People who receive stem cell therapy within a week of their first heart attack have nearly a 60 per cent lower risk of developing heart failure years later

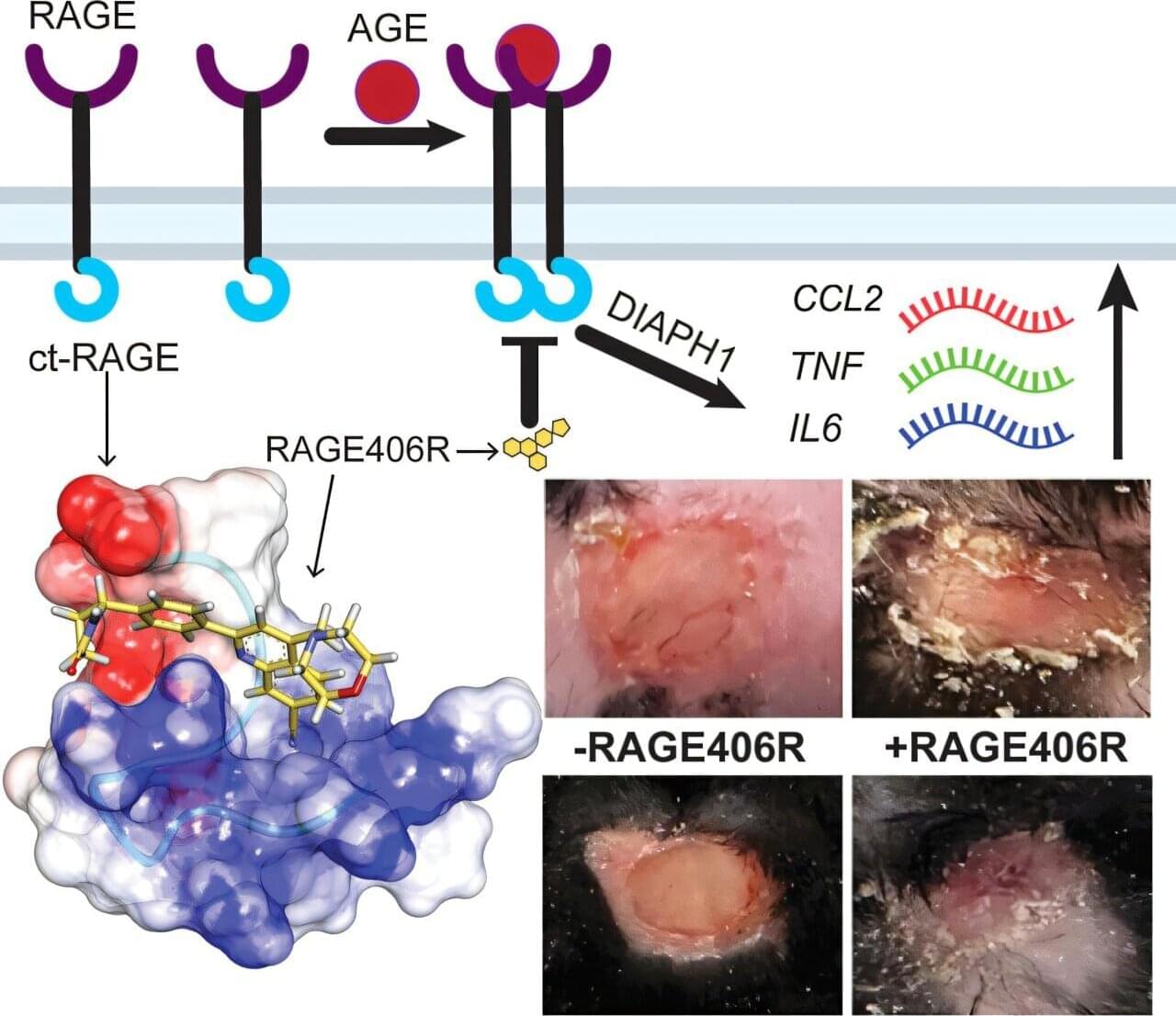
Researchers from the University at Albany and NYU Grossman School of Medicine have found a way to block a key cellular pathway known to drive chronic inflammation and impaired wound healing in people with diabetes.
The breakthrough could offer a new therapeutic option for stopping the harmful effects of both type 1 and type 2 diabetes at the source.
In their latest work, the researchers successfully identified—and developed a small molecule drug to disrupt—an intracellular chain reaction that is a major contributor to diabetes-induced complications. Their findings, published earlier this month, were featured on the cover of Cell Chemical Biology.
