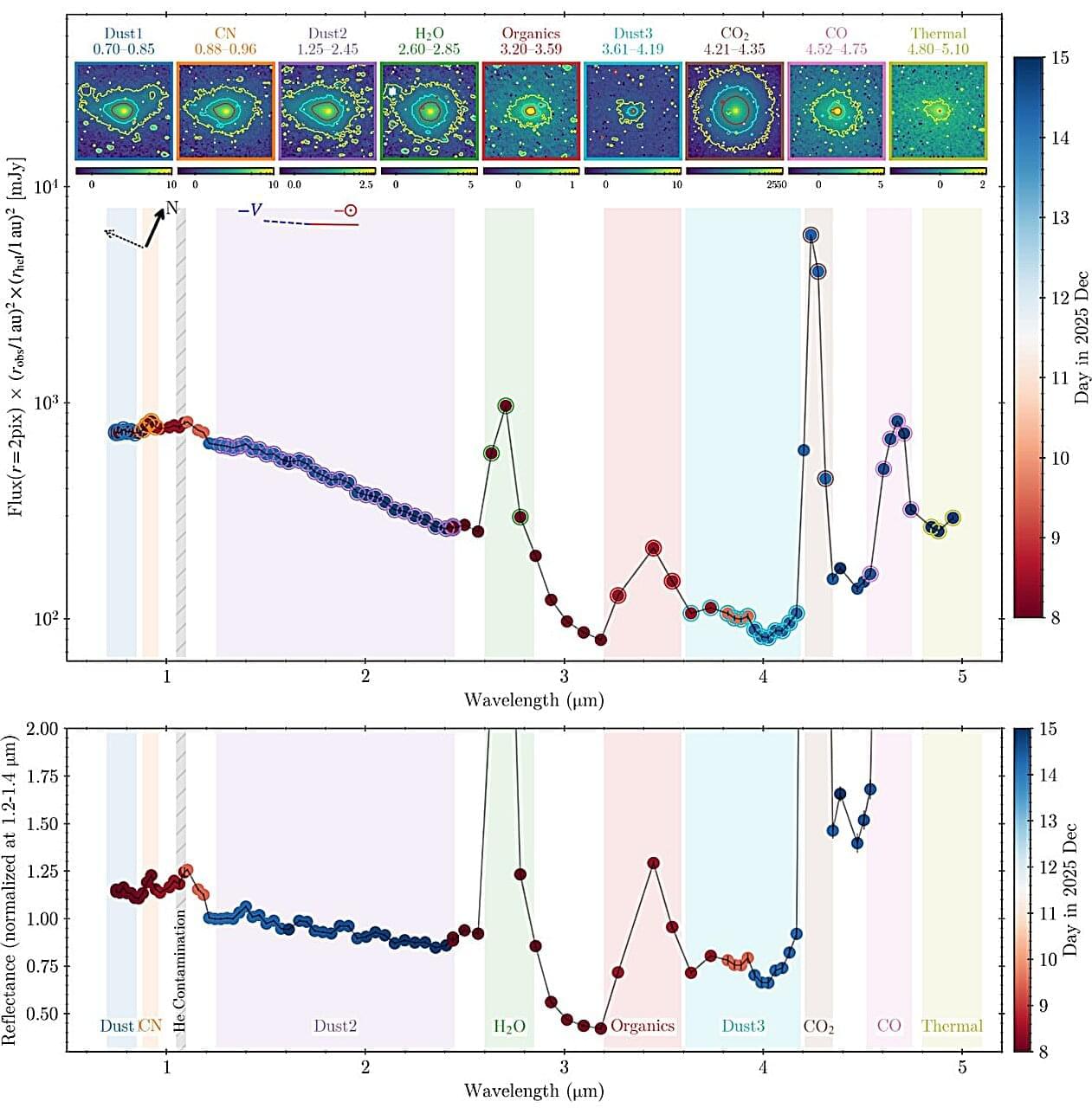
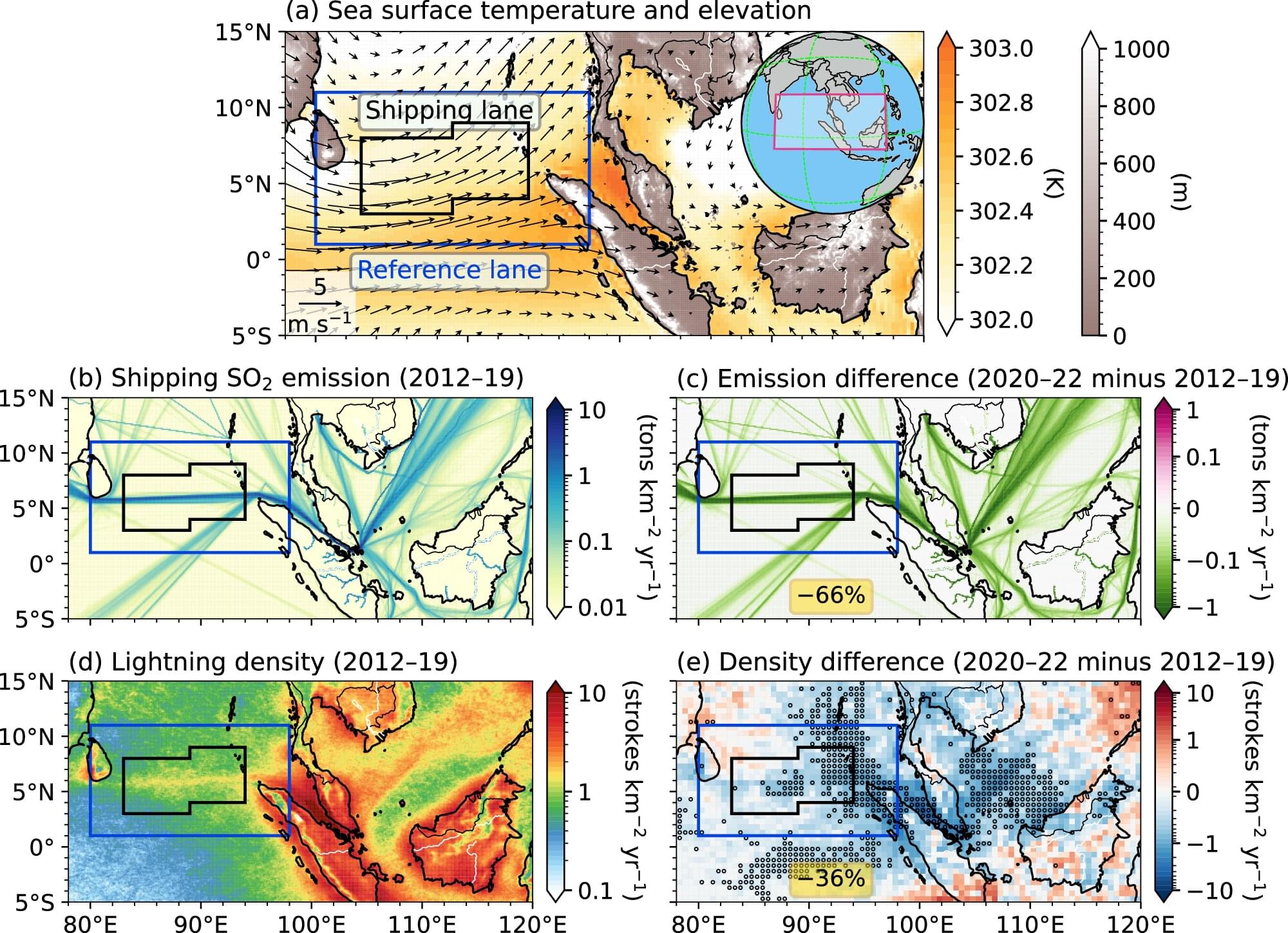
The interstellar object, 3I/ATLAS, was first discovered in July 2025, and made its closest approach to the sun (perihelion) in late October. New observations of 3I/ATLAS were taken in December from the SPHEREx observatory—a near-infrared space observatory used for spectrophotometry. The analysis of these observations was recently discussed by a team of scientists in a paper on arXiv, and reveals some dramatic differences from the data taken before 3I/ATLAS reached perihelion.
SPHEREx first analyzed spectrographic data from 3I/ATLAS in August, shortly after its discovery. At the time, the interstellar object was moving inward, getting closer to the sun, but still between Jupiter and Mars.
At this time, spectrographic analysis showed “barely detectable” H2O-gas, according to the study authors, along with a CO2 coma. The CO2 gas production was estimated in a prior study to be about 9.4 × 1026 molecules/sec, with upper limits for H2O and CO being much lower. Carbon-based organic compounds (collectively referred to as C-H), like methanol (CH3OH), formaldehyde (H2CO), methane (CH4), and ethane (C2H6) were not detectable at the time.