A new study reveals how humans use flexible problem-solving strategies like hierarchical and counterfactual reasoning when faced with complex tasks.


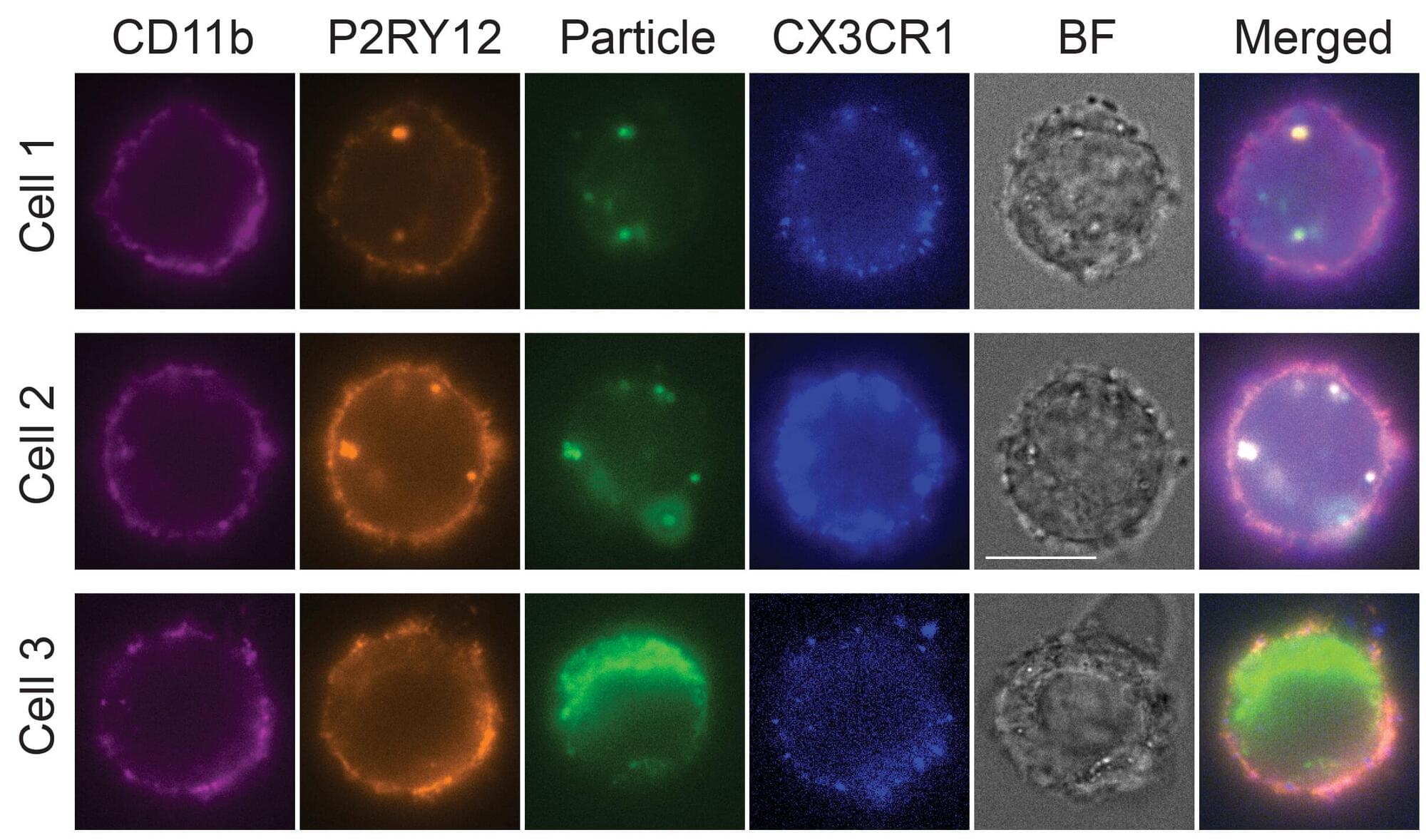
Microglia are a specialized type of immune cell that accounts for about 10% of all cells within the brain and spinal cord. They function by eliminating infectious microbes, dead cells, and aggregated proteins, as well as soluble antigens that may endanger the brain and, during development, also help shape neural circuits enabling specific brain functions.
