In a bid to treat blindness, Life Biosciences will try out potent cellular reprogramming technology on volunteers.
Category: biotech/medical – Page 177
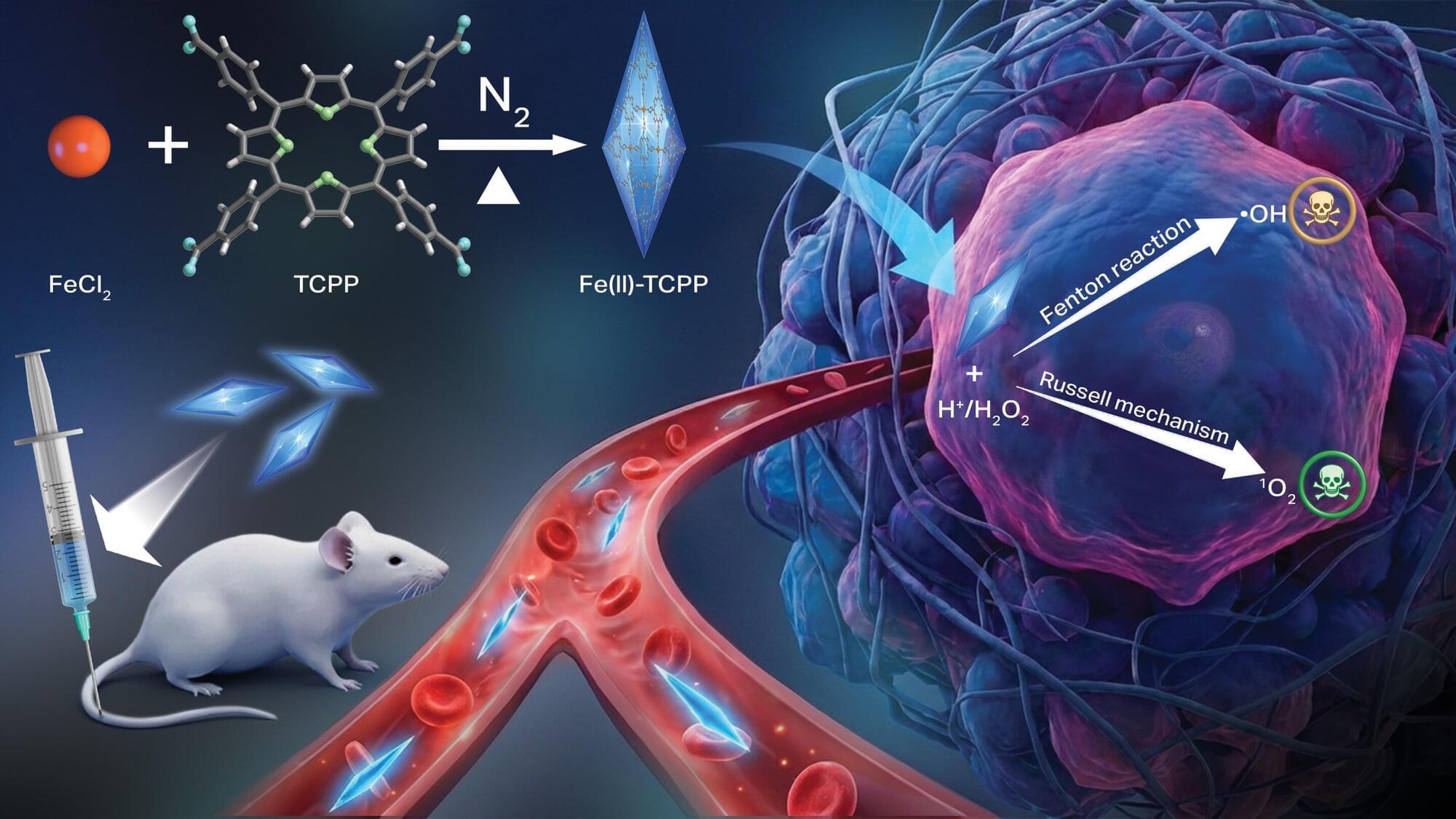
Novel nanomaterial uses oxidative stress to kill cancer cells
Scientists at Oregon State University have developed a new nanomaterial that triggers a pair of chemical reactions inside cancer cells, killing the cells via oxidative stress while leaving healthy tissues alone. The study led by Oleh and Olena Taratula and Chao Wang of the OSU College of Pharmacy appears in Advanced Functional Materials.
