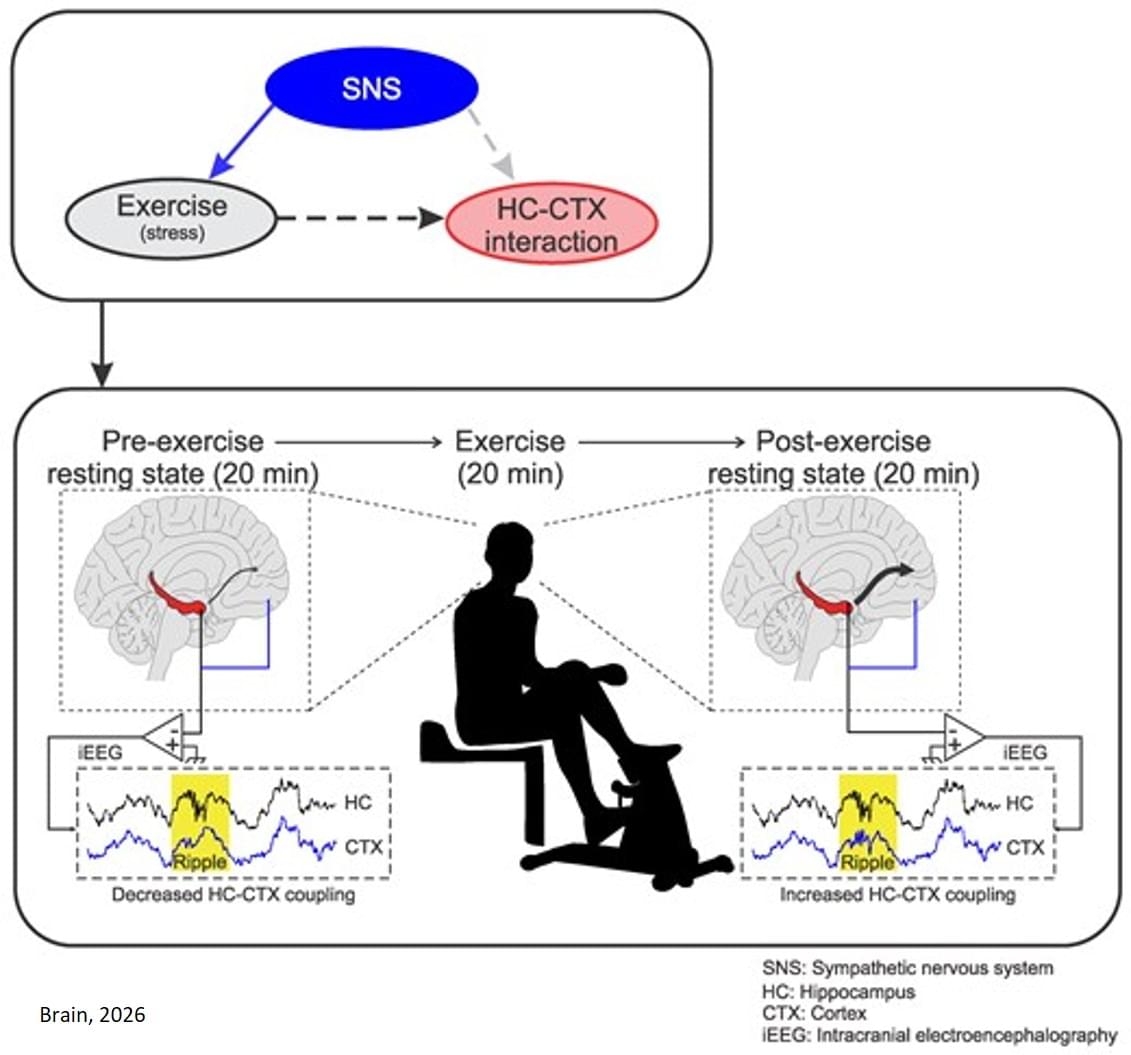
The team recruited 14 patients between 17 and 50 years of age, to participate. After a brief warmup, participants rode a stationary bike for 20 minutes at a pace they could maintain for the duration. Researchers recorded the participants’ brain activity before and after the cycling session using intracranial electroencephalography (iEEG), which utilizes implanted electrodes to measure neural activity in the brain. The recordings showed an increased rate of ripples originating in the hippocampus and connecting with cortical regions of the brain known to be involved in learning and memory performance.
“We’ve known for years that physical exercise is often good for cognitive functions like memory, and this benefit is associated with changes in brain health, largely from behavioral studies and noninvasive brain imaging,” says the study’s corresponding author. “By directly recording brain activity, our study shows, for the first time in humans, that even a single bout of exercise can rapidly alter the neural rhythms and brain networks involved in memory and cognitive function.”
The author says the results apply beyond the epileptic patients who participated. ScienceMission sciencenewshighlights.
A single session of physical exercise can spawn a boost of neural activity in brain networks that underlie learning and memory, according to a new study.
The researchers measured neural activity in the brains of patients with epilepsy before and after they completed a bout of physical exercise. The results showed that a single exercise session produced in the participants a burst of high-frequency brain waves, called ripples, emanating from the hippocampus to areas of the brain involved in learning and recall.
Neuroscientists have documented ripples relevant to memory in mice and rats, but they had not confirmed the link in humans, mainly because electrodes need to be implanted in the brain to obtain recordings. Instead, researchers had theorized the ripples’ role in humans, based on studies in people that measured changes in oxygenated blood in the brain after exercise. This new study marks the first time researchers have been able to see the neurons in action in people following exercise, the authors report.