A receptor involved in normal physiological processes may also drive breast cancer growth when overexpressed.

Solving Job Loss, and the Future of Work ## Andrew Yang advocates for the implementation of Universal Basic Income (UBI) as a necessary solution to address job loss, income inequality, and societal unrest caused by technological advancements and AI-driven changes in the economy ## ## Questions to inspire discussion.
Universal Basic Income Implementation.
🔹 Q: What UBI amount should be set to provide an effective safety net?
A: UBI should be set at twice the poverty level, around $25,000 per person per year, providing enough for survival but not happiness to maintain work incentives while protecting against economic collapse.
🔹 Q: How can UBI be funded without government action initially?
A: Well-resourced tech billionaires could fund UBI directly to local communities to keep the middle class afloat during AI-driven changes, potentially catalyzing further philanthropy and government action.
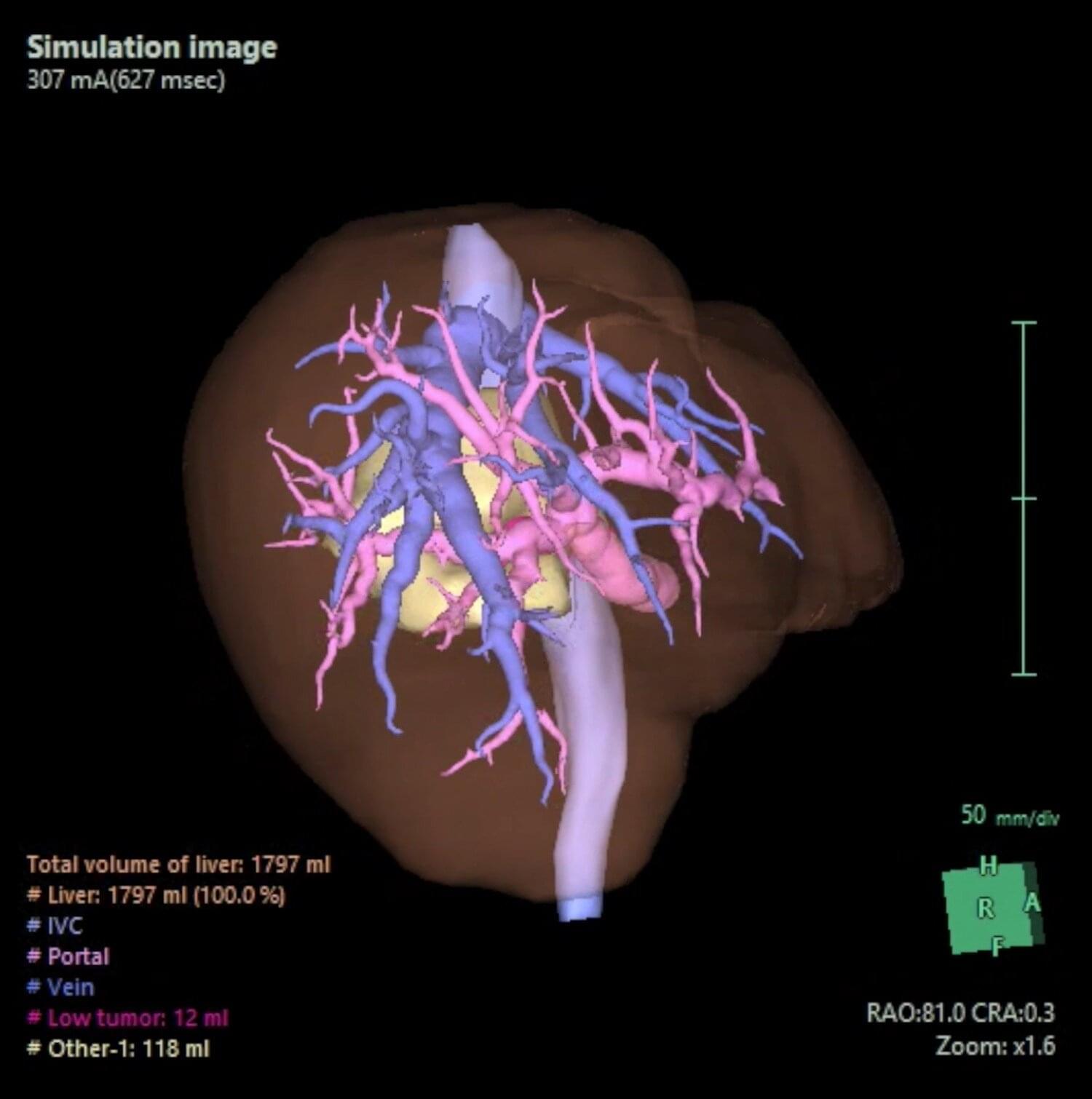
Resection of tumors in the caudate lobe (a deep, hard-to-reach part of the liver) is recognized as one of the most technically challenging procedures in hepatic surgery due to its unique anatomical position and complex vascular relationships. Researchers at Boston University Chobanian & Avedisian School of Medicine now show that it is possible to remove the caudate lobe safely using a surgical robot, even in an older patient, and still remove the cancer completely.
