Biological systems come in all shapes, sizes and structures. Some of these structures, such as those found in DNA, RNA and proteins, are formed through complex molecular interactions that are not easily duplicated by inorganic materials.
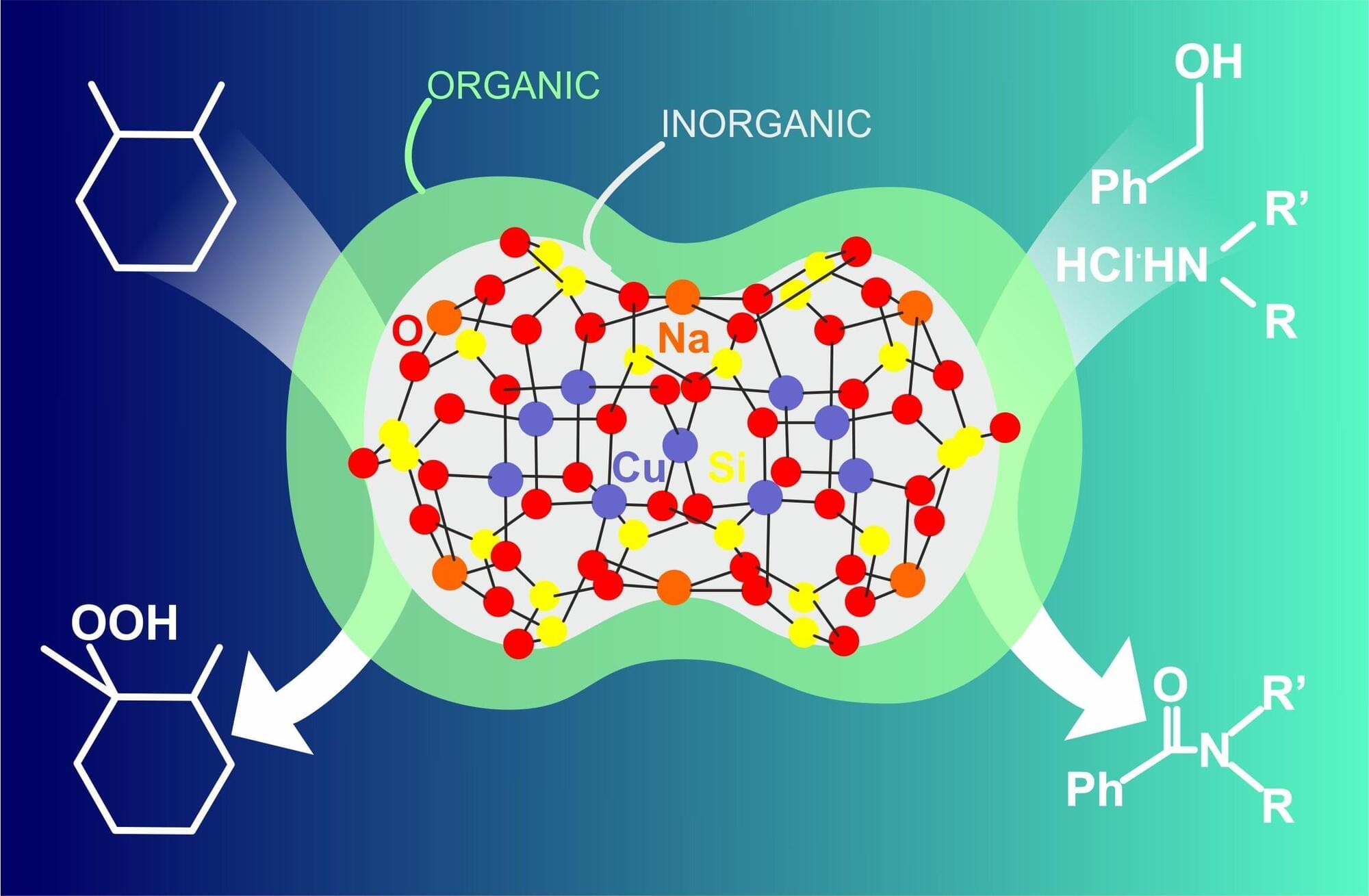
A research team led by Richard Robinson, associate professor of materials science and engineering, discovered a way to bind and stack nanoscale clusters of copper molecules that can self-assemble and mimic these complex biosystem structures at different length scales. The clusters provide a platform for developing new catalytic properties that extend beyond what traditional materials can offer.
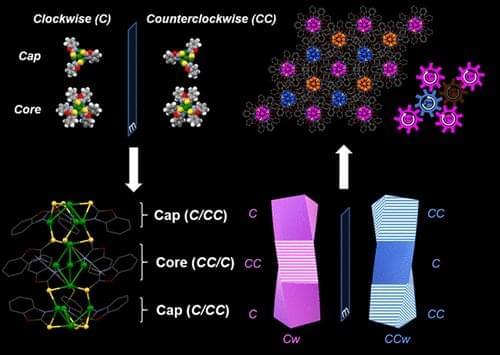
The nanocluster core connects to two copper caps fitted with special binding molecules, known as ligands, that are angled like propeller blades.