The AI-focused biotech insitro has laid off 22% of its workforce, the company said in a Thursday statement.


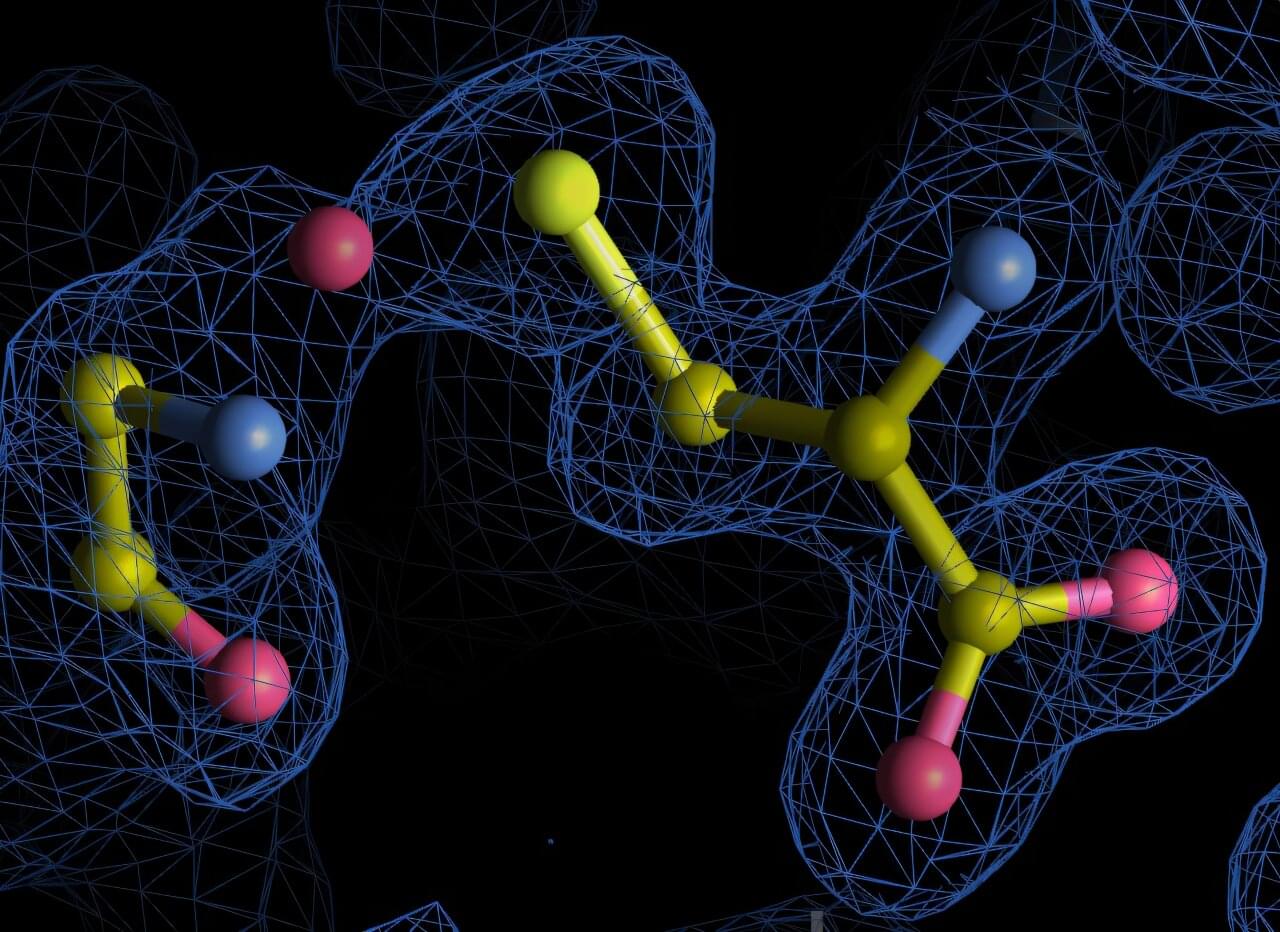
Proteins are among the most studied molecules in biology, yet new research from the University of Göttingen shows they can still hold surprising secrets. Researchers have discovered previously undetected chemical bonds within archived protein structures, revealing an unexpected complexity in protein chemistry.