When microglia don’t function properly, they can trigger neuroinflammation and fail to clear away damaged cells and harmful protein clumps—such as the neurofibrillary tangles and amyloid plaques seen in Alzheimer’s disease. This contributes to numerous neurodegenerative diseases, including Alzheimer’s, Parkinson’s and Huntington’s disease, as well as amyotrophic lateral sclerosis (ALS), multiple sclerosis, and other disorders. In fact, neuroinflammation can occur even before proteins start to form pathogenic aggregates and, in turn, accelerates protein aggregation.
Researchers and drug developers aiming to better understand and target microglia functions in the brain are challenged by the fact that human microglia can only be obtained through biopsies, and rodents’ microglia differ from their human counterparts in many critical features. This supply issue prompted them to work on methods to create microglia in the culture dish using stem cells as a starting point. However, to date, this process has remained inefficient, and requires weeks to complete at significant costs.

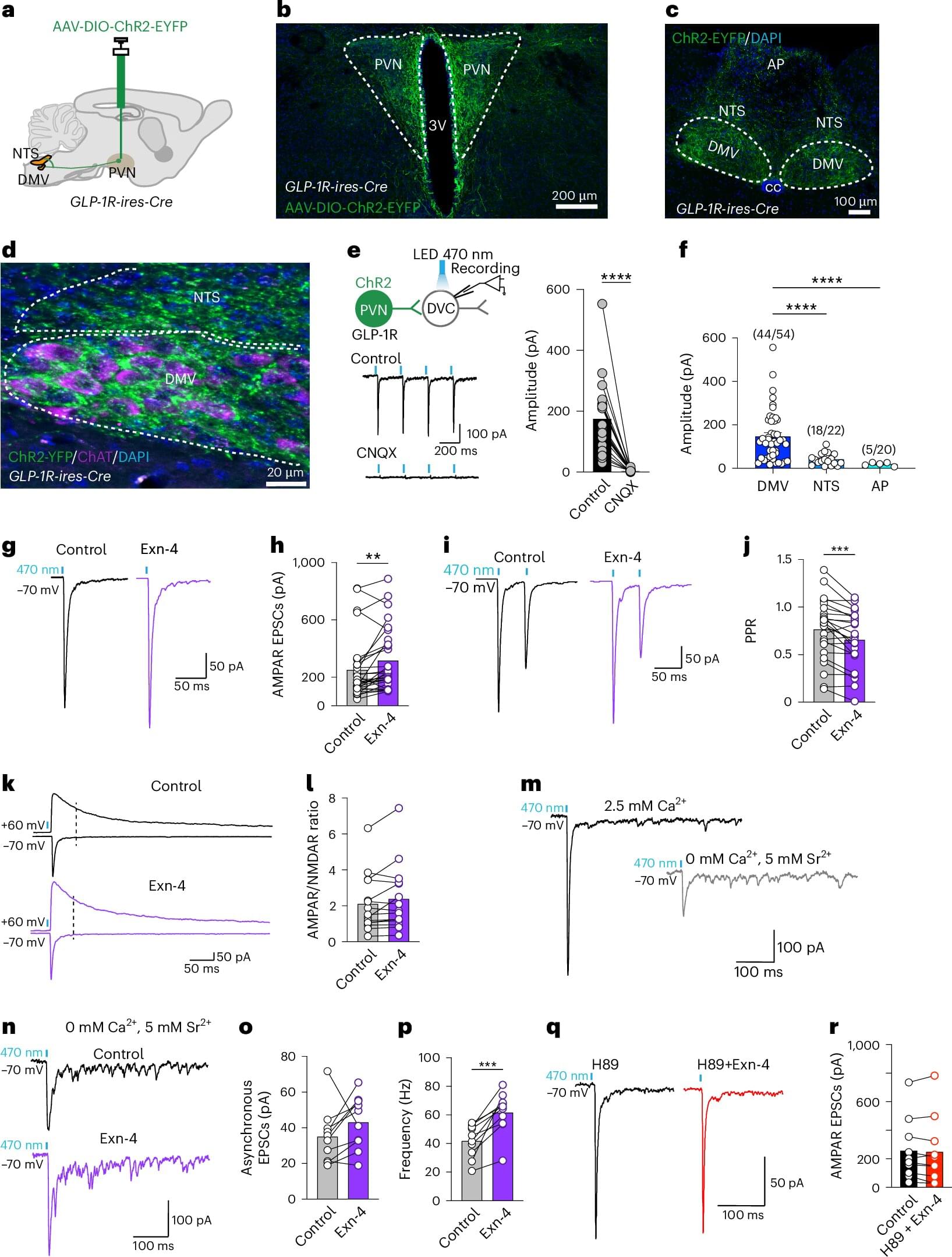
Scientists know the stomach talks to the brain, but two new studies from Rutgers Health researchers suggest the conversation is really a tug-of-war, with one side urging another bite, the other signaling “enough.”
