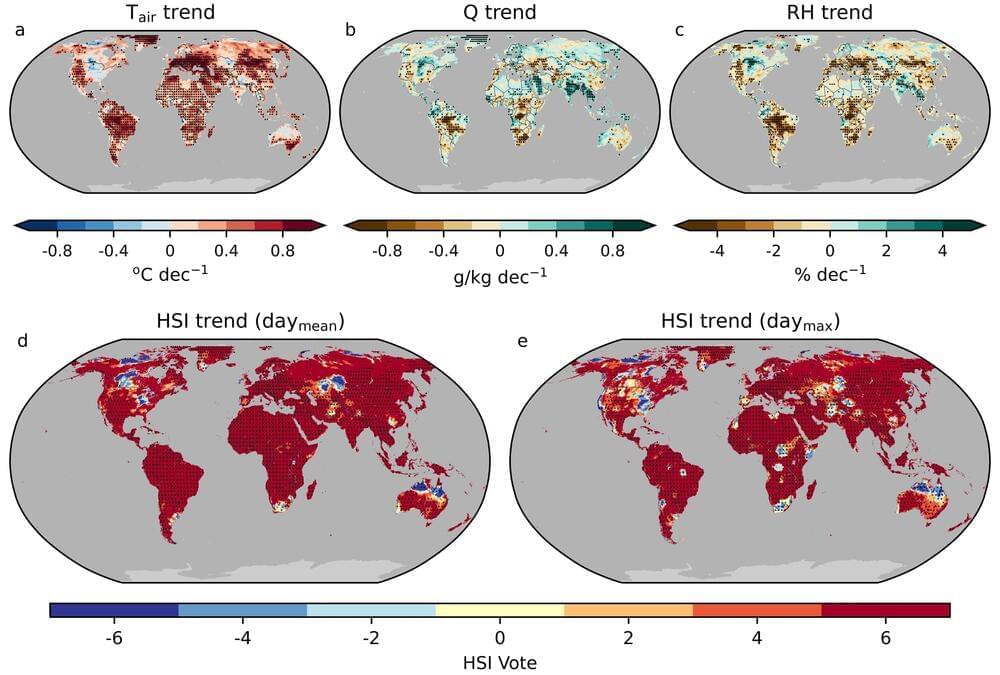
#bioink could be used to #Print and #Grow #Lung #Tissue.
Researchers describe their success in creating a mucus-based bioink for 3D printing lung tissue. This advancement could one day help study and treat chronic lung conditions. scitechupdates.com/mucus-based-bi…
Lung diseases kill millions of people around the world each year. Treatment options are limited, and animal models for studying these illnesses and experimental medications are inadequate. Now, writing in ACS Applied Bio Materials, researchers describe their success in creating a mucus-based bioink for 3D printing lung tissue. This advancement could one day help study and treat chronic lung conditions.
While some people with lung diseases receive transplants, donor organs remain in short supply. As an alternative, medications and other treatments can be used to manage symptoms, but no cure is available for disorders such as chronic obstructive pulmonary disease and cystic fibrosis. Researchers continue to seek better medications, often relying on testing in rodents. But these animal models may only partially capture the complexities of pulmonary diseases in humans, and they might not accurately predict the safety and efficacy of new drugs.
Meanwhile, bioengineers are exploring the production of lung tissue in the lab, either as a more accurate model to study human lungs or as a potential material to use in implants. One technique involves 3D printing structures that mimic human tissue, but designing a suitable bioink to support cell growth remains challenging. So, Ashok Raichur and colleagues set out to overcome this obstacle.