These newly identified nitrogen-oxygen-sulfur (NOS) linkages broaden our understanding of how proteins respond to oxidative stress, a condition where harmful oxygen-based molecules build up and can damage proteins, DNA, and other essential parts of the cell. The new findings are published in Communications Chemistry.
The research team systematically re-analyzed over 86,000 high-resolution protein structures from the Protein Data Bank, a global public repository of protein structures, using a new algorithm that they developed inhouse called SimplifiedBondfinder. This pipeline combines machine learning, quantum mechanical modeling, and structural refinement methods to reveal subtle chemical bonds that were missed by conventional analyses.

Swabs from China’s Tiangong space station reveal traces of a bacterium unseen on Earth, with characteristics that may help it function under stressful environmental conditions hundreds of kilometers above the planet’s surface.
Naming their discovery after the station, researchers from the Shenzhou Space Biotechnology Group and the Beijing Institute of Spacecraft System Engineering say the study of Niallia tiangongensis and similar species could be “essential” in protecting astronaut health and spacecraft functionality over long missions.
The swabs were taken from a cabin on board the space station in May 2023 by the Shenzhou-15 crew as part of one of two surveys by the China Space Station Habitation Area Microbiome Programme.

Vision is one of the most crucial human senses, yet over 300 million people worldwide are at risk of vision loss due to various retinal diseases. While recent advancements in retinal disease treatments have successfully slowed disease progression, no effective therapy has been developed to restore already lost vision-until now. KAIST researchers have successfully developed a novel drug to restore vision.
