Many AI models fail to recognise negation words such as “no” and “not”, which means they can’t easily distinguish between medical images labelled as showing a disease and images labelled as not showing the disease

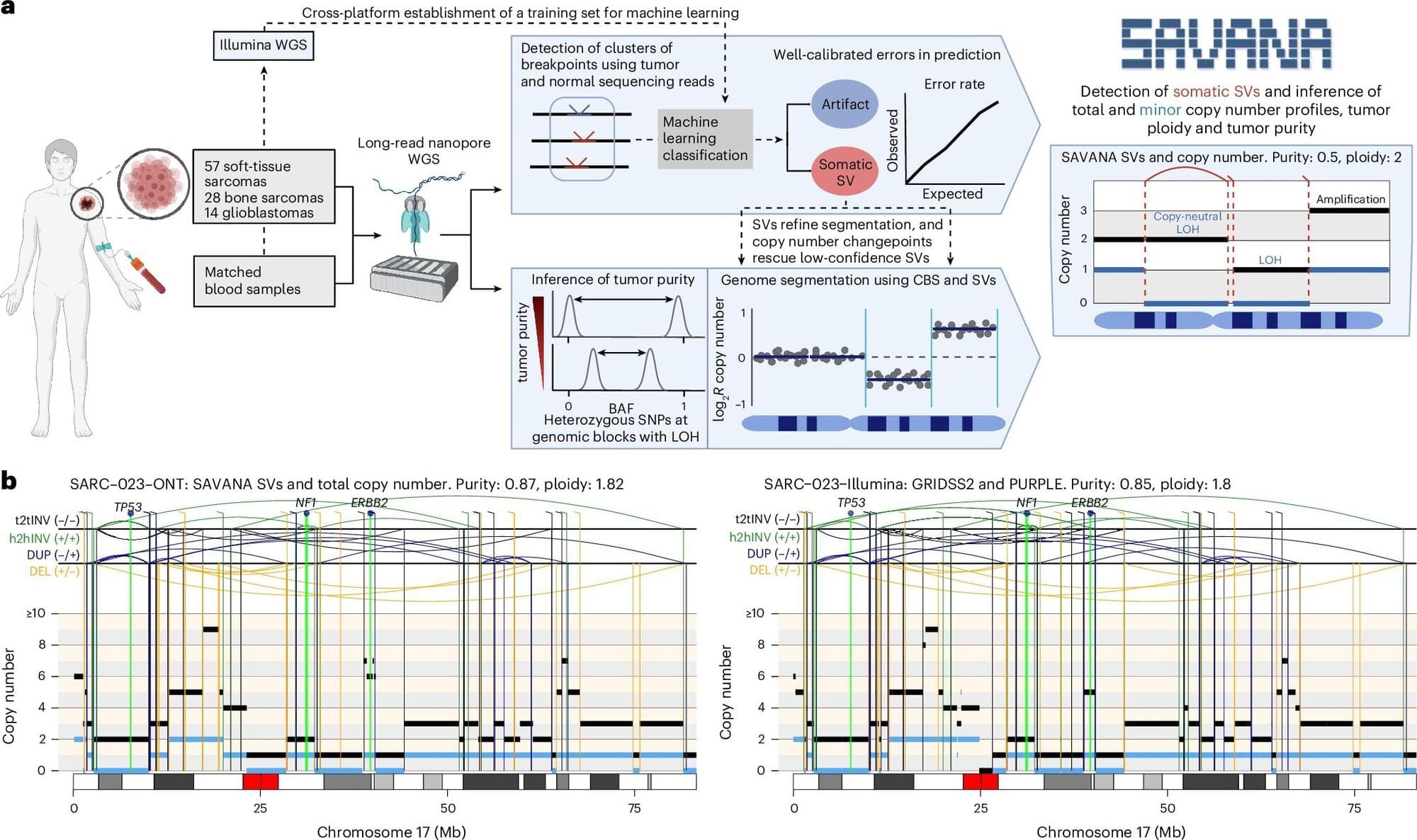
Long-read sequencing technologies analyze long, continuous stretches of DNA. These methods have the potential to improve researchers’ ability to detect complex genetic alterations in cancer genomes. However, the complex structure of cancer genomes means that standard analysis tools, including existing methods specifically developed to analyze long-read sequencing data, often fall short, leading to false-positive results and unreliable interpretations of the data.