The clinical case they describe in the journal Annals of Surgical Oncology, combines two “guidance” tools: a hanging/traction technique using the Arantius ligament and Indocyanine green (ICG) “negative staining” to clearly mark the caudate lobe boundaries and guide a margin-focused cancer operation in a very difficult area.
“The caudate lobe is one of the most technically demanding areas of the liver—it’s deep and surrounded by critical vessels,” said corresponding author Eduardo Vega, MD, assistant professor of surgery. “Robotic surgery can help us remove select tumors through smaller incisions, with less pain and blood loss and quicker recovery, while still aiming for cure.”
Senescent “zombie” cells accumulate as we age, releasing inflammatory signals that damage surrounding tissue. Senovax takes a novel approach: train the immune system to recognize these cells and eliminate them. By exposing dendritic immune cells to lab-generated senescent cells, the body learns the markers that identify aging cells. The result: the immune system creates a “wanted poster” and begins targeting senescent cells throughout the body. Unlike drugs that must reach specific tissues, the immune system already travels everywhere — and it remembers. One treatment could potentially provide long-lasting protection.
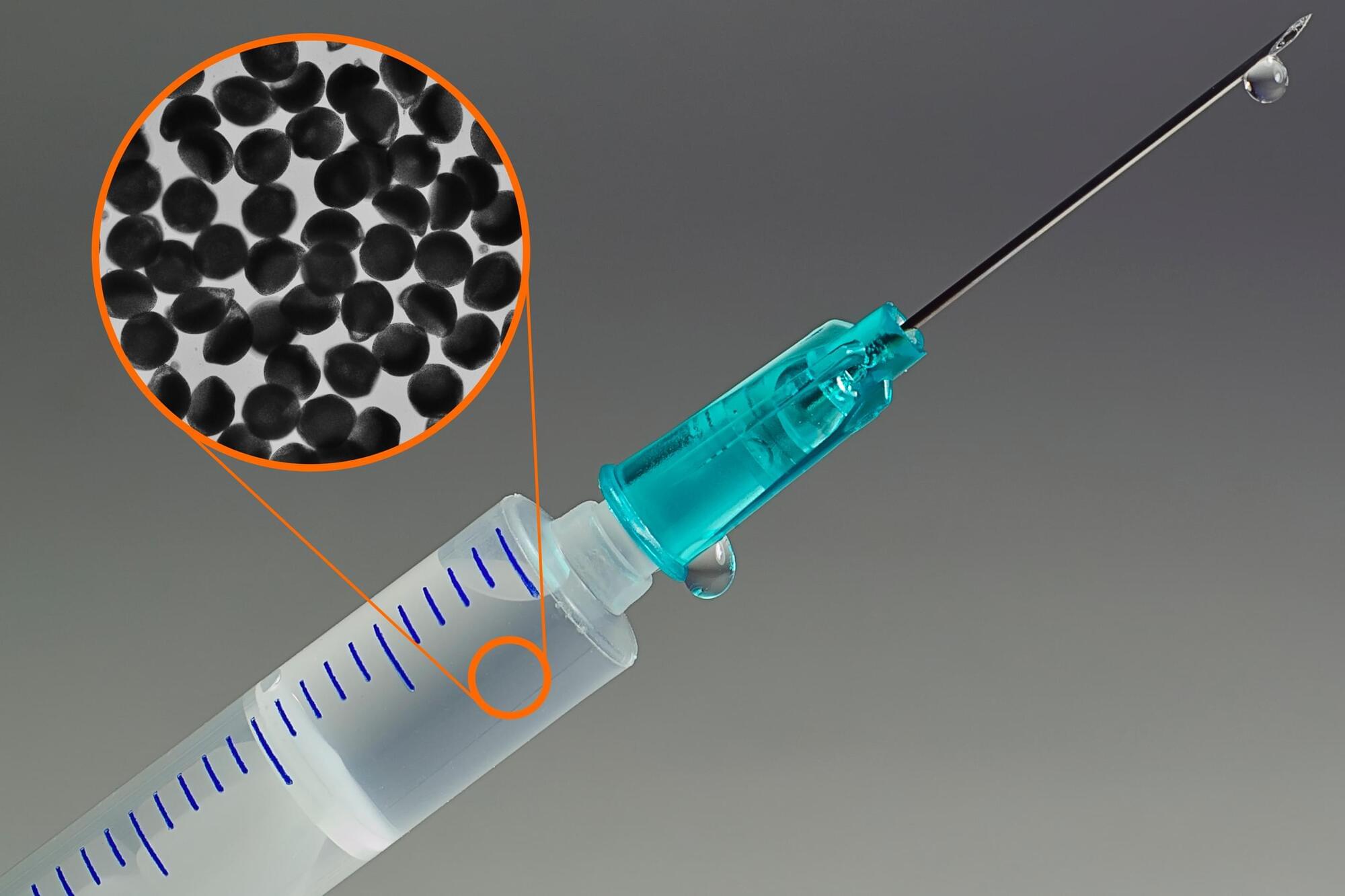
Antibody treatments for cancer and other diseases are typically delivered intravenously, because of the large volumes that are needed per dose. This means the patient has to go to a hospital for every treatment, where they may spend hours receiving the infusion.
MIT engineers have now taken a major step toward reformulating antibodies so that they can be injected using a standard syringe. The researchers found a way to create solid particles of highly concentrated antibodies, suspended in a solution. These particles carry enough antibodies that only about 2 milliliters of solution would be needed per dose.
This advance could make it much easier for patients to receive antibody treatments, and could make treatment more accessible for patients who have difficulty coming into a hospital, including older people.
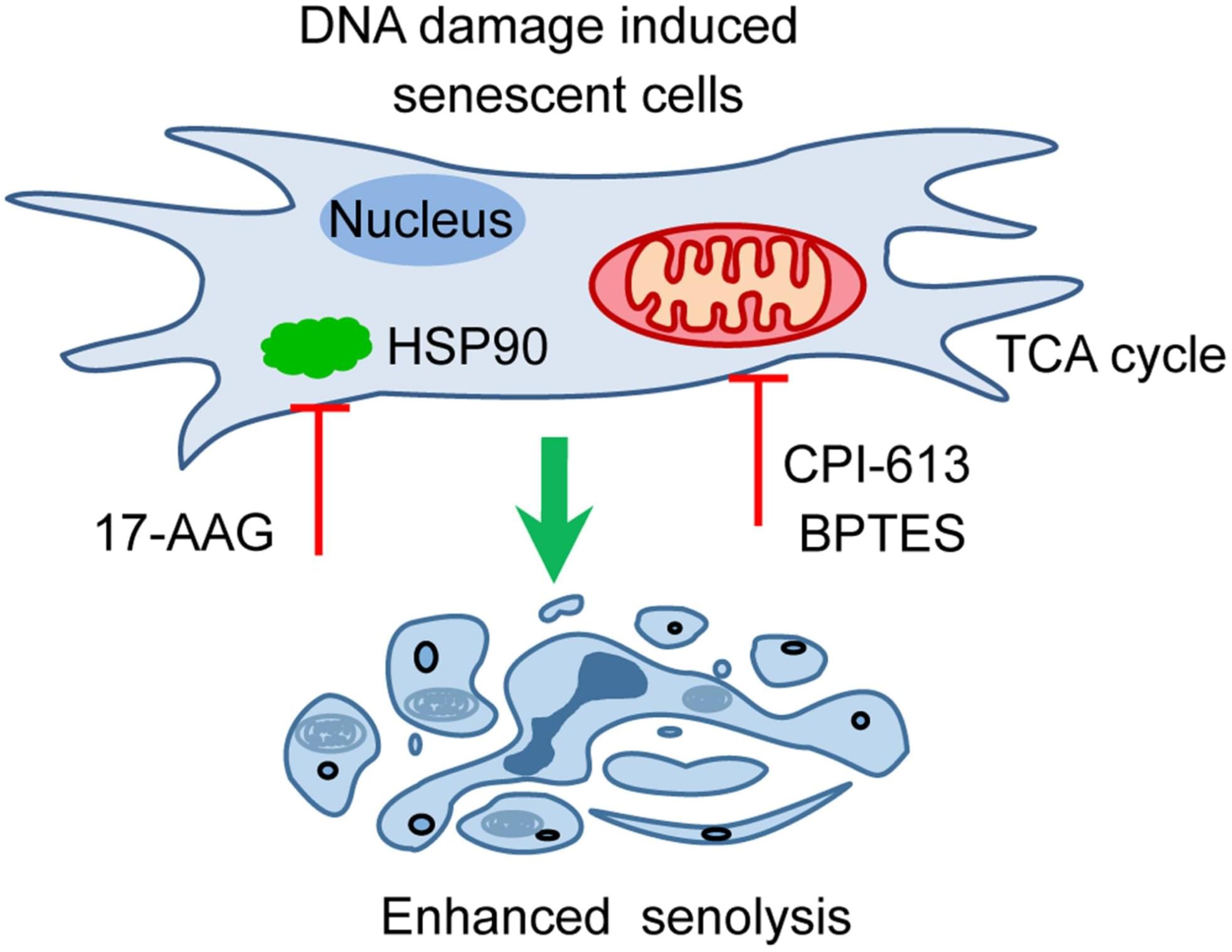
By a global proteomic profiling of senescent human BJ fibroblasts induced by ionizing radiation, 178 cellular proteins with at least 4-fold or greater changes in abundance were identified, representing the cellular landscape of the senescent fibroblasts. Functional enrichments and biological experiments demonstrated that the decreased glucose metabolism, reduced ATP and alpha-KG