KAIST (represented by President Kwang Hyung Lee) announced on the 30th of March that a research team led by Professor Jin Woo Kim from the Department of Biological Sciences has developed a treatment method that restores vision through retinal nerve regeneration.
The research team successfully induced retinal regeneration and vision recovery in a disease-model mouse by administering a compound that blocks the PROX1 (prospero homeobox 1) protein, which suppresses retinal regeneration. Furthermore, the effect lasted for more than six months.
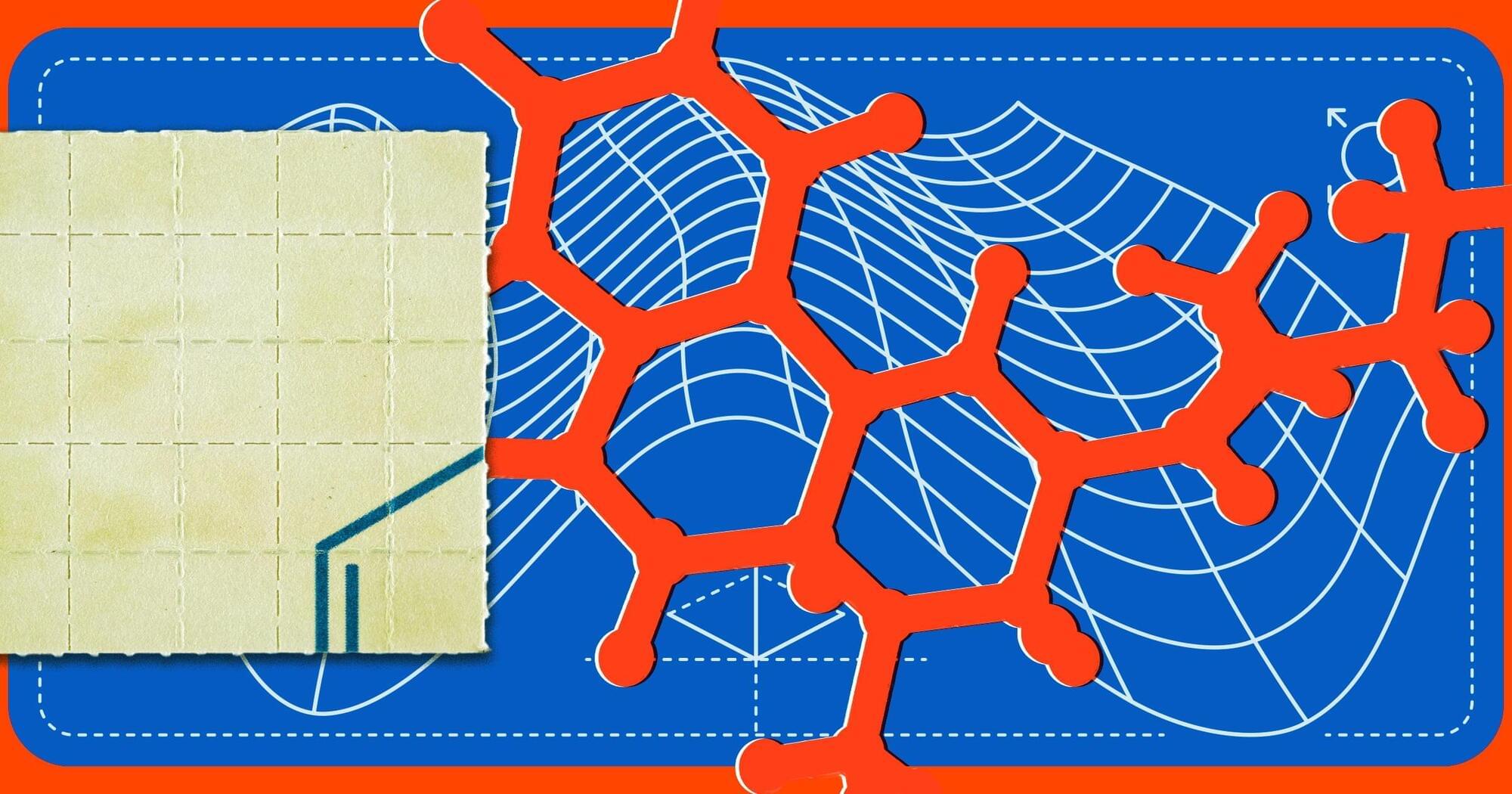
A team of researchers at the University of California, Davis, made small tweaks to the molecular structure of lysergic acid diethylamide (LSD) to see if it could be turned into an effective brain-healing treatment for patients that suffer from conditions like schizophrenia — without risking a potentially disastrous acid trip.
As detailed in a new paper published in the journal Proceedings of the National Academy of Sciences last month, the researchers created a new compound called JRT by shifting the position of just two atoms of the psychedelic’s molecular structure.
With the two atoms flipped, the new drug could still stimulate brain cell growth and repair damaged neural connections, while simultaneously minimizing psychedelic effects, in mice.
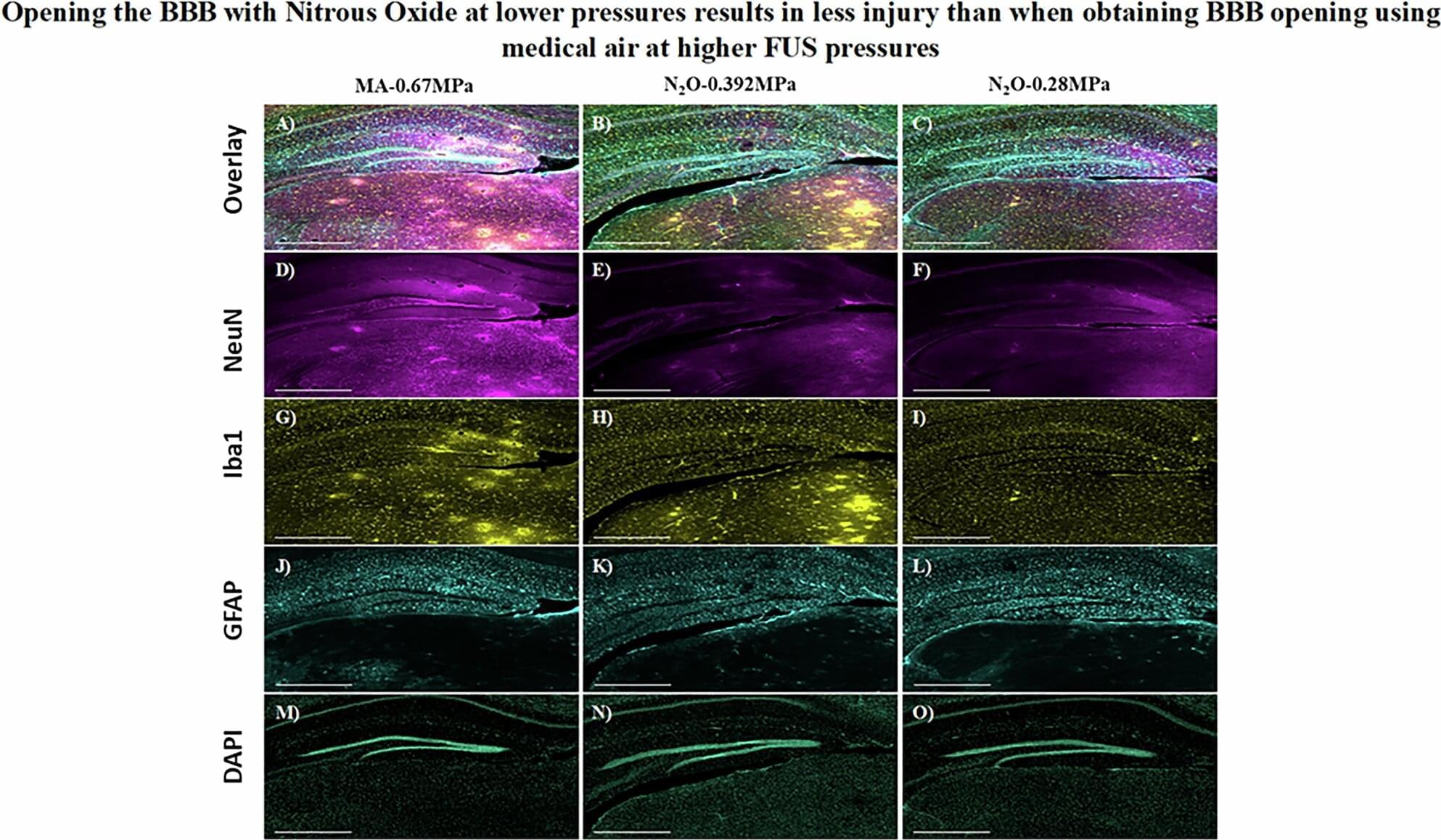
Nitrous oxide, a commonly used analgesic gas, temporarily improved the opening of the blood-brain barrier (BBB) to allow gene therapy delivery in mouse models using focused ultrasound (FUS), UT Southwestern Medical Center researchers report in a new study. Their findings, published in Gene Therapy, could eventually lead to new ways to treat a variety of brain diseases and disorders.
