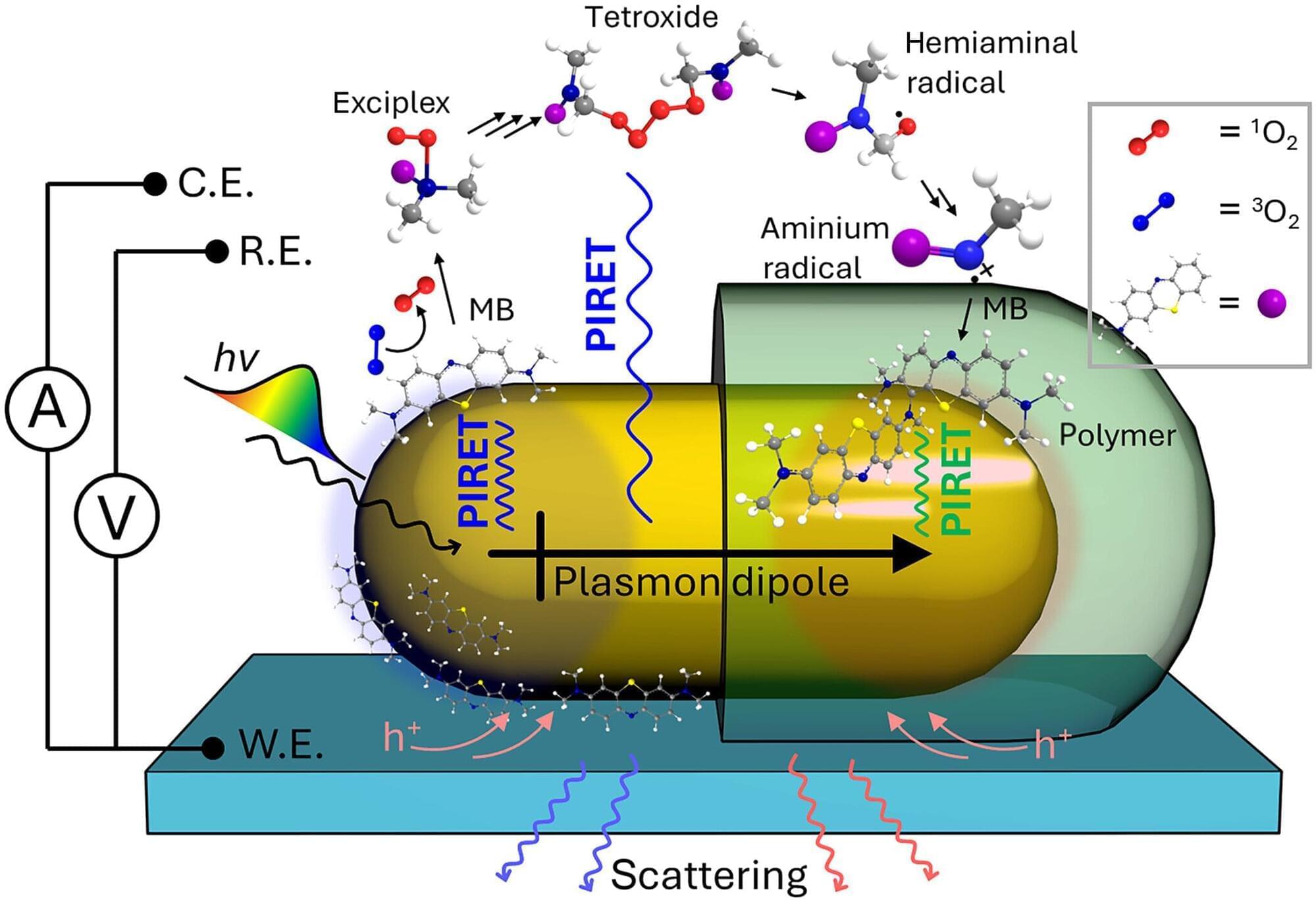
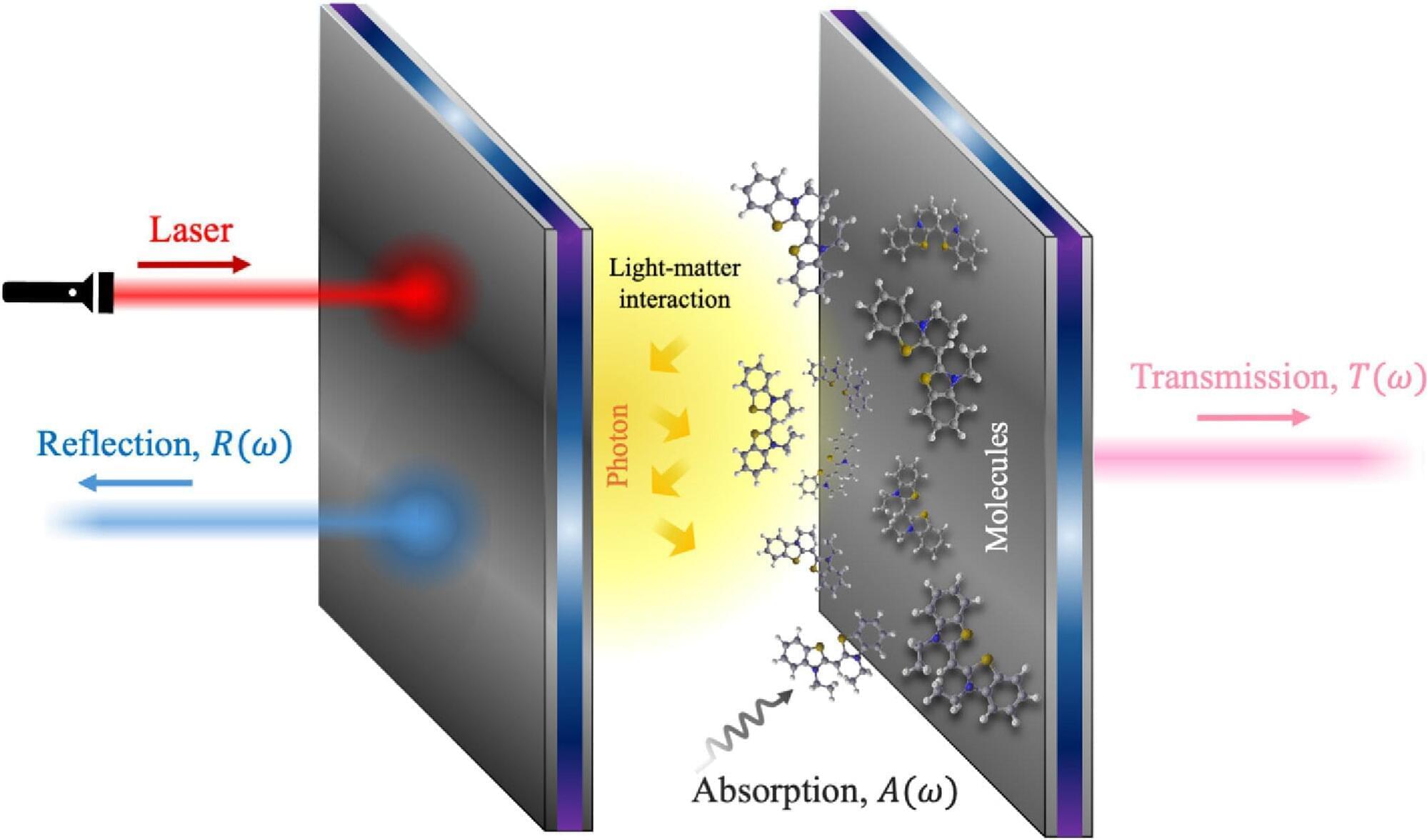
Scientists are exploring many ways to use light rather than heat to drive chemical reactions more efficiently, which could significantly reduce waste, energy consumption, and reliance on nonrenewable resources.
A team of chemistry researchers at the University of Illinois Urbana-Champaign has been studying plasmon-induced resonance energy transfer (PIRET)—conveying energy from a tiny metal particle to a semiconductor or molecule without the need for any physical contact.
“If you’d like to do chemistry with light, then your first step would be to use that light as efficiently as possible,” said Illinois chemistry professor Christy Landes, who co-leads the research team exploring this innovative research. “And one of the most efficient ways to use light is to use plasmonic metal nanoparticles, because they are better than just about any other material at absorbing and scattering light.”