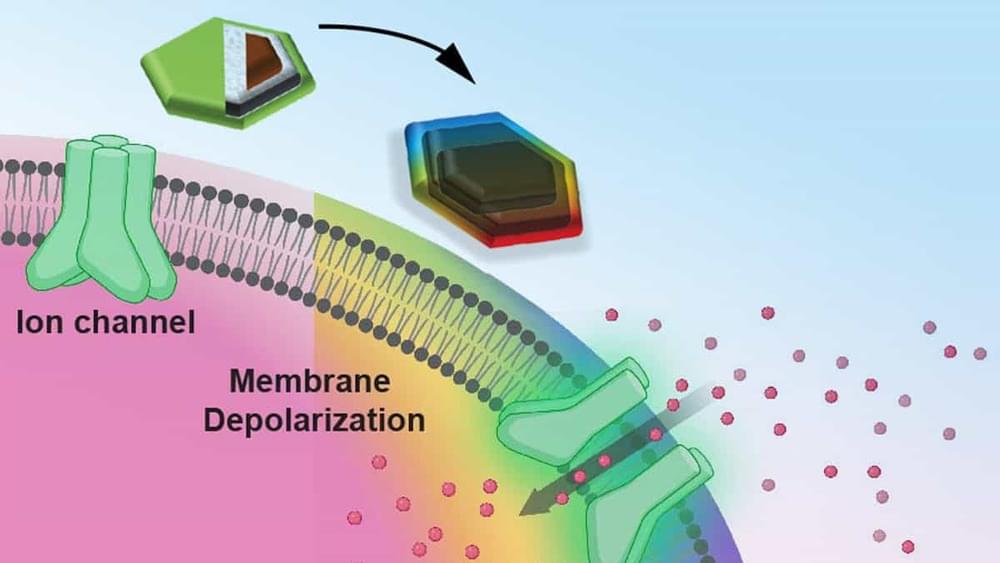
Injectable magnetoelectric nanodiscs may activate neurons in localized brain regions when stimulated by a weak external magnetic field, say MIT scientists.

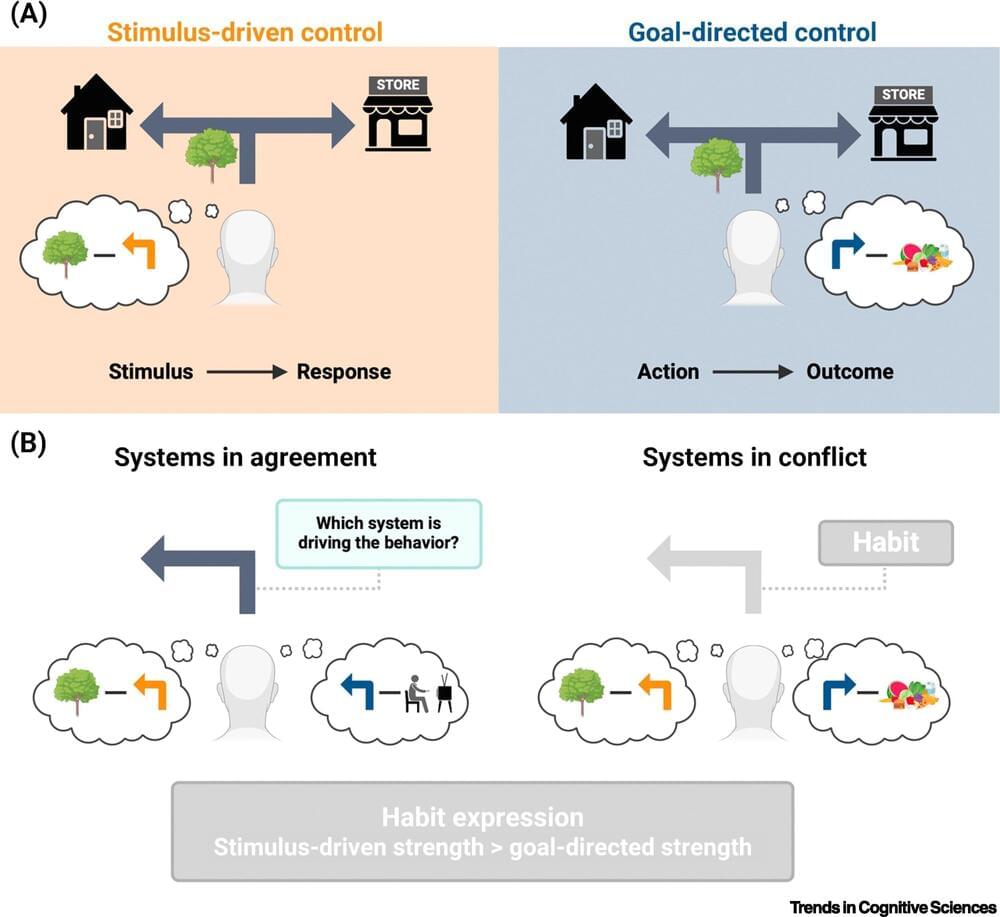
Cognitive neuroscientists at Trinity College Dublin have published new research describing a brand new approach to making habit change achievable and lasting.
This innovative framework has the potential to significantly improve approaches to personal development, as well as the clinical treatment of compulsive disorders (for example obsessive compulsive disorder, addiction, and eating disorders).
The research was led by Dr. Eike Buabang, Postdoctoral Research Fellow in the lab of Professor Claire Gillan in the School of Psychology, has been published as a paper titled “Leveraging cognitive neuroscience for making and breaking real-world habits” in the journal Trends in Cognitive Sciences.

