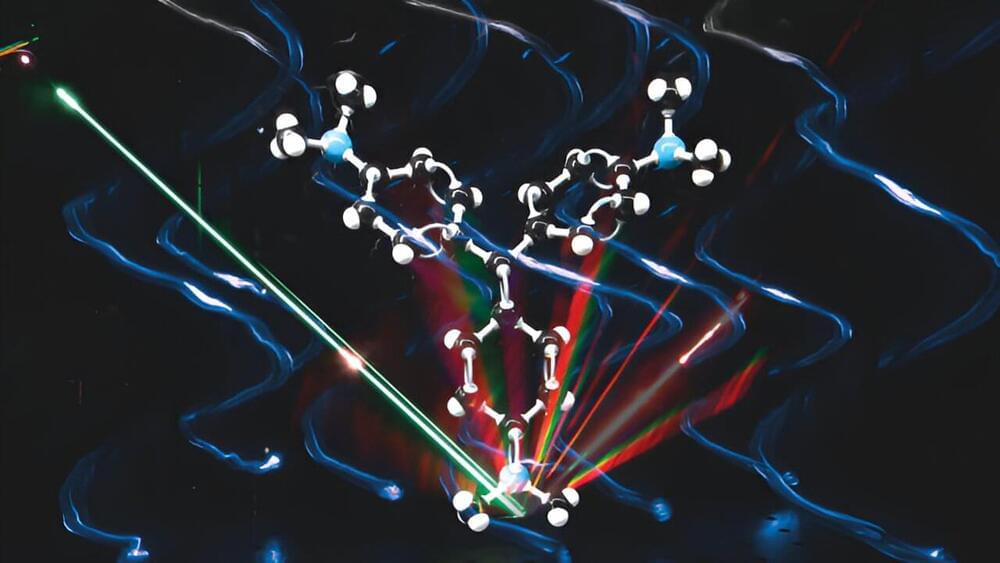
Thinking of X-rays might trigger memories of broken bones or dental check-ups. But this extremely energetic light can show us more than just our bones: it is also used to study the molecular world, even biochemical reactions in real-time. One issue, though, is that researchers have never been able to study a single atom with X-rays. Until now.
Scientists have been able to characterize a single atom using X-rays. Not only were they able to distinguish the type of atoms they were seeing (there were two different ones), but they also managed to study the chemical behavior these atoms were showing.
“Atoms can be routinely imaged with scanning probe microscopes, but without X-rays, one cannot tell what they are made of. We can now detect exactly the type of a particular atom, one atom-at-a-time, and can simultaneously measure its chemical state,” senior author Professor Saw Wai Hla, from the University of Ohio and the Argonne National Laboratory, said in a statement.