Together, the papers in Nature Metabolism and Nature Communications trace the first complementary wiring diagram of hunger and satiety in ways that could refine today’s blockbuster weight-loss drugs and blunt their side effects.
One study, led by Zhiping Pang of Robert Wood Johnson Medical School’s Center for NeuroMetabolism, pinpointed a slender bundle of neurons that runs from the hypothalamus to the brainstem.

Our brain is adept at synchronizing with rhythmic sounds, whether it’s the beat of a song or the steady patter of rain. This ability helps us recognize and process sounds more effectively.
A research team led by the Max Planck Institute for Empirical Aesthetics (MPIEA) in Frankfurt am Main has shown that stimulation with weak electrical currents, known as transcranial alternating current stimulation (tACS), can influence this ability. The new study is published in the journal PLOS Biology.
The study builds on previous work showing that tACS can either enhance or suppress brain rhythms depending on how it’s timed with incoming sound. In order to investigate the interaction between electrical stimulation and brain rhythms, 50 participants took part in three experiments where they listened to noisy sounds and were asked to identify short, barely perceptible pauses. The researchers then transmitted electrical rhythms to the participants’ brains via electrodes placed on their scalps several times to see how this influenced their brain activity.
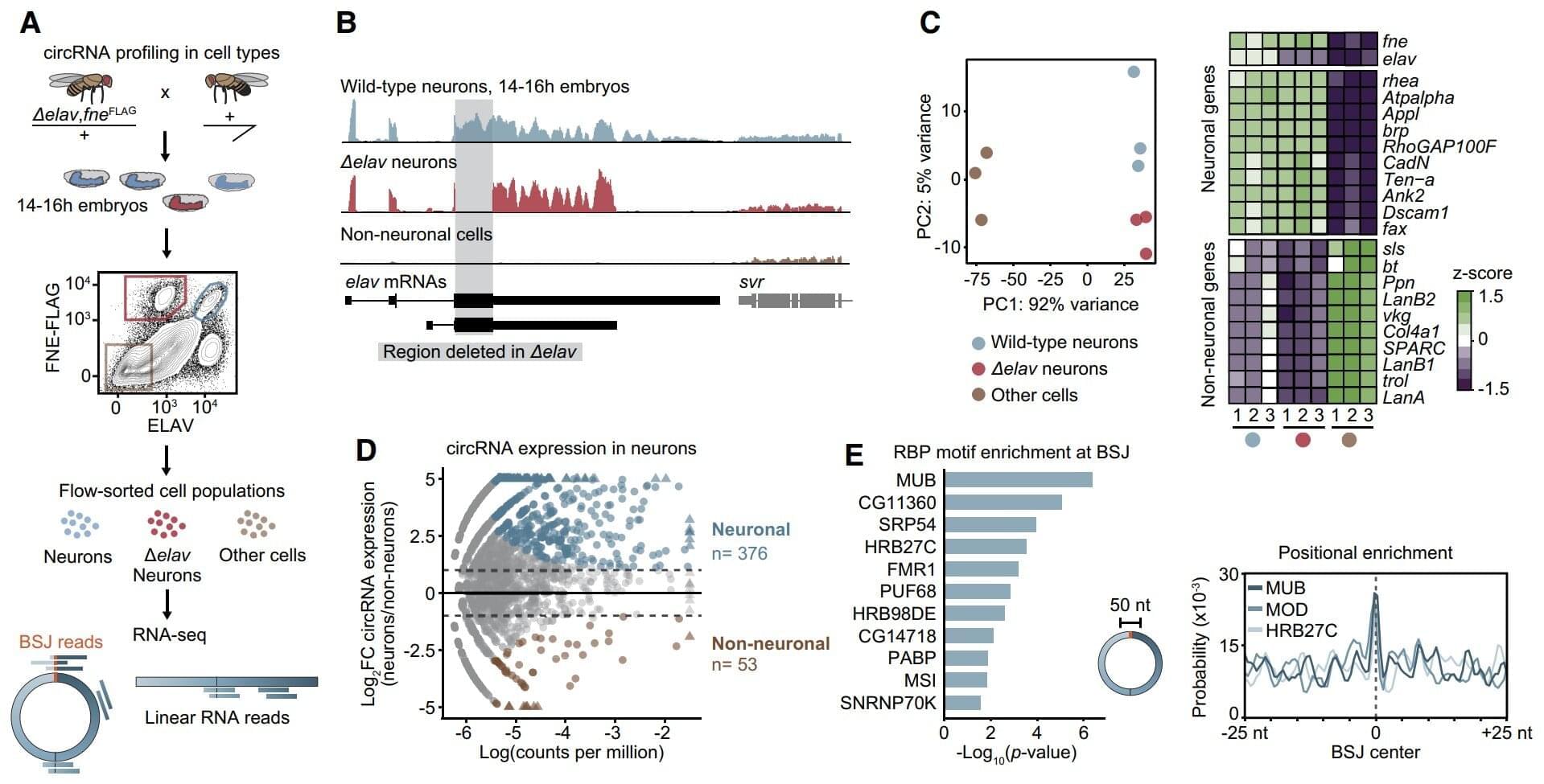
Deep within our nerve cells, a molecule is at work that has no beginning and no end. Instead of a straight chain, as is common for most RNA strands, it forms a closed loop. Known as circular RNAs (circRNAs), these molecules are crucial for development, thought, and synaptic function, yet their high prevalence in neurons has long been a scientific mystery. How does the brain produce so many of them?
Now, Max Planck researchers from Freiburg have discovered a crucial mechanism that explains the remarkable abundance of circRNAs in the nervous system. Published in Genes & Development, the study reveals that the protein ELAV acts as a global master switch for the production of these molecules.
CircRNAs are found across all life forms. They are expressed in specific patterns during different developmental stages and in different cells—especially abundant in cells of the nervous system. While their roles are not as well-studied as normal, linear RNAs, circRNAs are known to be important in brain development, cognition, and even in conditions like neurodegeneration and addiction.