production, and declined chaperones are the most striking features associated with senescent fibroblasts. Moreover, these proteomic features are closely correlated with their transcription alterations confirmed by RT-PCR. Respectively, inhibiting pyruvate dehydrogenase (critical enzyme to supply acetyl-CoA to TCA cycle) or glutaminase GLS1 (crucial enzyme to supplement TCA cycle intermediate alpha-KG) or inhibiting Hsp90 (important member of chaperones) led to the selective killing of senescent fibroblasts, indicating the essential roles of the TCA cycle or chaperones in the survival and maintenance of cellular senescence. Most importantly, co-inhibiting the TCA cycle and Hsp90 gave rise to the enhanced selective killing of senescent fibroblasts as well as the therapy-induced senescent cancer cells and the alleviation of physical dysfunctions in aged mice, suggesting the synergistic regulation of cellular senescence by the TCA cycle and chaperones. Thus, our profiling revealed key cellular features for the survival and maintenance in senescent normal cells, demonstrating that pyruvate dehydrogenase is a novel and potent senolytic target for the selective elimination of senescence.
Cyclin D-binding myb-like transcription factor 1 or DMTF1a key protein in the brain can help to regenerate neural stem cells and improve aging-associated memory decline. NUS scientists found that this protein’s levels are repressed in the “aged” neural stem cells, Health & Wellness News, Health and Me
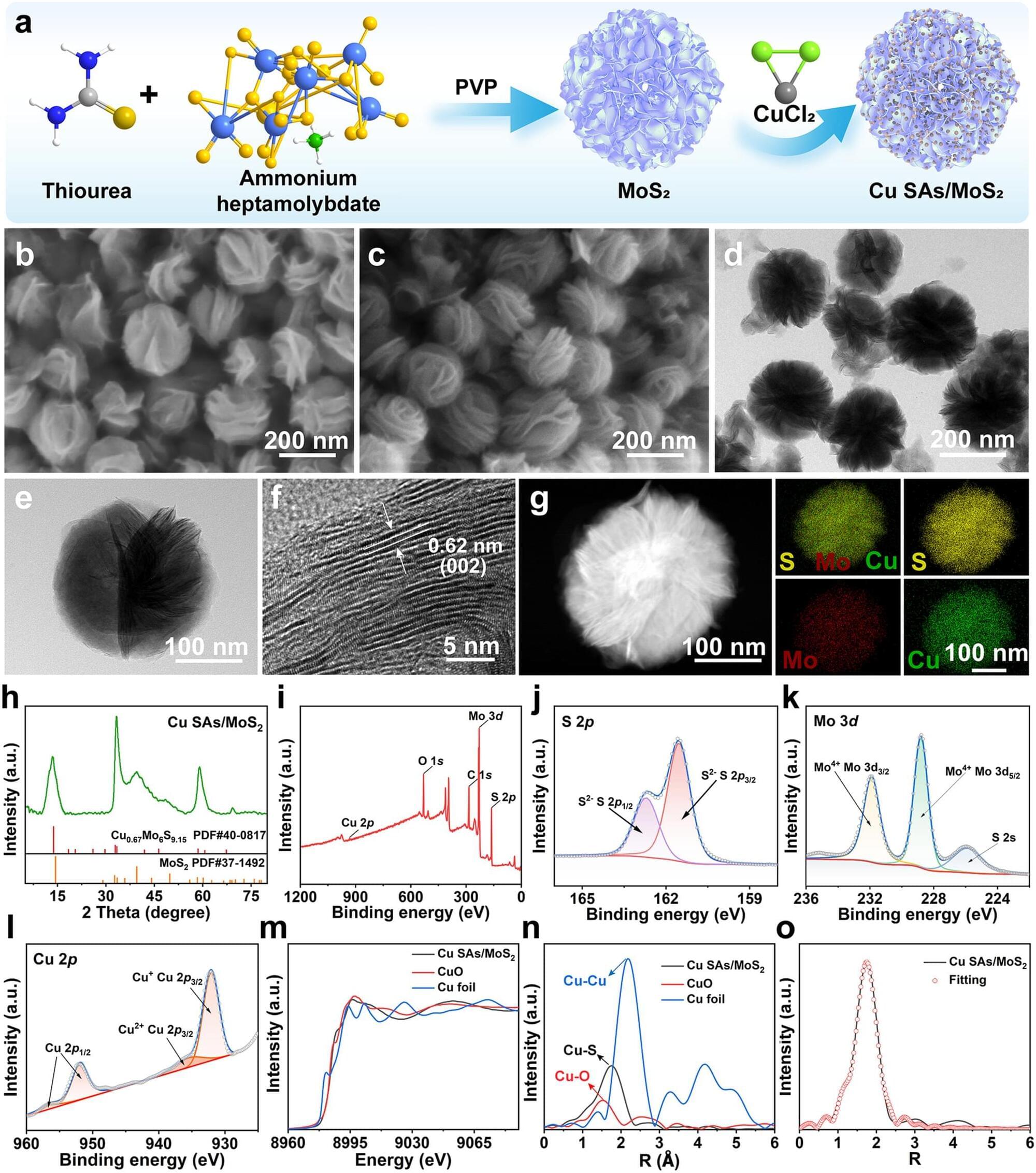
111. Wenqi Wang, Xiaolong Wei, Bolong Xu, Hengshuo Gui, Yan Yan*, Huiyu Liu* & Xianwen Wang* Nano-Micro Lett. 18,111 (2026).
This work is led by Prof. Dr. Xianwen Wang (Anhui Medical University) and co-workers. Prof. Wang’s research centers on burn wounds and tissue regeneration, burn infection, design and development of antimicrobial nanomaterials, development of anti-inflammatory nano-formulations and study on their anti-inflammatory mechanisms. This article develops copper single-atom-loaded MoS₂ nanozymes (Cu SAs/MoS₂) that combat drug-resistant bacteria through a triple mechanism of oxidative damage, cuproptosis-like death, and disrupted cell wall synthesis. Density functional theory reveals that Cu coordination enhances H₂O₂ adsorption, reducing activation energy by 17% and boosting peroxidase-like activity, while glutathione peroxidase-like activity disrupts redox homeostasis and inhibition of peptidoglycan synthesis blocks cell wall remodeling, collectively enabling efficient bacterial killing and decelerating resistance development.