Life is a series of small events: making morning coffee, letting the dog out, opening a laptop, letting the dog back in. Add them all up and you have a full day. Our brains are committed to observing and processing the events that make up our daily lives, said Jeff Zacks, the Edgar James Swift Professor in Arts & Sciences and chair of the Department of Psychological & Brain Sciences. “Knowing where events begin and where they end is crucial to understanding the world,” Zacks said.
In a pair of new papers, Zacks and other researchers in Arts & Sciences and the McKelvey School of Engineering explore this key process of human cognition.
Zacks led a study that trained computer models to observe more than 25 hours of video of people performing simple, everyday tasks such as cleaning a kitchen or cooking a meal before making predictions about what happens next. The study came to a surprising conclusion: The computer models were most accurate when they responded to uncertainty. When the model was especially unsure about what would happen next, it would reset and reassess the scene, an approach that improved its overall comprehension.

A new study demonstrates that even simple single-cell organisms, such as ciliates and amoebae, exhibit habituation, a basic form of learning previously thought to be exclusive to more complex beings.
This revelation not only changes our understanding of cellular capabilities but also opens up possibilities for applications in cancer immunology, suggesting that our immune cells might be reprogrammed to better recognize and attack cancer cells.
A dog learns to sit on command. A person tunes out the steady hum of a washing machine while engrossed in a book. The ability to learn and adapt is a cornerstone of evolution and survival.

Scientists have discovered your cholesterol levels could be significantly linked to your risk of developing dementia. And it’s not just high cholesterol that matters: fluctuating levels over several years could increase your chances of the disease by 60 per cent, suggests a new study of 10,000 people.
The research also suggests that, even if you don’t develop dementia, a large cholesterol variability – swinging from high to low levels – is linked to an increased risk of general cognitive decline by 23 per cent.

And should we — abolish death? Neuroscientist and author Ariel Zeleznikow-Johnston will be speaking at Bush House, King’s College London, on Tues 3rd December, in an event that London Futurists is happy to draw to the attention of all members and friends.
In this event organised by King’s College London, neuroscientist and author Ariel Zeleznikow-Johnston will be speaking about his new book The Future Loves You, which explores how brain preservation techniques might preserve us forever.
Ariel will be in discussion with historians, literary scholars, ethicists, and futurists, including Richard Ashcroft, Steve Connor, Caitjan Gainty, Catriona Byers, and Fay Bound Alberti.
This DFI Centre for Technology and the Body event will be held on 3 December at 6pm-8.30pm at Bush House, Lecture Room 2.
A series of studies on humans and mice examined sex differences in reactions to anesthetics, revealing that female brains are more resistant to the hypnotic effects of these drugs. Testosterone administration increased sensitivity to anesthetics in mice, while castration enhanced anesthetic resistance. In humans, females regained consciousness and recovered cognitive function faster than males after identical exposure to anesthetics. The study was published in Neuroscience.
General anesthetics are drugs that induce a reversible loss of consciousness, primarily used during surgical procedures to block pain and prevent awareness. They are essential in medicine because they enable complex surgeries that would otherwise be intolerable due to pain, allowing patients to undergo invasive procedures safely and comfortably.
The history of general anesthesia dates back to the 19th century, with the first successful public demonstration by Dr. William Morton in 1846. Before anesthetics, surgery was excruciating and dangerous, often performed only in dire cases due to the severe pain and risks. Over time, safer and more effective agents, such as chloroform and eventually modern inhaled and intravenous anesthetics, were developed. Today, general anesthesia is administered by specialized professionals called anesthesiologists, who monitor and adjust the dosage to ensure patient safety.
Brain-Computer Interfaces fascinate the sci-fi and medical communities in equal measure. Here’s how close the transformative technology is to everyday use.
