We’re crawling with microbes, and scientists want to use them to treat disease.



A Learn more on https://scim.ag/5KM
Shots enter early clinical trials for healthy people at high risk for disease.
Multifocal Choroiditis (MFC) patients with inflammatory choroidal neovascularization (iCNV) face challenges in visual and treatment outcomes. A recently published study delves into the effectiveness of Immunomodulatory Therapy (IMT) compared to an approach using steroids as needed, shedding light on visual and treatment outcomes. This study was published in the American Journal of Ophthalmology by Matteo Airaldi and colleagues.

Research conducted by a team of scientists from Kaunas universities, Lithuania, revealed that low-frequency ultrasound influences blood parameters. The findings suggest that ultrasound’s effect on haemoglobin can improve oxygen’s transfer from the lungs to bodily tissues.
The research was undertaken on 300 blood samples collected from 42 pulmonary patients. The samples were exposed to six different low-frequency ultrasound modes at the Institute of Mechatronics of Kaunas University of Technology (KTU).
The changes in 20 blood parameters were registered using the blood analysing equipment at the Lithuanian University of Health Sciences (LSMU) laboratories. For the prediction of ultrasound exposure, artificial intelligence, i.e. analysis of variance (ANOVA), non-parametric Kruskal-Wallis method and machine learning algorithms were applied. The calculations were made at the KTU Artificial Intelligence Centre.

It’s been more than three years since the National Aeronautics and Space Administration made space-grown lettuce an item on the menu for astronauts aboard the International Space Station. Alongside their space diet staples of flour tortillas and powdered coffee, astronauts can munch on a salad, grown from control chambers aboard the ISS that account for the ideal temperature, amount of water and light that plants need to mature.
But there is a problem. The International Space Station has a lot of pathogenic bacteria and fungi. Many of these disease-causing microbes at the ISS are very aggressive and can easily colonize the tissue of lettuce and other plants. Once people eat lettuce that’s been overrun by E. coli or Salmonella, they can get sick.
With billions of dollars poured into space exploration each year by NASA and private companies like SpaceX, some researchers are concerned that a foodborne illness outbreak aboard the International Space Station could derail a mission.

Ball is not alone in calling for a drastic rethink of how scientists discuss biology. There has been a flurry of publications in this vein in the past year, written by me and others2–4. All outline reasons to redefine what genes do. All highlight the physiological processes by which organisms control their genomes. And all argue that agency and purpose are definitive characteristics of life that have been overlooked in conventional, gene-centric views of biology.
This burst of activity represents a frustrated thought that “it is time to become impatient with the old view”, as Ball says. Genetics alone cannot help us to understand and treat many of the diseases that cause the biggest health-care burdens, such as schizophrenia, cardiovascular diseases and cancer. These conditions are physiological at their core, the author points out — despite having genetic components, they are nonetheless caused by cellular processes going awry. Those holistic processes are what we must understand, if we are to find cures.
Ultimately, Ball concludes that “we are at the beginning of a profound rethinking of how life works”. In my view, beginning is the key word here. Scientists must take care not to substitute an old set of dogmas with a new one. It’s time to stop pretending that, give or take a few bits and pieces, we know how life works. Instead, we must let our ideas evolve as more discoveries are made in the coming decades. Sitting in uncertainty, while working to make those discoveries, will be biology’s great task for the twenty-first century.
An MIT biotechnology student has gotten “Doom” to run on a cell array full of gut bacteria, proving that it really does run on everything.
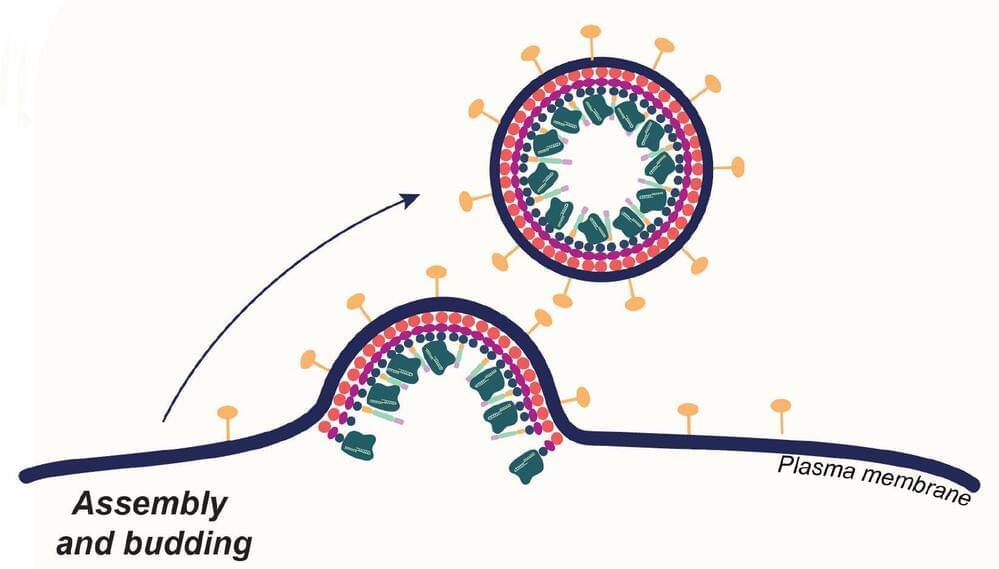
Most approved gene therapies today, including those involving CRISPR-Cas9, work their magic on cells removed from the body, after which the edited cells are returned to the patient.
This technique is ideal for targeting blood cells and is currently the method employed in newly approved CRISPR gene therapies for blood diseases like sickle cell anemia, in which edited blood cells are reinfused in patients after their bone marrow has been destroyed by chemotherapy.
A new, precision-targeted delivery method for CRISPR-Cas9, published in the journal Nature Biotechnology, enables gene editing on very specific subsets of cells while still in the body—a step toward a programmable delivery method that would eliminate the need to obliterate patients’ bone marrow and immune system before giving them edited blood cells.
Basic biology textbooks will tell you that all life on Earth is built from four types of molecules: proteins, carbohydrates, lipids, and nucleic acids. And each group is vital for every living organism.
But what if humans could actually show that these “molecules of life,” such as amino acids and DNA bases, can be formed naturally in the right environment? Researchers at the University of Florida are using the HiPerGator—the fastest supercomputer in U.S. higher education—to test this experiment.
HiPerGator—with its AI models and vast capacity for graphics processing units, or GPUs (specialized processors designed to accelerate graphics renderings)—is transforming the molecular research game.