Could Alien Civilizations Build Dyson Bubbles? Scientists Say Stability Might Be Achievable
Get the latest international news and world events from around the world.
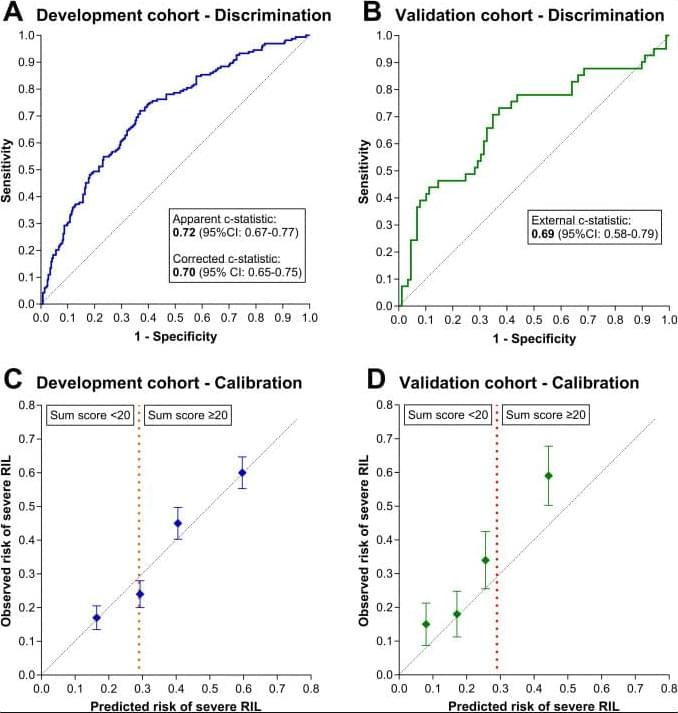
Pretreatment risk model for radiation-induced lymphopenia is associated with adjuvant durvalumab efficacy in patients with unresectable stage III NSCLC
Can we predict immunotherapy success by forecasting severe radiation-induced lymphopenia (RIL)? A pretreatment NSCLC nomogram shows only low RIL-risk patients benefit from adjuvant durvalumab, potentially enabling personalized RIL mitigation and optimized immunotherapy. Read it here in the RedJournal.
Severe radiation-induced lymphopenia (RIL) during concurrent chemoradiotherapy (CCRT) for NSCLC has been associated with poorer outcomes and reduced immunotherapy efficacy. Because RIL often develops late during CCRT, identifying patients at risk before treatment may be clinically relevant. This study aimed to develop and validate a nomogram based on pretreatment predictors for severe RIL, and secondarily to explore associations between predicted RIL risk and adjuvant durvalumab-associated survival.
Tubulin prevents toxic protein clump formation
“This led us to the following idea: what if instead of preventing the formation of droplets, we created conditions that would drive Tau and alpha synuclein inside the droplets toward their healthy path, discouraging them from taking the disease path?” said a co-corresponding author of the work.
The team worked with biochemical and biophysical techniques, high-resolution microscopy and neuronal-based assays to investigate tubulin’s role in modulating and preventing the formation of toxic aggregates in droplets.
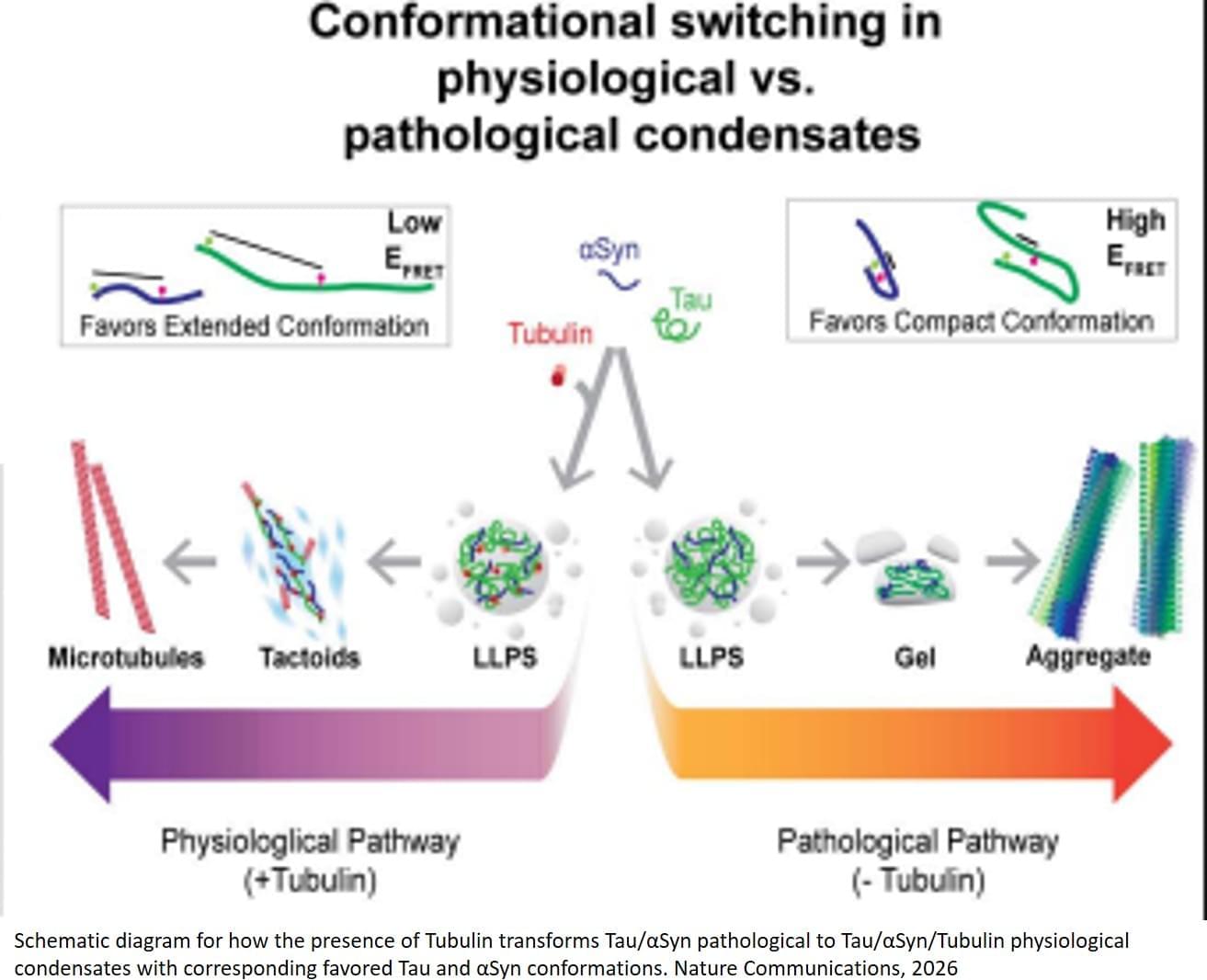
The researchers show that Tubulin modulates Tau:αSyn condensates by promoting microtubule interactions and inhibiting homotypic and heterotypic pathological oligomers. Tubulin partitioning into condensates promotes microtubule polymerization and prevents Tau and αSyn oligomerization.
