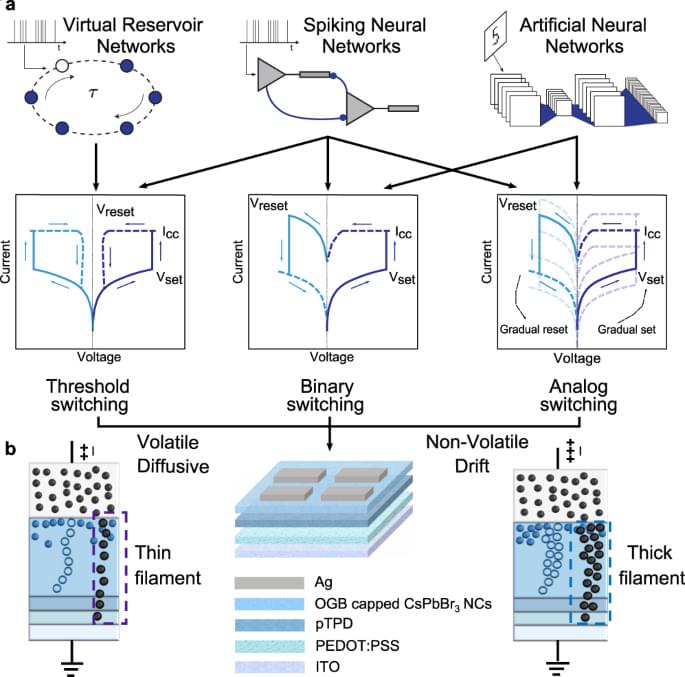
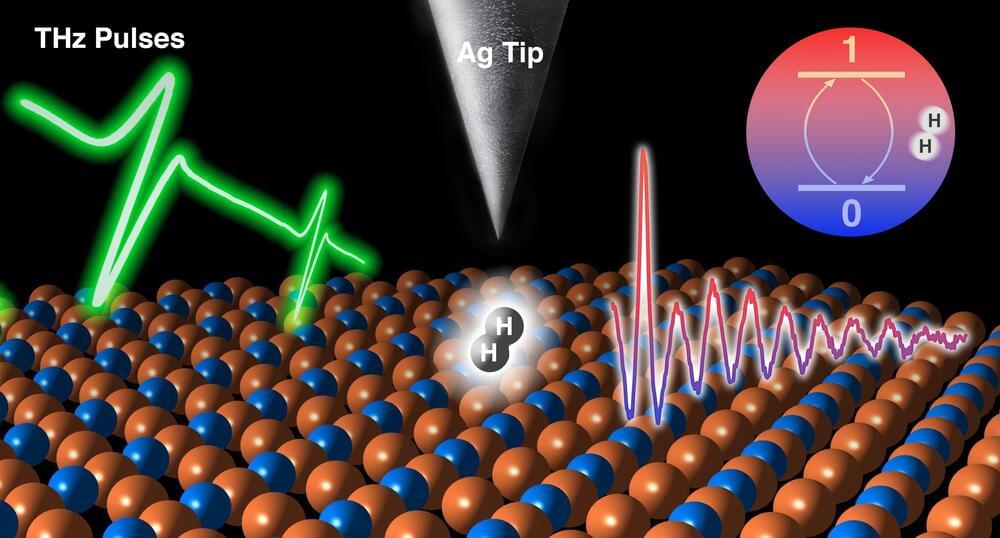
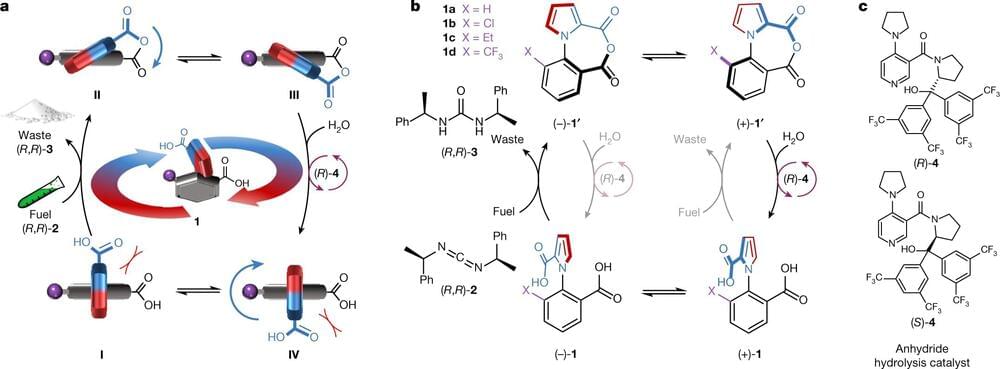
Josh SeehermanI don’t think he’s wrong.
Art ToegemannIt’s adjusting to users sharing a password.
Shubham Ghosh Roy shared a link.
At the interface between chemistry and physics, the process of crystallization is omnipresent in nature and industry. It is the basis for the formation of snowflakes but also of certain active ingredients used in pharmacology. For the phenomenon to occur for a given substance, it must first go through a stage called nucleation, during which the molecules organize themselves and create the optimal conditions for the formation of crystals. While it has been difficult to observe pre-nucleation dynamics, this key process has now been revealed by the work of a research team from the University of Geneva (UNIGE). The scientists have succeeded in visualizing this process spectroscopically in real time and on a micrometric scale, paving the way to the design of safer and more stable active substances. These results can be found in the Proceedings of the National Academy of Sciences (PNAS).
Crystallization is a chemical and physical process used in many fields, from the pharmaceutical industry to food processing. It is used to isolate a gaseous or liquid substance in the form of crystals. However, this phenomenon is not unique to industry; it is ubiquitous in nature and can be seen, for example, in snowflakes, coral or kidney stones.
For crystals to form from substances, they must first go through a crucial stage called nucleation. It is during this first phase that the molecules begin to arrange themselves to form “nuclei,” stable clusters of molecules, which leads to the development and growth of crystals. This process occurs stochastically, meaning it is not predictable when and where a nucleus form. “Until now, scientists have been struggling to visualize this first stage at the molecular level. The microscopic picture of crystal nucleation has been under intense debate. Recent studies suggest that molecules seem to form some disordered organization before the formation of nuclei. Then how does the crystalline order emerge from them? That is a big question,” explains Takuji Adachi, assistant professor in the Department of Physical Chemistry at the UNIGE Faculty of Science.