“The approach we explored in this study has the potential to advance care for diseases of the brain that can be treated by targeted therapeutic delivery,” said study leader Bhavya R. Shah, M.D., Associate Professor of Radiology, Neurological Surgery, and in the Advanced Imaging Research Center at UT Southwestern. He’s also an Investigator in the Peter O’Donnell Jr. Brain Institute and a member of the Center for Alzheimer’s and Neurodegenerative Diseases. Deepshikha Bhardwaj, Ph.D., Senior Research Associate at UTSW, was the study’s first author.
The BBB is a highly selective border of semipermeable cells that line tiny blood vessels supplying blood to the brain. It is thought to have developed during evolution to protect the brain from toxins and infections in the blood. However, the BBB also impedes the delivery of drugs that could be used to treat neurologic or neuropsychiatric conditions, such as Alzheimer’s disease, multiple sclerosis, or brain tumors. Consequently, researchers have worked for decades to develop solutions that can temporarily open the BBB to allow treatments to enter.

Researchers at the University of Alabama in the United States have used duct tape from a store to create a triboelectric nanogenerator capable of collect electricity from the human body and the environment.
Their development is capable of powering small devices such as biosensors by converting mechanical energy from friction and movement into electricity. The generator is made of metallized films polyethylene terephthalate, which act as electrodes, and layers of adhesive tape.
The developers emphasize that power generation occurs through interaction polypropylene and of the acrylic adhesive layer when they are pressed and released. At the same time, due to the weak intermolecular interaction (Van der Waals forces) on the borders of the atomic-sized gaps.