Related articles: Cactus Thorn-Inspired Janus Nanofiber Membranes as a Water Diode for Light-Enhanced Diabetic Wound Healing https://doi.org/10.1007/s40820-025-01904-z Synergistic Ferroptosis–Immunotherapy Nanoplatforms: Multidimensional Engineering for Tumor Microenvironment Remodeling and Therapeutic Optimization https://doi.org/10.1007/s40820-025-01862-6 Wearable Ultrasound Devices for Therapeutic Applications https://doi.org/10.1007/s40820-025-01890-2
The development of highly efficient and multifunctional nanozymes holds promise for addressing the challenges posed by drug-resistant bacteria. Here, copper single-atom-loaded MoS2 nanozymes (Cu SAs/MoS2) were developed to effectively combat drug-resistant bacteria by synergistically integrating the triple strategies of oxidative damage, cuproptosis-like death and disruption of cell wall synthesis. Density functional theory revealed that each Cu center coordinated with three sulfur ligands, enhancing the adsorption of H2O2, which reduced the activation energy of the key step by 17%, thereby improving peroxidase-like (POD-like) activity. The generation of reactive oxygen species in combination with Cu SAs/MoS2 glutathione peroxidase-like (GSH-Px-like) for glutathione scavenging resulted in an imbalance in redox homeostasis within bacteria.
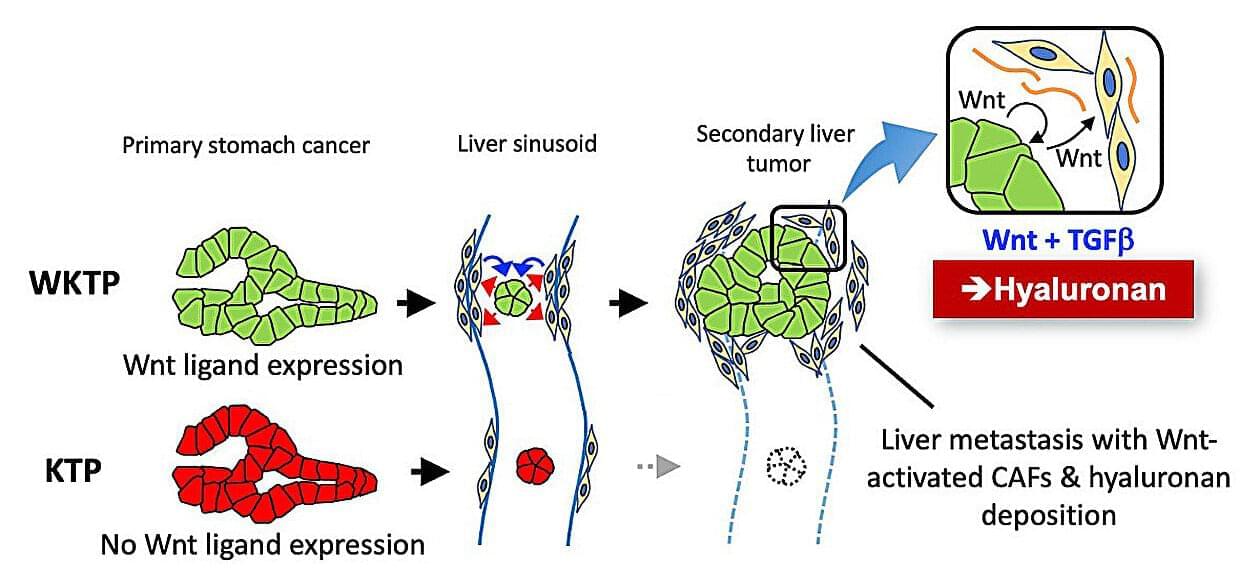
Researchers at the Cancer Research Institute and the Nano Life Science Institute (WPI-NanoLSI), Kanazawa University, have uncovered a critical mechanism that enables gastric cancer to spread to distant organs. Their study shows that cancer cells stimulate Wnt signaling in surrounding stromal fibroblasts to produce hyaluronan, creating a supportive microenvironment that promotes metastasis. These findings provide new insight into how metastatic tumors establish themselves and suggest promising strategies to prevent gastric cancer progression. The work is published in the journal Nature Communications.
Gastric cancer remains one of the leading causes of cancer-related deaths worldwide, largely because it frequently spreads to other organs such as the liver. While genetic mutations that initiate tumors have been extensively studied, the biological mechanisms that allow cancer cells to colonize new tissues remain poorly understood.
“Wnt signaling”—a pathway essential for stem cell maintenance and tissue regeneration—is often activated in gastric cancer through external ligand stimulation rather than genetic mutation. This study further identifies that Wnt signaling in the tumor microenvironment also plays a crucial role in disease progression.
