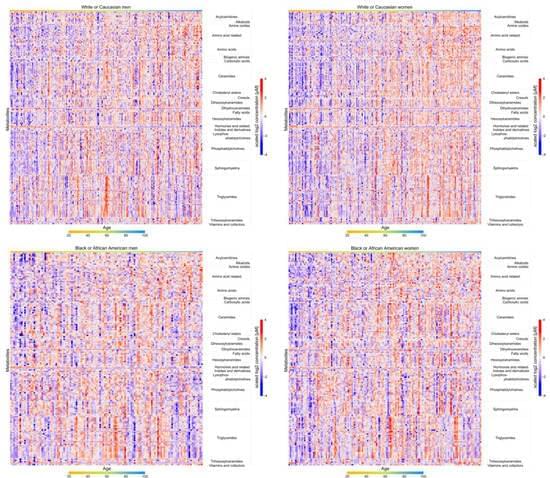
As one of the OMICS in systems biology, metabolomics defines the metabolome and simultaneously quantifies numerous metabolites that are final or intermediate products and effectors of upstream biological processes. Metabolomics provides accurate information that helps determine the physiological steady state and biochemical changes during the aging process. To date, reference values of metabolites across the adult lifespan, especially among ethnicity groups, are lacking. The “normal” reference values according to age, sex, and race allow the characterization of whether an individual or a group deviates metabolically from normal aging, encompass a fundamental element in any study aimed at understanding mechanisms at the interface between aging and diseases.