In the absence of Tubulin, Tau-driven condensation accelerates formation of pathogenic Tau:αSyn heterodimers and amyloid fibrils. The authors also identify distinct Tau and αSyn structural states in pathological Tubulin-absent versus physiological Tubulin-rich condensates.
“When tubulin levels are low, as it has been found in Alzheimer’s disease, microtubules are less abundant and Tau and alpha synuclein can form toxic aggregates,” the author said. “But when tubulin is present, Tau and alpha‑synuclein shift away from harmful aggregates and instead promote the assembly of healthy microtubules,” the author said. “Tubulin redirects the activity of these proteins by giving them something productive to do.” ScienceMission sciencenewshighlights.
Researchers have discovered a potential new strategy to fight back against Alzheimer’s and Parkinson’s diseases, conditions that are linked to the toxic accumulation of Tau and alpha synuclein protein clumps in the brain. The team reports in Nature Communications that tubulin, the building block of microtubules, the cell’s internal ‘railway tracks,’ can stop Tau and alpha synuclein from forming toxic clumps and instead steer them into their normal, healthy roles.
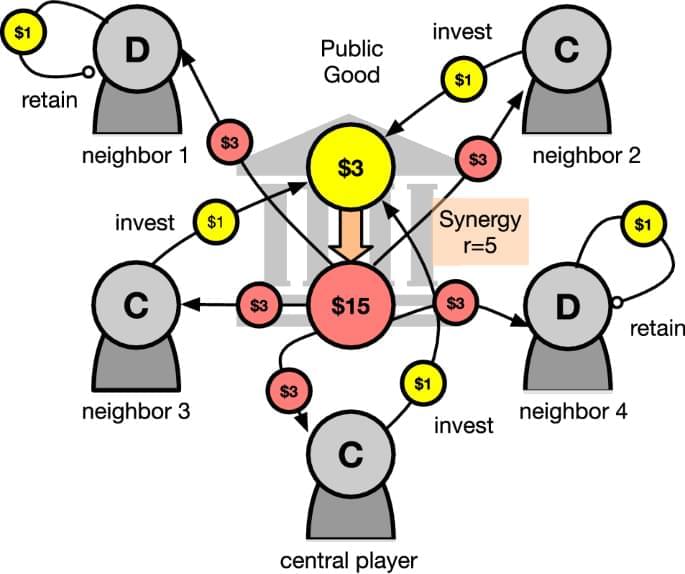
Promoting cooperation in the public goods game using artificial intelligent agents
Hintze, A., Adami, C. Promoting cooperation in the public goods game using artificial intelligent agents. npj Complex 3, 3 (2026). https://doi.org/10.1038/s44260-025-00065-9
Scientists discover the switch that revives exhausted cancer-fighting T cells
Scientists have uncovered new genetic rules that determine whether the immune system’s “killer” T cells remain powerful long-term defenders or become worn out and ineffective. By building a detailed genetic atlas of CD8 T cell states, researchers identified key molecular switches that push these cells toward either resilience or exhaustion. Remarkably, disabling just two previously unknown genes restored the tumor-killing power of exhausted T cells while preserving their ability to provide lasting immune protection.

How AI could unlock deep-sea secrets of marine life
The reef is a home and feeding ground for dozens of species that depend on it the way a woodland creature depends on trees. It has survived ice ages – but whether it will survive increasing pressures from industrial fishing, deep-sea mining and climate change is, in part, a question about data. If we don’t know it exists, how can we protect it?
A new project called Deep Vision could fundamentally transform our understanding of the deep ocean by digging into pictures and videos sat largely unexamined in research archives around the world. By using AI, thousands of hours of seafloor footage can be analysed to produce the first comprehensive maps of vulnerable marine ecosystems across the entire Atlantic basin.
Over the past two decades, robotic and autonomous underwater vehicles have collected vast quantities of footage from the deep sea. This represents an extraordinary resource – a record of ecosystems that most humans will never see.