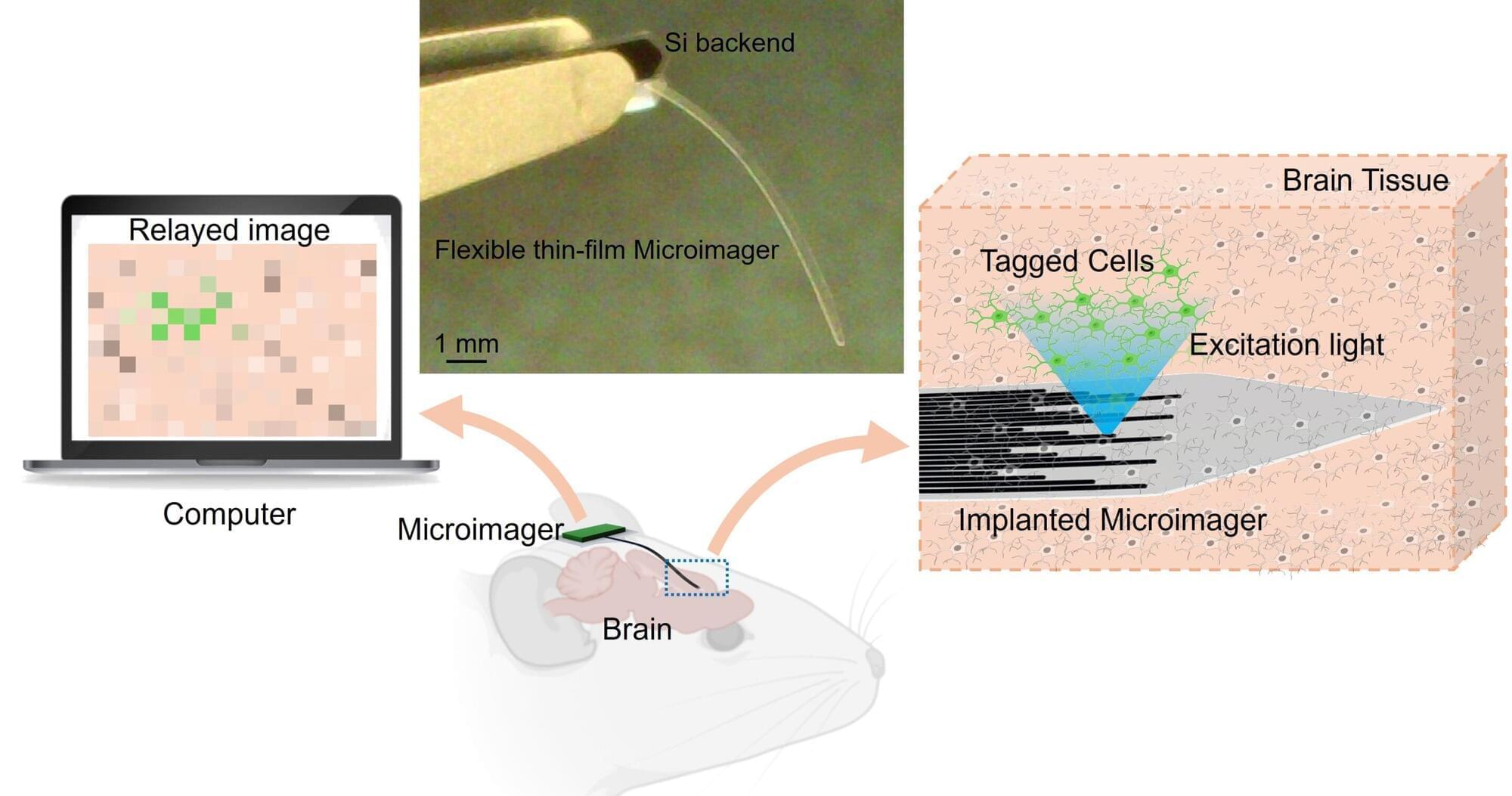
Researchers have developed an extremely thin, flexible imager that could be useful for noninvasively acquiring images from inside the body. The new technology could one day enable early and precise disease detection, providing critical insights to guide timely and effective treatment.
“As opposed to existing prohibitively large endoscopes made of cameras and optical lenses or bulky fiber optic bundles, our microimager is very compact,” said research team leader Maysam Chamanzar from Carnegie Mellon University. “Much thinner than a typical eyelash, our device is ideal for reaching deep regions of the body without causing significant damage to the tissue.”
In the journal Biomedical Optics Express, the researchers showed that the microimager, which is only 7 microns thick—a tenth of an eyelash diameter—and about 10 mm long, can be used in a mouse brain for structural and functional imaging of brain activity. The width of the thin film imager can be customized based on the desired field of view and resolution.