Among the billions of neurons in the brain, fewer than 500 are responsible for suppressing binge drinking, according to new research by Gilles E. Martin, Ph.D., associate professor of neurobiology.
Published in Nature Neuroscience, these findings provide insights into binge-drinking behavior and alcohol dependency that may lead to new therapeutic targets.
“It’s really hard to comprehend how only a few neurons can have such a profound effect on behavior,” said Dr. Martin, a member of the Brudnick Neuropsychiatric Research Institute at UMass Chan. “This is exciting because we are starting to understand how only a handful of cells are involved in very specific behaviors. Truly, this study is about finding a needle in a haystack.”
Some autistic traits related to challenges with social interaction, mental flexibility and visual perception could be alleviated through a new, noninvasive therapy. A team of researchers, including those from the University of Tokyo, found that stimulating nerve cells when the brain becomes “stuck” in a certain state improves flexibility and relieves some autistic behaviors. The procedure utilized transcranial magnetic stimulation (TMS), which is already used to treat certain mood disorders, in a novel manner.
The study is published in the journal Nature Neuroscience.
Over 40 adults with a mild form of autism participated, and the therapeutic effects lasted for up to two months after the last session. This study could contribute toward projects enabling new treatments.

Using an algorithm they call the Krakencoder, researchers at Weill Cornell Medicine are a step closer to unraveling how the brain’s wiring supports the way we think and act. The study, published June 5 in Nature Methods, used imaging data from the Human Connectome Project to align neural activity with its underlying circuitry.
Mapping how the brain’s anatomical connections and activity patterns relate to behavior is crucial not only for understanding how the brain works generally but also for identifying biomarkers of disease, predicting outcomes in neurological disorders and designing personalized interventions.
The brain consists of a complex network of interconnected neurons whose collective activity drives our behavior. The structural connectome represents the physical wiring of the brain, the map of how different regions are anatomically connected.
