For much of the 20th century it was thought that the adult brain was incapable of regeneration. This view has since shifted dramatically and neurogenesis—the birth of new neurons—is now a widely accepted phenomenon in the adult brain, offering promising avenues for treating many neurological conditions.
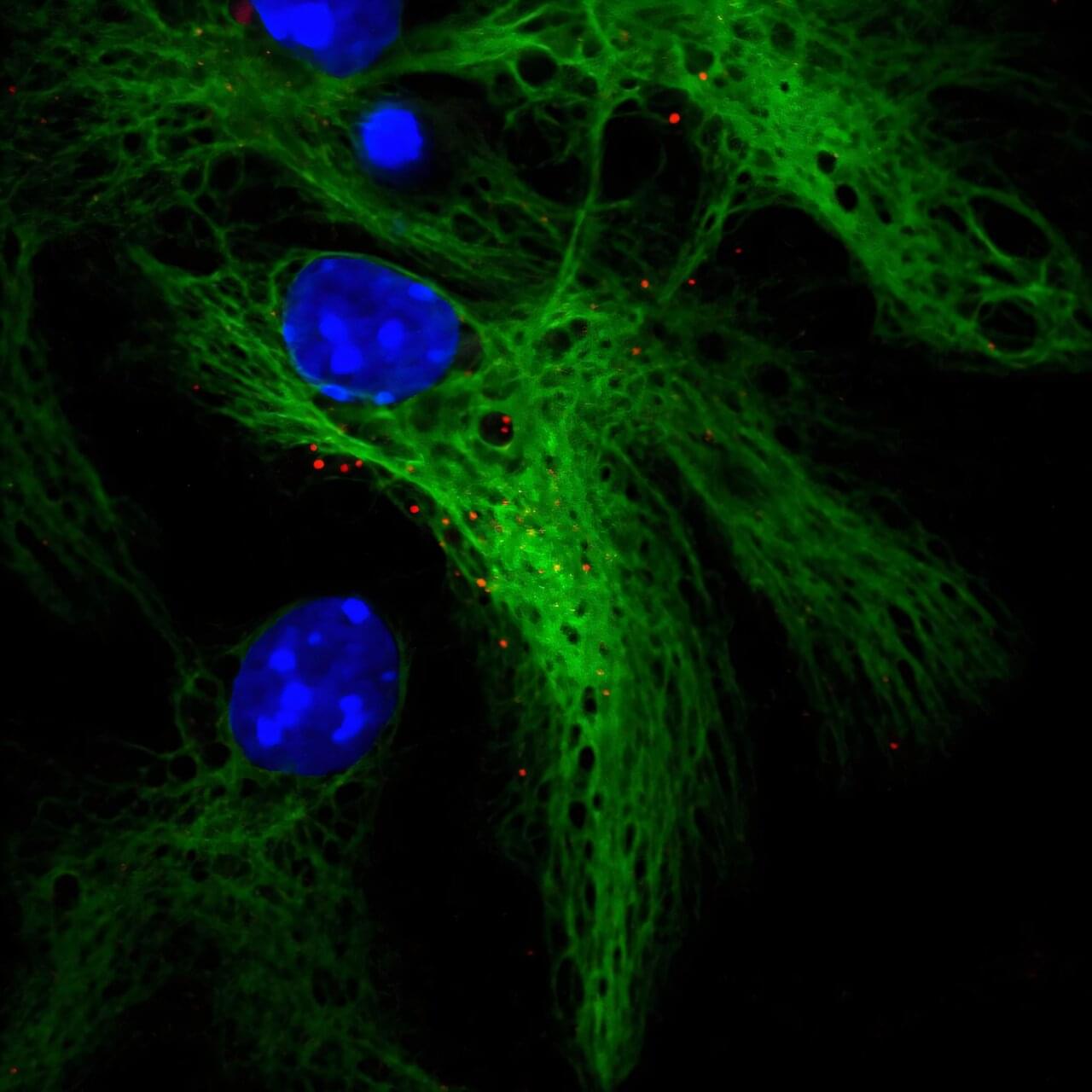
One of the main challenges in the field has been identifying neural stem and progenitor cells (NPCs) responsible for generating these new neurons. NPCs are rare, diverse and difficult to isolate from other brain cells due to overlapping molecular signatures. As a result, understanding their biology—and particularly their role in human brain disorders—has remained elusive.
In a study published in Stem Cell Reports, a team led by researchers at Baylor College of Medicine and the Jan and Dan Duncan Neurological Research Institute (Duncan NRI) at Texas Children’s Hospital reveals specific genes that define NPCs.