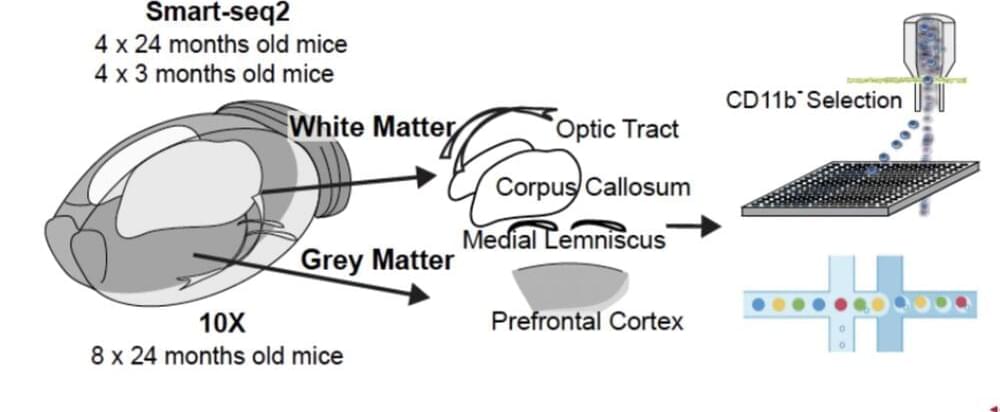
At first glance, the human body seems to be symmetrical: two arms, two legs, two eyes, two ears, and even the nose and mouth appear to be mirrored on an imaginary axis that divides most people’s faces. Finally, the brain is split into two nearly equal-sized halves, and the furrows and bulges follow a similar pattern. The initial impression, however, is misleading since there are small, functionally relevant differences between the left and right sides of the different brain regions.
The two hemispheres have distinct functional specializations. For instance, most individuals process language mostly in their left hemisphere whereas spatial attention is primarily processed in their right hemisphere. Work can thus be distributed more effectively to both sides, and the overall range of tasks is expanded.
However, this so-called lateralization, or the tendency for brain regions to process certain functions more in the left or right hemisphere, differs between people. And not only in the minority whose brains are mirror-inverted in comparison to the majority. Even people with classically arranged brains have varying degrees of asymmetry. Previous research has indicated that this, in turn, may have an effect on the functions themselves.