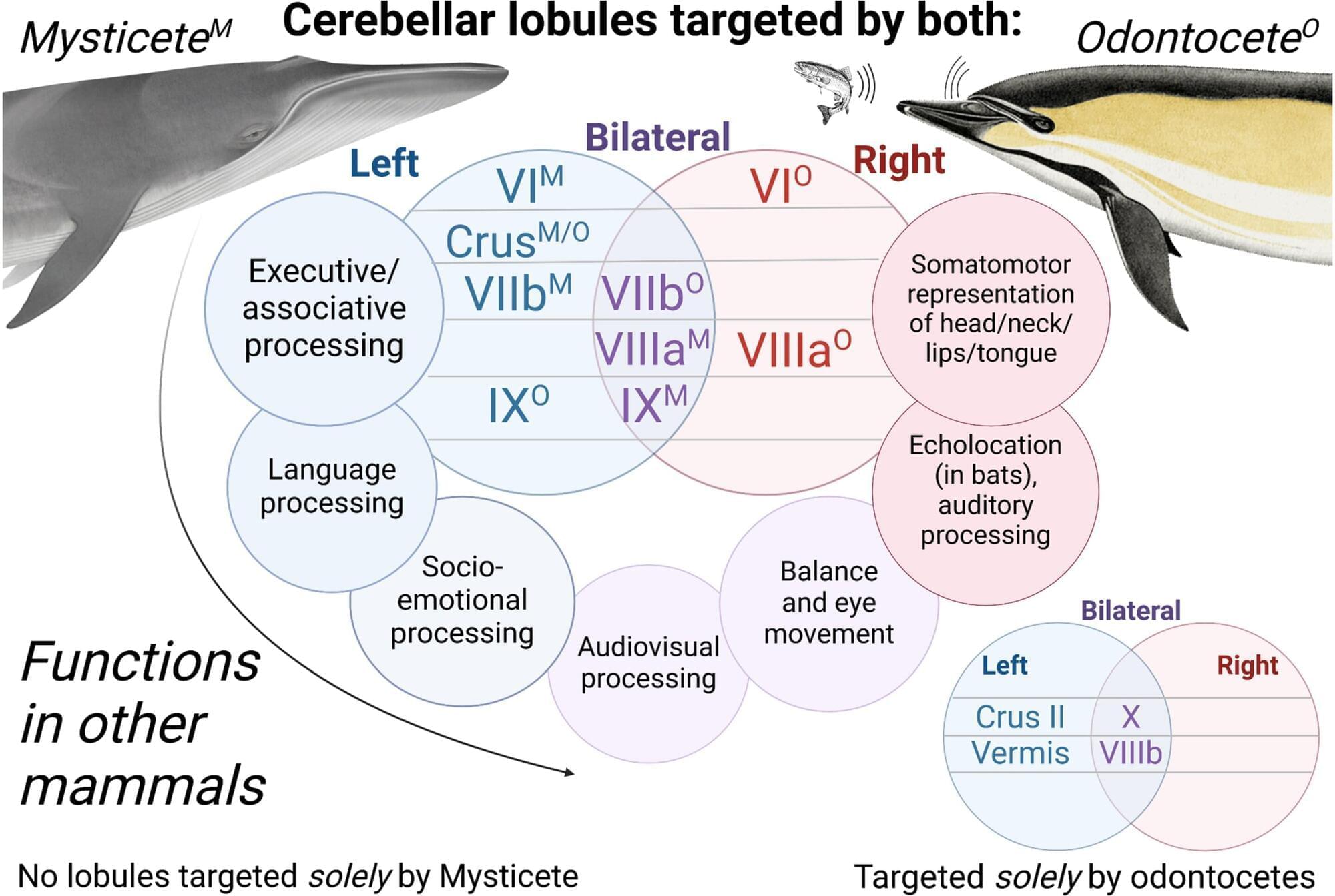
Toothed whales use sound to find their way around, detect objects, and catch fish. They can investigate their environment by making clicking sounds, and then decoding the “echoic return signal” created when the clicking sounds bounce off objects and return to their ears. This “biosonar,” called echolocation, is rare in the animal kingdom.
Now, a new study by researchers at the Woods Hole Oceanographic Institution, New College of Florida, UC Berkeley, and Oxford University, and published in PLOS One, brings us closer to understanding how dolphin brains have evolved to support echolocation.
The research team applied new techniques for mapping networks in the excised brains of dead, stranded cetaceans to examine and compare the auditory pathways in echolocating dolphins and a non-echolocating baleen whale called a sei whale. A partnership with the International Fund for Animal Welfare (IFAW) and others is critical to advancing this work.