These misleading results can compromise our understanding of how tumors evolve, respond to treatment, and ultimately how patients are diagnosed and treated.
To address this challenge, researchers developed SAVANA, a new algorithm which they describe in the journal Nature Methods.
The brain is a unique place. It is shielded from much of the body by the blood-brain barrier, meaning it’s protected from pathogens and potentially dangerous substances that might be in our blood. And historically, many scientists believed that separation extended to the immune system as well: the brain has its own specialized immune cells called microglia, but immune cells present in the rest of the body were long thought to steer clear of the brain unless there was a disease or other problem requiring their presence.
Now, a team of scientists from Yale School of Medicine (YSM) has shown that immune cells known as T cells reside in the healthy brains of mice and humans, trafficked there from the gut and fat. This is the first time T cells have been shown to inhabit the brain under normal, non-diseased conditions.
The findings are published in Nature.
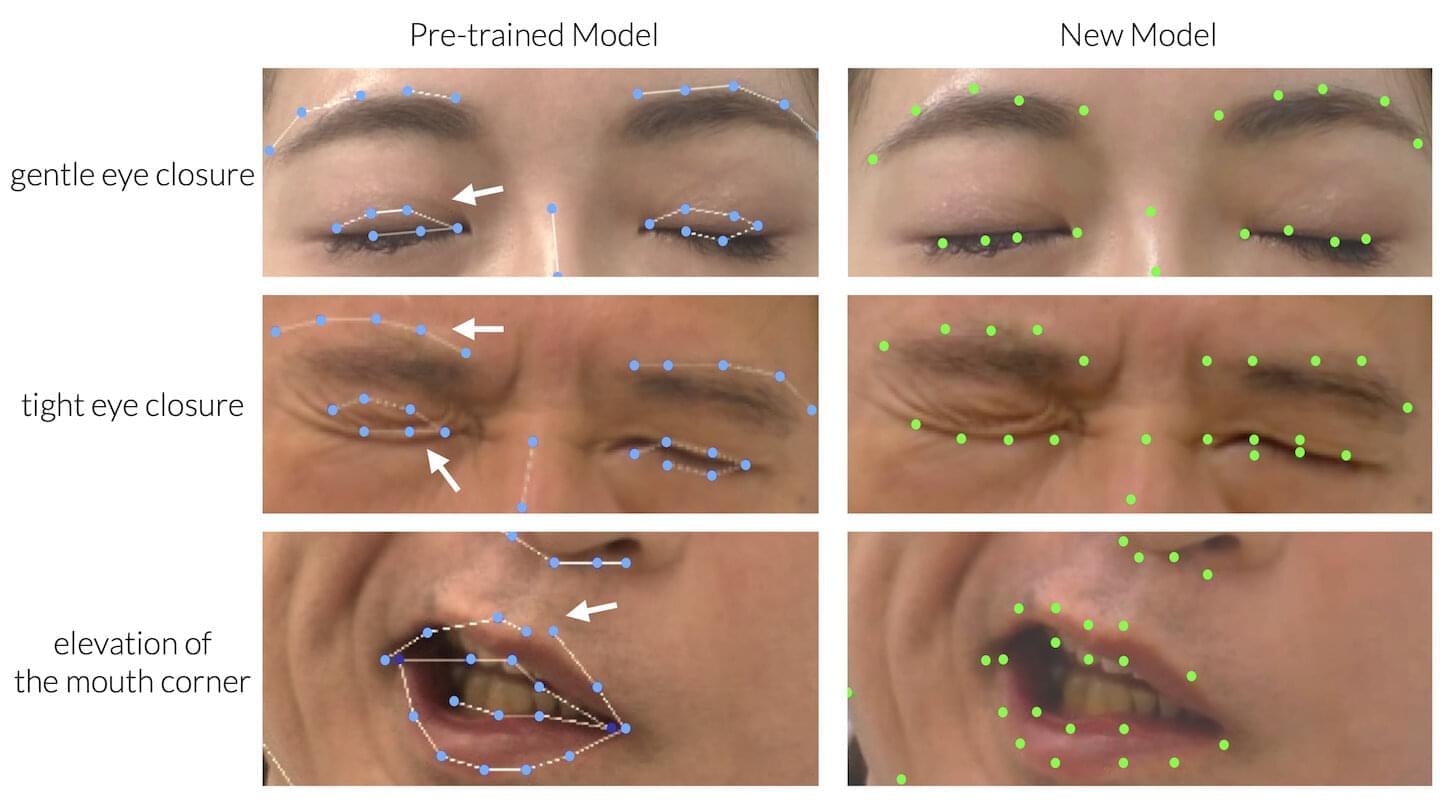
A “fine-tuned” artificial intelligence (AI) tool shows promise for objective evaluation of patients with facial palsy, reports an experimental study in the June issue of Plastic and Reconstructive Surgery.
