The decision by the L.A. County public health system not to test patients with coronavirus symptoms could make it difficult to know how many people are infected.



Bulky, buzzing and beeping hospital rooms demonstrate that monitoring a patient’s health status is an invasive and uncomfortable process, at best, and a dangerous process, at worst. Penn State researchers want to change that and make biosensors that could make health monitoring less bulky, more accurate—and much safer.
The key would be making sensors that are so stretchable and flexible that they can easily integrate with the human body’s complex, changing contours, said Larry Cheng, the Dorothy Quiggle Professor in Engineering and an affiliate of the Institute for Computational and Data Sciences. His lab is making progress on designing sensors that can do just that.
If biosensors that are both energy efficient and stretchable can be achieved at scale, the researchers suggest that engineers can pursue—and, in some cases, are already pursuing—a range of options for sensors that can be worn on the body, or even placed inside the body. The payoff would be smarter, more effective and more personalized medical treatment and improved health decision-making—without a lot of bulky, buzzing and beeping pieces of monitoring equipment.
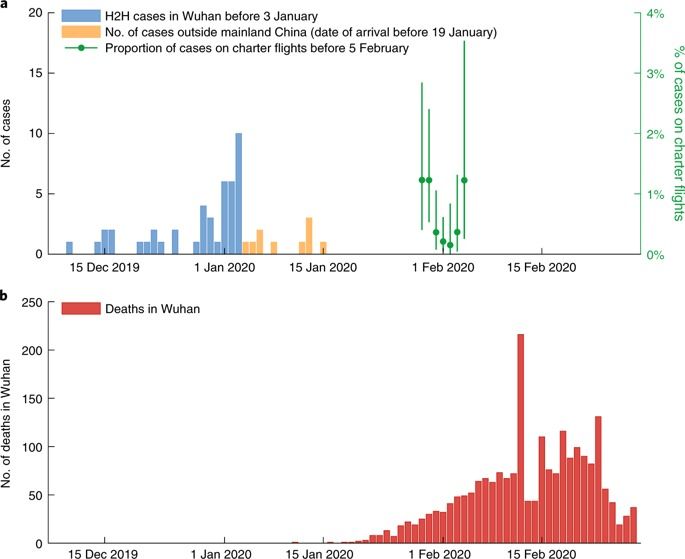
As of 29 February 2020 there were 79,394 confirmed cases and 2,838 deaths from COVID-19 in mainland China. Of these, 48,557 cases and 2,169 deaths occurred in the epicenter, Wuhan. A key public health priority during the emergence of a novel pathogen is estimating clinical severity, which requires properly adjusting for the case ascertainment rate and the delay between symptoms onset and death. Using public and published information, we estimate that the overall symptomatic case fatality risk (the probability of dying after developing symptoms) of COVID-19 in Wuhan was 1.4% (0.9–2.1%), which is substantially lower than both the corresponding crude or naïve confirmed case fatality risk (2,169/48,557 = 4.5%) and the approximator1 of deaths/deaths + recoveries (2,169/2,169 + 17,572 = 11%) as of 29 February 2020. Compared to those aged 30–59 years, those aged below 30 and above 59 years were 0.6 (0.3–1.1) and 5.1 (4.2–6.1) times more likely to die after developing symptoms. The risk of symptomatic infection increased with age (for example, at ~4% per year among adults aged 30–60 years).

This Is-Real!!! Israeli pharmaceutical company, Teva, has announced that they will donate more than 6 million tablets through wholesalers to hospitals across the United States, from March 31.
As the coronavirus is spreading across the world, and the number of people infected is increasing everyday, there is an urgent need to find treatments against COVID-19 that could reduce complications and improve recovery. Recently, the Israeli Health Ministry has approved multiple experiment treatments, and companies worldwide are attempting to determine what could be used to treat COVID-19.
As such, Israeli pharmaceutical company, Teva, has announced that they will donate more than 6 million doses of hydroxychloroquine sulfate tablets through wholesalers to hospitals across the United States, from March 31. Over 10 million tablets are expected to be shipped within a month.


In addition to working with the World Health Organization to end smallpox, Larry Brilliant has fought flu, polio, and blindness. He says we will, eventually, get back to normal. But that’s not going to occur until three important things happen first. LARRY BRILLIANT SAYS he doesn’t have a crystal ball. But 14 years ago, Brilliant, the epidemiologist who helped eradicate smallpox, spoke to a TED audience and described what the next pandemic would look like. At the time, it sounded almost too horrible to take seriously. “A billion people would get sick,” he said. “As many as 165 million people would die. There would be a global recession and depression, and the cost to our economy of $1 to $3 trillion would be far worse for everyone than merely 100 million people dying, because so many more people would lose their jobs and their health care benefits, that the consequences are almost unthinkable.”
Epidemiologist Larry Brilliant, who warned of pandemic in 2006, says we can beat the novel coronavirus—but first, we need lots more testing.