The findings advance the field of chemodynamic therapy (CDT), an emerging treatment approach based on the distinctive biochemical environment found in cancer cells. Compared to healthy tissues, malignant tumors are more acidic and have elevated concentrations of hydrogen peroxide, the scientists explain.
Conventional CDT works by using the tumor microenvironment to trigger the chemical production of hydroxyl radicals—molecules, made up of oxygen and hydrogen—with an unpaired electron. These reactive oxygen species are able to damage cells through oxidation by stealing electrons from molecules like lipids, proteins, and DNA.

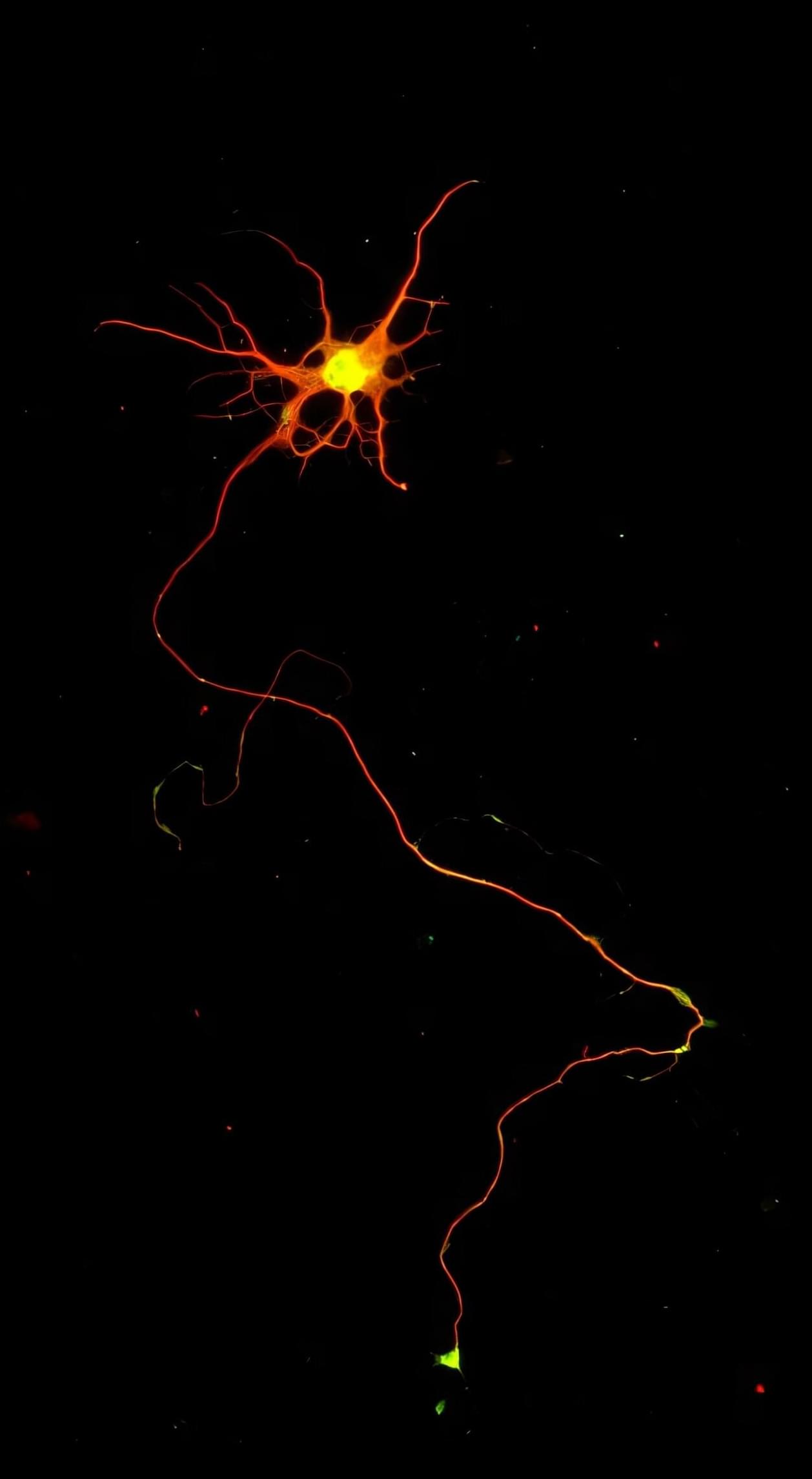
Scientists grow specialized nerve cells that degenerate in ALS and are damaged in spinal cord injury
Researchers have developed a way to grow a highly specialized subset of brain nerve cells that are involved in motor neuron disease and damaged in spinal injuries. Their study, published today in eLife, presents fundamental findings on the directed differentiation of a rare population of special brain progenitors—also known as adult or parent stem cells—into corticospinal-like neurons. The editors note that the work provides compelling data demonstrating the success of this new approach.