“We believe that our research offers valuable insights into the realm of facial palsy evaluation and presents a significant advancement in leveraging AI for clinical applications,” comments lead author Takeichiro Kimura, MD, of Kyorin University, Mitaka, Tokyo.
Patients with facial palsy have paralysis or partial loss of movement of the face, caused by nerve injury due to tumors, surgery, trauma, or other causes. Detailed assessment is essential for evaluating treatment options, such as nerve transfer surgery, but poses difficult challenges.

American scientists plan to implement a project to test quantum communication in free space. Using lasers, they want to launch qubits over the Long Island Sound.
It is noted, that three laser beams from the telescope on top of the Kline Tower on the Yale University campus will be directed across the Long Island Sound at a distance of nearly 43.5 km and captured on the opposite side by a similar telescope on the roof of the University Hospital Stony Brook.
The goal of the Quantum Laser Across the Sound project is to expand the ability to send and receive quantum information and demonstrating the potential for possible future quantum computing infrastructures. The telescope on top of the Kline Tower will send entangled photons 43.4 km across the Long Island Sound.

In this episode of The Moss Report, Ben Moss sits down with Dr. Ralph Moss to explore the real-world pros and cons of using artificial intelligence in cancer research and care.
From AI-generated health advice to PubMed citations that don’t exist, this honest conversation covers what AI tools are getting right—and where they can dangerously mislead.
Dr. Moss shares the results of his own AI test across five major platforms, exposing their strengths and surprising failures.
Whether you’re a cancer patient, caregiver, or simply curious about how AI is shaping the future of medicine, this episode is essential listening.
Links and Resources:
🌿 The Moss Method – Fight Cancer Naturally – (Paperback, Hardcover, Kindle) https://amzn.to/4dGvVjp.

Scientists in Europe have tested an anti-aging drug cocktail in mice and found that it extended the animals’ lifespans by around 30 percent. The mice stayed healthier for longer too, with less chronic inflammation and delayed cancer onset.
The two drugs are rapamycin and trametinib, which are both used to treat different types of cancer. Rapamycin is also often used to prevent organ rejection, and has shown promise in extending lifespans in animal tests. Trametinib, meanwhile, has been shown to extend the lifespan of fruit flies, but whether that worked in larger animals remained to be seen.
So for a new study, a research team led by scientists from the Max Planck Institute in Germany investigated how both drugs, on their own and together, could extend lifespan in mice.
