Histone proteins provide essential structural support for DNA in chromosomes, acting as spools around which DNA strands wrap. These proteins have been well studied, but most current tools to study gene expression rely on RNA sequencing. Histone RNA is unique in that its structure prevents the RNA molecules from being detected by current methods.
Thus, the expression of histone genes may be significantly underestimated in tumor samples. The researchers hypothesized that the increased proliferation of cancer cells leads to a very elevated expression, or hypertranscription, of histones to meet the added demands of cell replication and division.
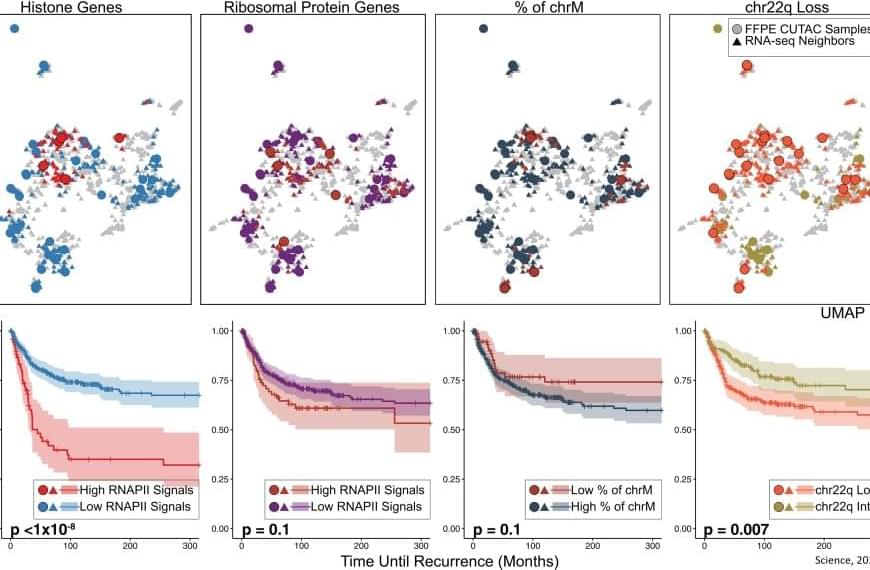
To test their hypothesis, the researchers used CUTAC profiling to examine and map RNAPII, which transcribes DNA into precursors of messenger RNA. They studied 36 FFPE samples from patients with meningioma – a common and benign brain tumor – and used a novel computational approach to integrate this data with nearly 1,300 publicly available clinical data samples and corresponding clinical outcomes.
In tumor samples, the RNAPII enzyme signals found on histone genes were reliably able to distinguish between cancer and normal samples.
RNAPII signals on histone genes also correlated with clinical grades in meningiomas, accurately predicting rapid recurrence as well as the tendency of whole-arm chromosome losses. Using this technology on breast tumor FFPE samples from 13 patients with invasive breast cancer also predicted cancer aggressiveness.
Using a new technology and computational method, researchers have uncovered a biomarker capable of accurately predicting outcomes in meningioma brain tumors and breast cancers.