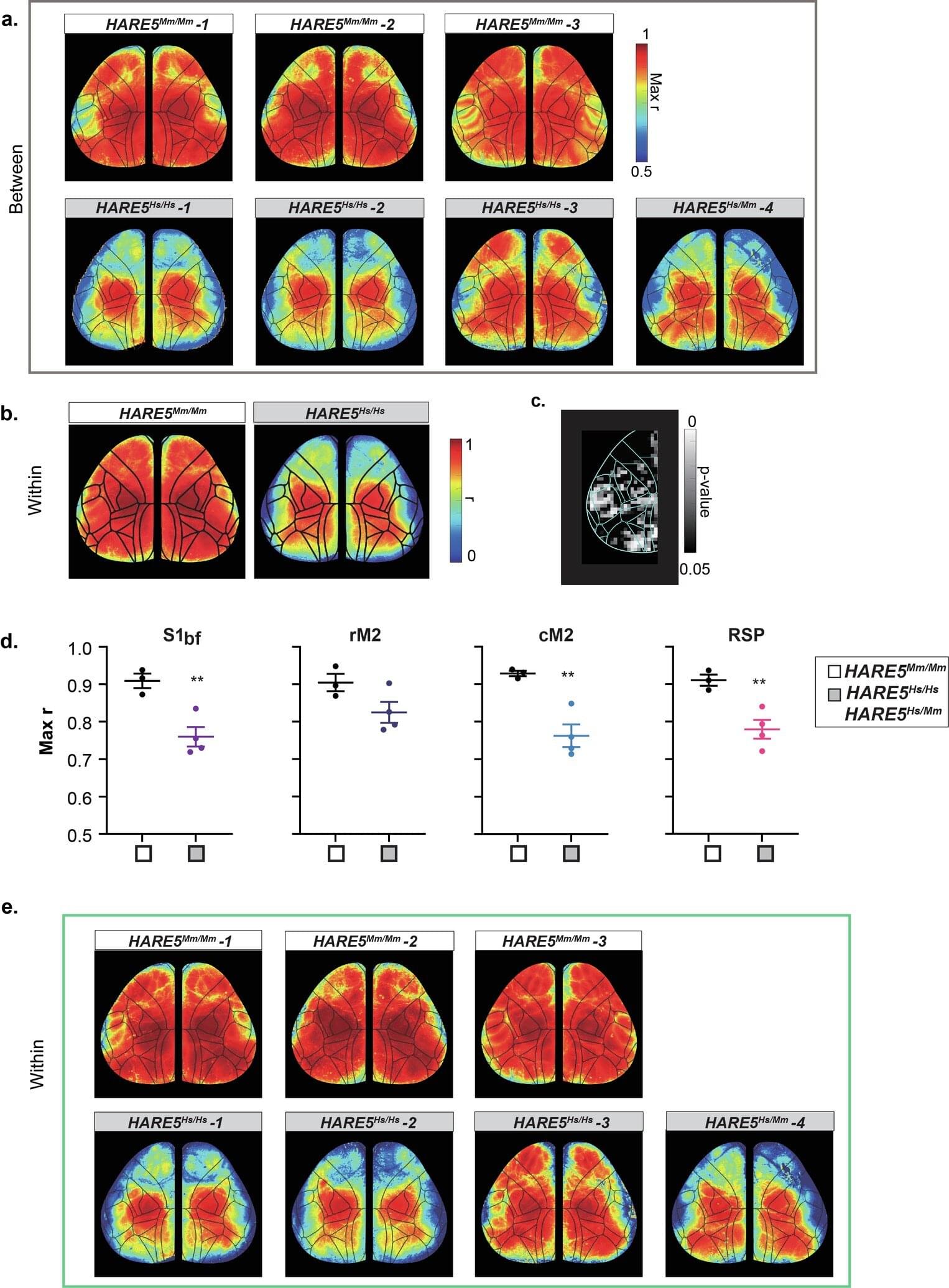
Duke University Medical Center-led research has identified a human-specific DNA enhancer that regulates neural progenitor proliferation and cortical size. Small genetic changes in HARE5 amplify a key developmental pathway, resulting in increased cortical size and neuron number in experimental models. Findings have implications for understanding the genetic mechanisms underlying neurodevelopmental disorders.
Humans possess a significantly larger and more complex cerebral cortex compared to other species, contributing to advanced cognitive functions. Comparative genomics research has identified Human Accelerated Regions (HARs), segments of non-coding DNA with human-specific genetic changes. Many HARs are located near genes associated with brain development and neural differentiation.
Because thousands of HARs have been identified and linked to brain-related genes, the next critical step is to investigate how these regulatory elements actively shape human brain features.