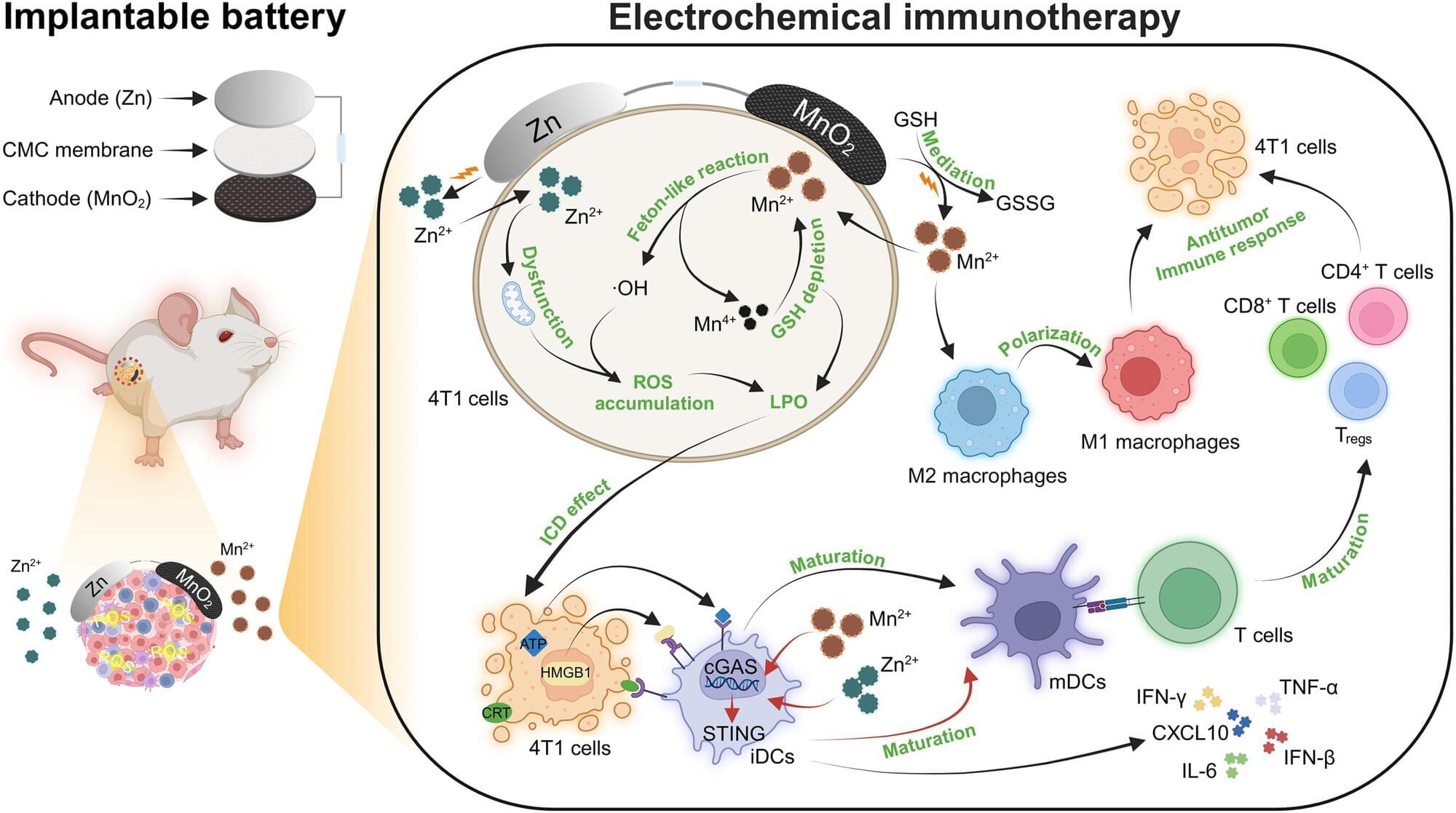
A pioneering biobattery has been shown to reduce tumor growth in the body and could hold the key to a new drug-free immunotherapy treatment in cancer patients.
The breakthrough, a global collaboration between Distinguished Professor Gordon Wallace and Professor Caiyun Wang from the Intelligent Polymer Research Institute (IPRI) at the University of Wollongong (UOW) and researchers from Jilin University in China, is outlined in a new paper published in Science Advances.
Biobatteries have the same basic parts as regular batteries—two electrodes (anode and cathode), a separator and an electrolyte—but use biological processes to create electricity. The paper examines how biobatteries can be used to target tumors and spark a localized immunotherapy response in the body.

Published in Brain, Behavior and Immunity—is the first to suggest that a tumor-driving gene known as AEG-1 actively regulates the inflammation responsible for causing chemotherapy-induced peripheral neuropathy (CIPN), a common and painful side effect of cancer treatment. Eliminating the function of this gene using targeted therapies could become a critical strategy for managing a debilitating side effect experienced by many cancer patients.