This study explores how muscle contractions, such as those that occur during exercise, influence motor neurons—the cells responsible for controlling muscle movement.
There’s no doubt that exercise does a body good. Regular activity not only strengthens muscles but can bolster our bones, blood vessels, and immune system.
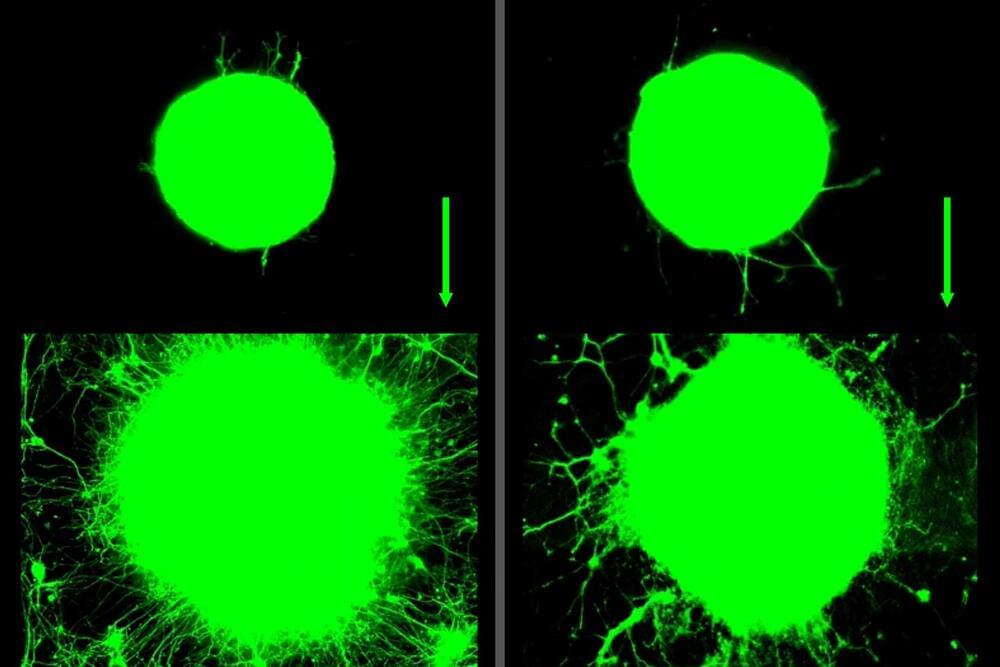
Now, MIT engineers have found that exercise can also have benefits at the level of individual neurons. They observed that when muscles contract during exercise, they release a soup of biochemical signals called myokines.
In the presence of these muscle-generated signals, neurons grew four times further compared to neurons that were not exposed to myokines. These cellular-level experiments suggest that exercise can have a significant biochemical effect on nerve growth.