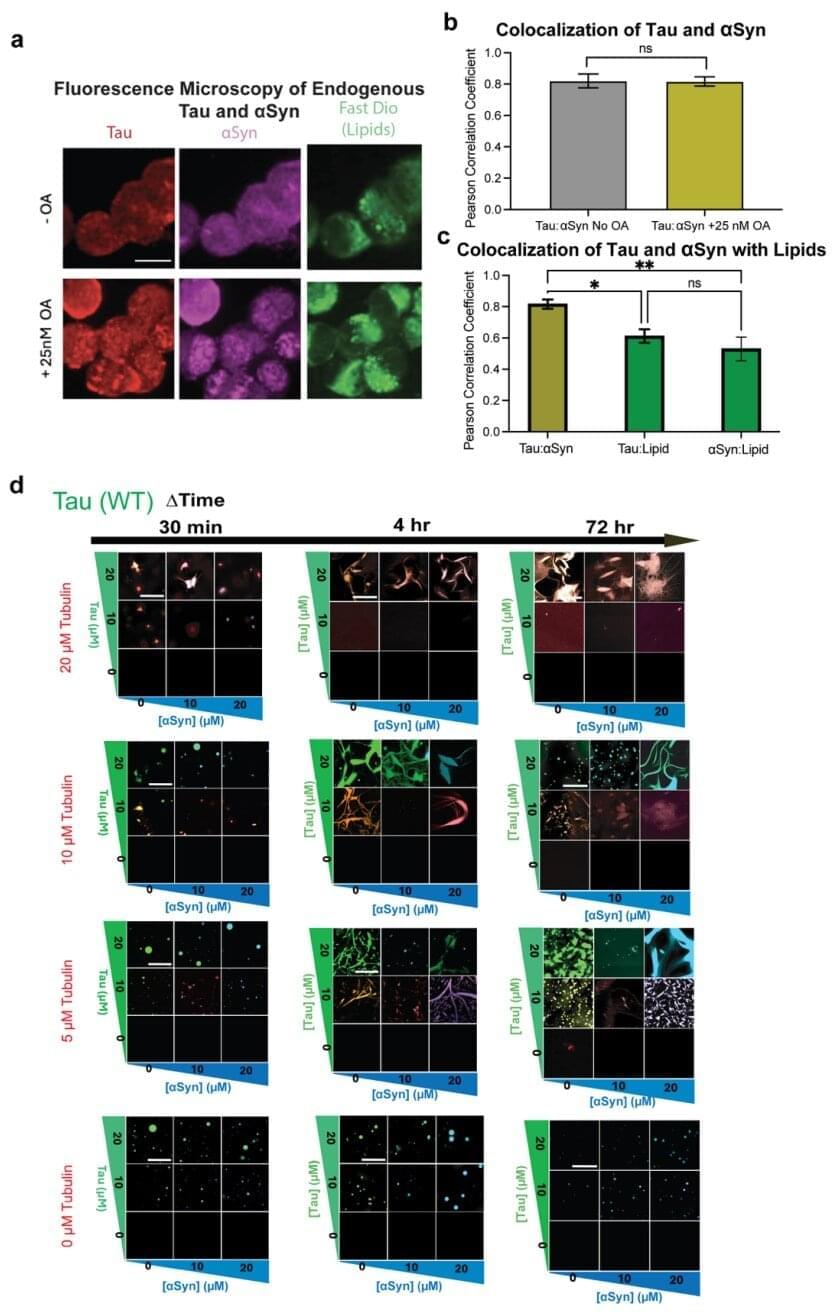
Researchers at Baylor College of Medicine have discovered a potential new strategy to fight back against Alzheimer’s and Parkinson’s diseases, conditions that are linked to the toxic accumulation of Tau and alpha synuclein protein clumps in the brain. The team reports in Nature Communications that tubulin, the building block of microtubules, the cell’s internal ‘railway tracks, can stop Tau and alpha synuclein from forming toxic clumps and instead steer them into their normal, healthy roles.
“Tau and alpha synuclein are well known for their roles in neurodegenerative diseases like Alzheimer’s and Parkinson’s. In these conditions, these proteins can misfold, stick together and form harmful aggregates that damage neurons and contribute to memory loss, movement problems and other symptoms,” said first author Dr. Lathan Lucas, postdoctoral associate of biochemistry and molecular pharmacology in Dr. Allan Ferreon’s lab.
“But Tau and alpha synuclein also fulfill essential functions in healthy neurons—they help maintain cell structure and support communication by interacting with tubulin and contributing to microtubule assembly and stabilization.”