to landfill sites.
Wind power is one of the best ways to decarbonise the world’s electricity. Recent years have seen explosive growth in capacity additions, as well as gigantic new turbine designs able to generate as much as 18 MW. The costs keep falling, while efficiencies continue to improve. The trend is now obvious: renewable energy is the future and will inevitably displace fossil fuels.
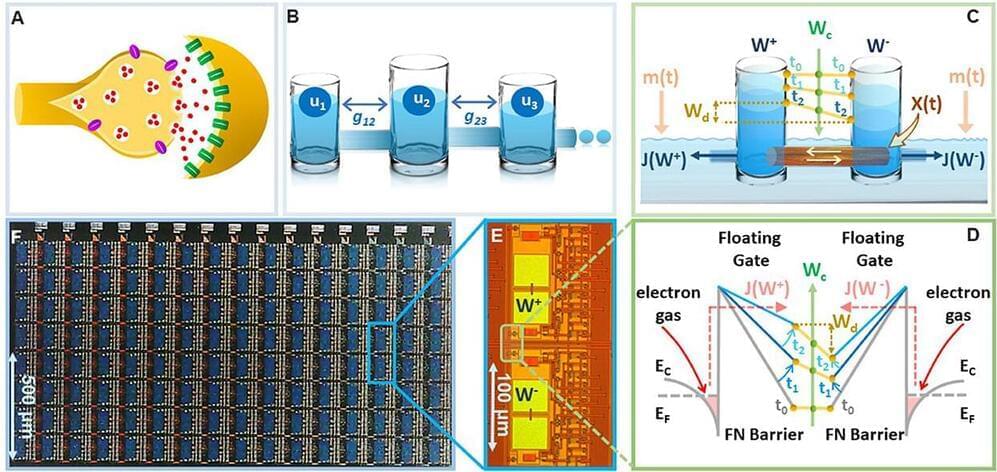
Artificial intelligence and machine learning have made tremendous progress in the past few years including the recent launch of ChatGPT and art generators, but one thing that is still outstanding is an energy-efficient way to generate and store long-and short-term memories at a form factor that is comparable to a human brain. A team of researchers in the McKelvey School of Engineering at Washington University in St. Louis has developed an energy-efficient way to consolidate long-term memories on a tiny chip.
Shantanu Chakrabartty, the Clifford W. Murphy Professor in the Preston M. Green Department of Electrical & Systems Engineering, and members of his lab developed a relatively simple device that mimics the dynamics of the brain’s synapses, connections between neurons that allows signals to pass information. The artificial synapses used in many modern AI systems are relatively simple, whereas biological synapses can potentially store complex memories due to an exquisite interplay between different chemical pathways.
Chakrabartty’s group showed that their artificial synapse could also mimic some of these dynamics that can allow AI systems to continuously learn new tasks without forgetting how to perform old tasks. Results of the research were published Jan. 13 in Frontiers in Neuroscience.
Is Director of the Division of Research, Innovation and Ventures (DRIVe — https://drive.hhs.gov/) at the Biomedical Advanced Research and Development Authority (https://aspr.hhs.gov/AboutASPR/ProgramOffices/BARDA/Pages/default.aspx), a U.S. Department of Health and Human Services (HHS) office responsible for the procurement and development of medical countermeasures, principally against bioterrorism, including chemical, biological, radiological and nuclear (CBRN) threats, as well as pandemic influenza and emerging diseases.
Dr. Patel is committed to advancing high-impact science, building new products, and launching collaborative programs and initiatives with public and private organizations to advance human health and wellness. As the DRIVe Director, Dr. Patel leads a dynamic team built to tackle complex national health security threats by rapidly developing and deploying innovative technologies and approaches that draw from a broad range of disciplines.
Dr. Patel brings extensive experience in public-private partnerships to DRIVe. Prior to joining the DRIVe team, he served as the HHS Open Innovation Manager. In that role, he focused on advancing innovative policy and funding solutions to complex, long-standing problems in healthcare. During his tenure, he successfully built KidneyX, a public-private partnership to spur development of an artificial kidney, helped design and execute the Advancing American Kidney Health Initiative, designed to catalyze innovation, double the number of organs available for transplant, and shift the paradigm of kidney care to be patient-centric and preventative, and included a Presidential Executive Order signed in July 2019. He also created the largest public-facing open innovation program in the U.S. government with more than 190 competitions and $45 million in awards since 2011.
Prior to his tenure at HHS, Dr. Patel co-founded Omusono Labs, a 3D printing and prototyping services company based in Kampala, Uganda; served as a scientific analyst with Discovery Logic, (a Thomson Reuters company) a provider of systems, data, and analytics for real-time portfolio management; and was a Mirzayan Science and Technology Policy Fellow at The National Academies of Science, Engineering, and Medicine. He also served as a scientist at a nanotechnology startup, Kava Technology.
Dr. Patel holds a US patent issued in 2005 and has authored over a dozen peer-reviewed articles in areas such as nanotechnology, chemistry, innovation policy, and kidney health.
Dr. Patel earned his Ph.D. in physical chemistry from the Georgia Institute of Technology, and has a bachelor’s degree in chemistry from Washington University in St. Louis.