The findings set the stage for further research into whether these molecularly directed neurons can form functional connections in the body, and to explore their potential use in human diseases where corticospinal neurons are compromised.
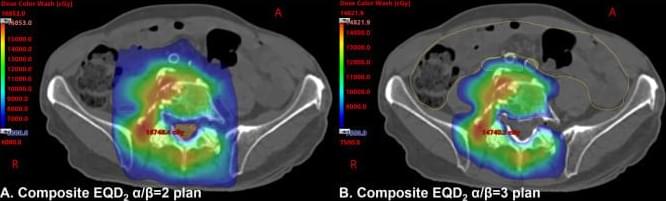
Reirradiation With Stereotactic Body Radiation Therapy for Spinal Metastases: Planning Procedure From a High-Volume Multidisciplinary Spine Oncology Program (SOaR2)
New in practicalRO.
We sought to develop a systematic spine reirradiation planning protocol prioritizing patient safety and maximizing tumor dose delivery. Patients were presented at a Multidisciplinary Spine Oncology Tumor Board to confirm suspicion for recurrent or progressive malignancy and were evaluated in the clinic by the Department of Radiation Oncology and Neurosurgery. Suitable patients proceeded to computed tomography (CT)/magnetic resonance imaging scan simulation. A dedicated physics pathway was activated with the fusion of the magnetic resonance imaging scan and planned CT scan, verified independently by 2 physicists.
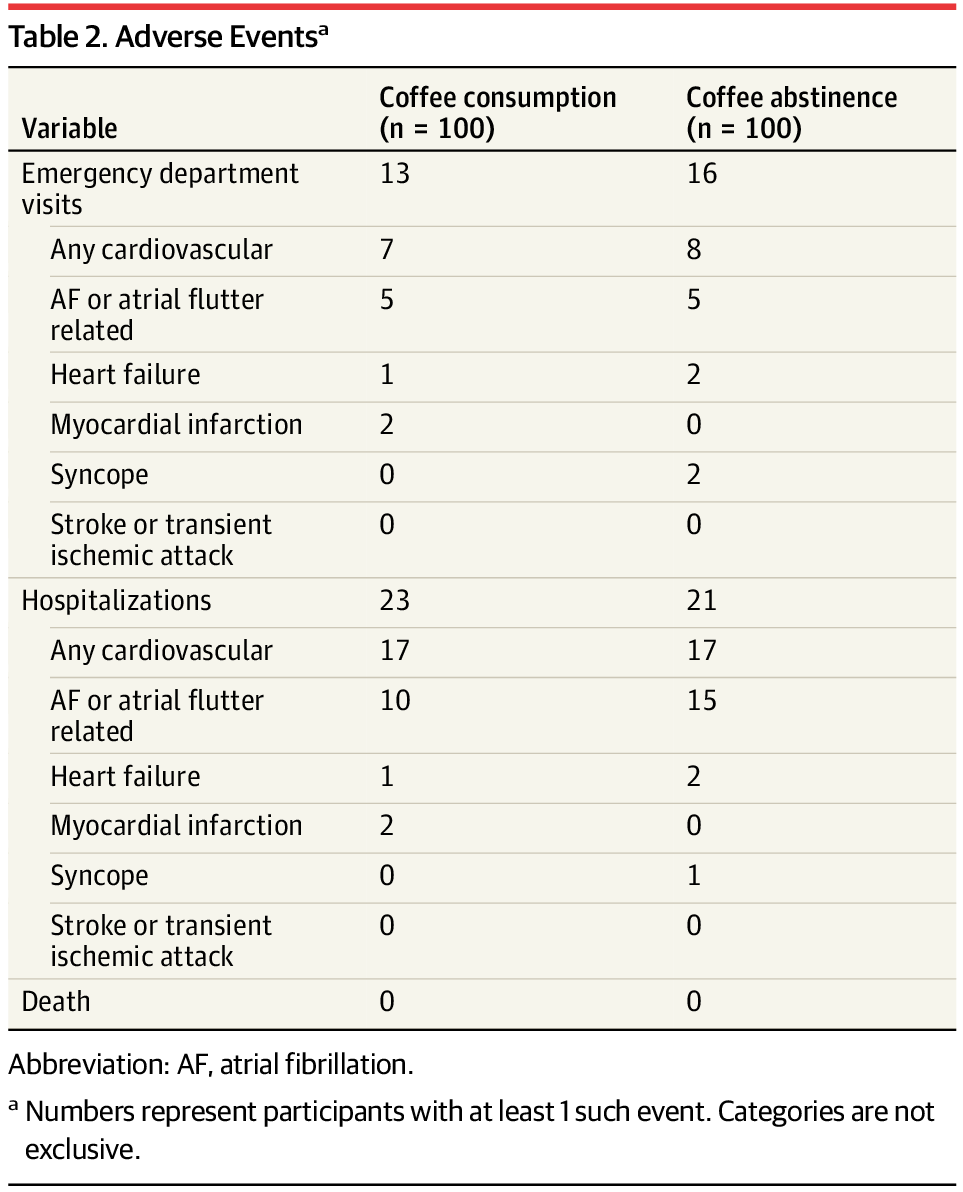
Caffeinated Coffee Consumption or Abstinence to Reduce Atrial Fibrillation: The DECAF Randomized Clinical Trial
RCT: Patients with atrial fibrillation (AF) who continued their usual caffeinated coffee intake after cardioversion experienced less recurrence of AF or atrial flutter compared to those who abstained from coffee and caffeine.
Main Outcomes and Measures The primary end point was clinically detected recurrence of AF or atrial flutter over 6 months.
Results Two hundred patients (mean [SD] age, 69 [11] years; 71% male) were randomized to caffeinated coffee consumption (n = 100) or coffee abstinence (n = 100). Baseline coffee intake was 7 cups (IQR, 7–18) per week in both groups. During follow-up, coffee intake in the consumption and abstinence groups was 7 (IQR, 6–11) and 0 (IQR, 0–2) cups per week, respectively, resulting in a between-group difference of 7 cups (95% CI, 7–7) per week. In the primary analysis, AF or atrial flutter recurrence was less in the coffee consumption (47%) than the coffee abstinence (64%) group, resulting in a 39% lower hazard of recurrence (hazard ratio, 0.61 [95% CI, 0.42–0.89]; P = .01). A comparable benefit of coffee consumption was observed with AF recurrence only. There was no significant difference in adverse events.
Conclusions and Relevance In this clinical trial of coffee drinkers after successful cardioversion, allocation to consumption of caffeinated coffee averaging 1 cup a day was associated with less recurrence of AF or atrial flutter compared with abstinence from coffee and caffeinated products.