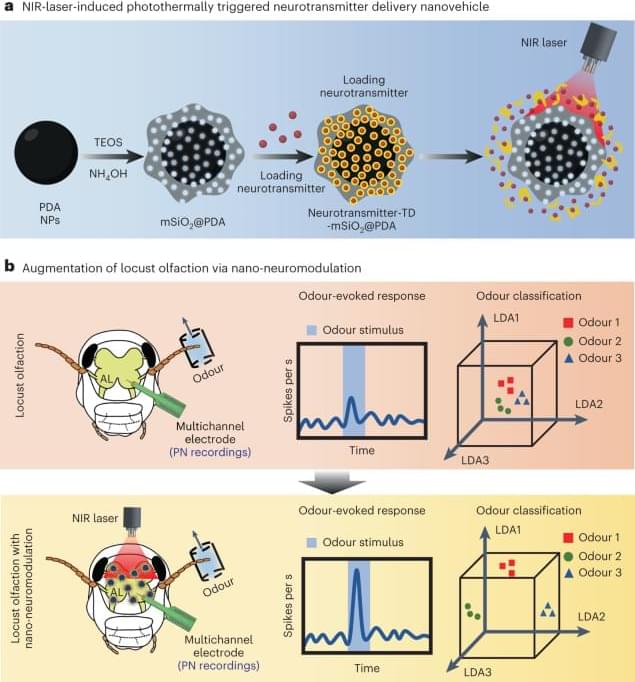
The findings revealed that out of six children subjected to the gene therapy, five exhibited restored hearing and enhanced speech recognition.
Furthermore, the remaining incidents were ephemeral and had no lasting impact, showing that the detected adverse effects were just short and did not raise long-term issues.
“We are the first to initiate the clinical trial of OTOF gene therapy. It is thrilling that our team translated the work from basic research in animal model of DFNB9 to hearing restoration in children with DFNB9,” said Yilai Shu, lead study author from the Eye & ENT Hospital of Fudan University, in a press release.
In another notable event, an 11-year-old boy has become the first to undergo gene therapy in the United States to address congenital deafness.