ZURICH (Reuters) — Switzerland’s health care system could collapse by the end of the month if the new coronavirus keeps spreading at current rates, a government official warned on Tuesday.
Swiss authorities estimated that 2,650 people had tested positive for the coronavirus and said 19 people had died, while predicting cases will likely soar in the weeks ahead.
Exact figures were unavailable. Daniel Koch, head of the Federal Office of Health’s communicable diseases division, said the rapid rise had outstripped the state’s ability to record new cases in real time.
Sam Harris discusses the coronavirus withAmesh Adalja.
In this episode of the podcast, Sam Harris speaks with Amesh Adalja about the spreading coronavirus pandemic. They discuss the contagiousness of the virus and the severity of the resultant illness, the mortality rate and risk factors, vectors of transmission, how long coronavirus can live on surfaces, the importance of social distancing, possible anti-viral treatments, the timeline for a vaccine, the importance of pandemic preparedness, and other topics.
Amesh Adalja, MD, is an infectious disease specialist at the Johns Hopkins University Center for Health Security. His work is focused on emerging infectious disease, pandemic preparedness, and biosecurity. Amesh has served on US government panels tasked with developing guidelines for the treatment of plague, botulism, and anthrax. He is an Associate Editor of the journal Health Security, co-editor of the volume Global Catastrophic Biological Risks, and a contributing author for the Handbook of Bioterrorism and Disaster Medicine. Amesh actively practices infectious disease, critical care, and emergency medicine in the Pittsburgh metropolitan area.
Website: www.trackingzebra.com
Twitter: @AmeshAA
Francisco Garcia, a youth team coach at Malaga-based club Atletico Portada Alta, had an unknown pre-existing health condition that resulted in him being more vulnerable to the virus than usual for an individual of his age, though he was only informed of having cancer after going to hospital with symptoms of coronavirus.
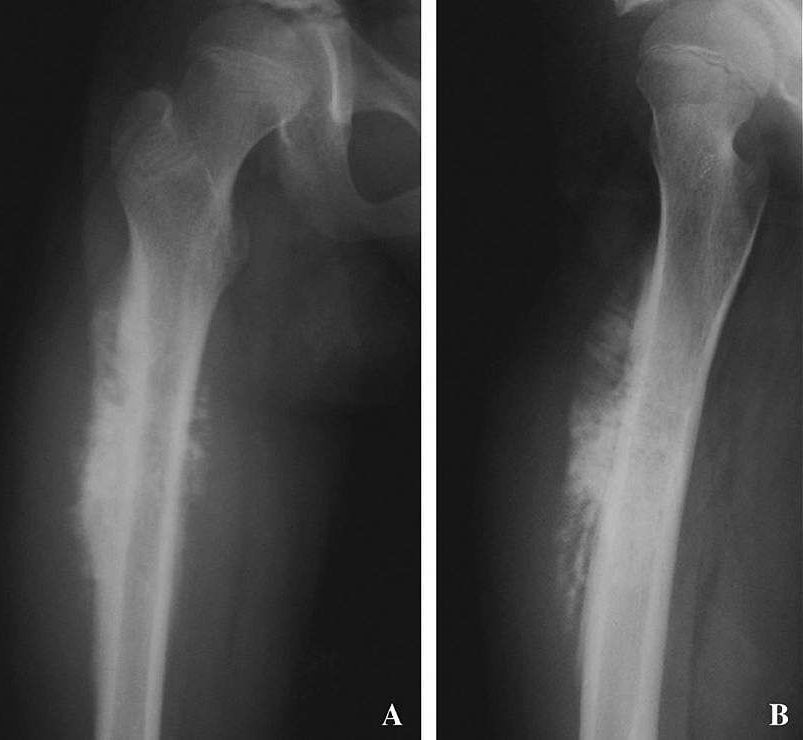
A study by researchers at the National Cancer Institute (NCI), part of the National Institutes of Health, offers new insight into genetic alterations associated with osteosarcoma, the most common cancerous bone tumor of children and adolescents. The researchers found that more people with osteosarcoma carry harmful, or likely harmful, variants in known cancer-susceptibility genes than people without osteosarcoma. This finding has implications for genetic testing of children with osteosarcoma, as well as their families.
The study was published March 19, 2020, in JAMA Oncology.
“With this study, we wanted to find out how many people with osteosarcoma may have been at high risk for it because of their genetics,” said Lisa Mirabello, Ph.D., of NCI’s Division of Cancer Epidemiology and Genetics (DCEG), who led the research. “We not only learned that at least a quarter of the people in the study with osteosarcoma had a variant in a gene known to predispose someone to cancer, we also uncovered variants that had never before been associated with this cancer.”