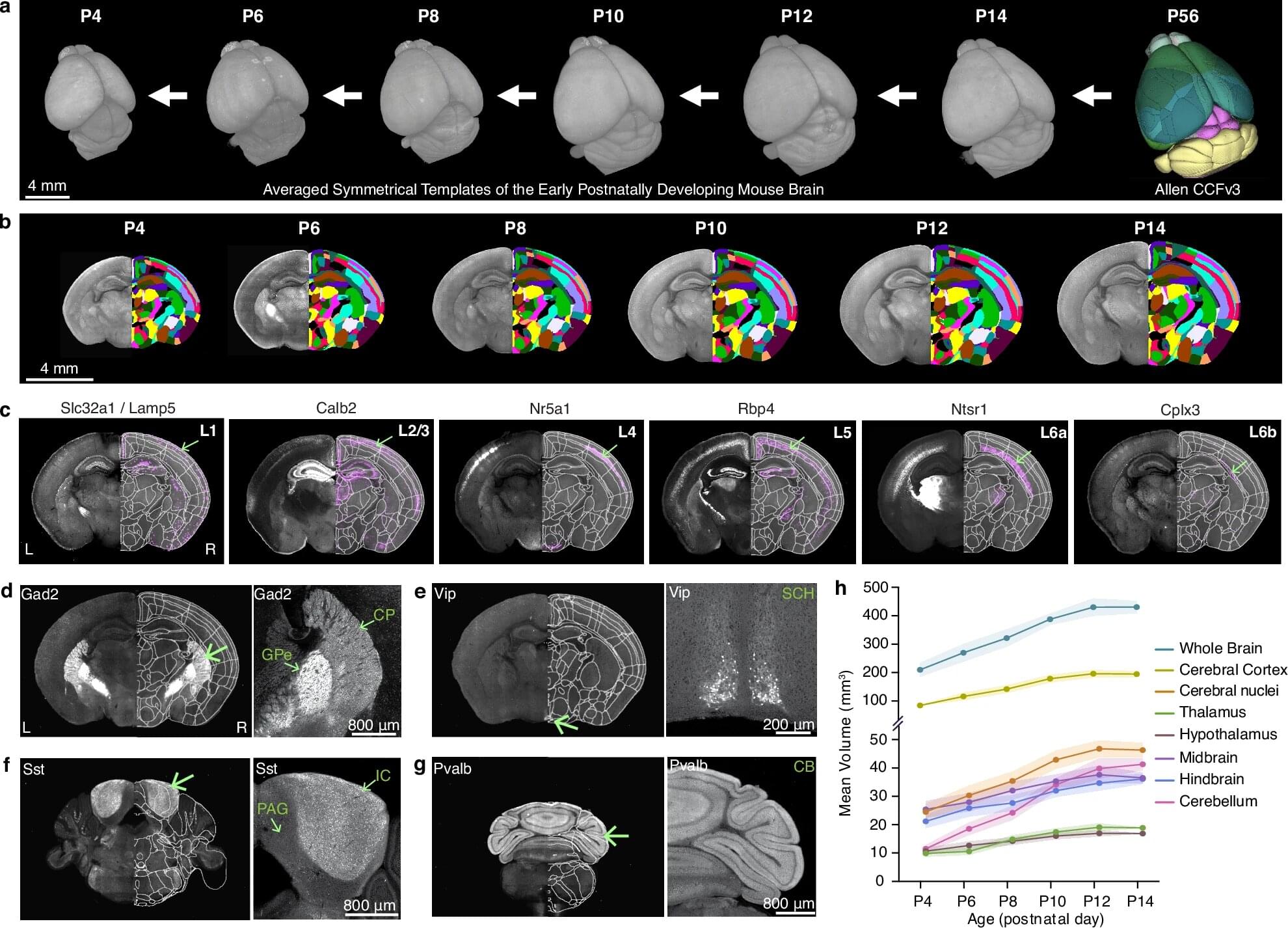
Brain growth and maturation doesn’t progress in a linear, stepwise fashion. Instead, it’s a dynamic, choreographed sequence that shifts in response to genetics and external stimuli like sight and sound. This is the first high-resolution growth chart to explain changes of key brain cell types in the developing mouse brain, led by a team at Penn State College of Medicine and the Allen Institute for Brain Science.
Using advanced imaging techniques, the researchers constructed a series of 3D atlases that are like time-lapsed maps of the brain during its first two weeks after birth, offering an unparalleled look at a critical period of brain development. It’s a powerful tool to understand healthy brain development and neurodevelopmental disorders, the researchers explained.
The study, published in Nature Communications, also detailed how regions of the brain change in volume and explained the shift in density of key cell types within them.
Scientists at Wake Forest University School of Medicine, in collaboration with researchers at Weill Cornell Medicine, have made a breakthrough in understanding why many cancer patients develop nerve damage after chemotherapy. Their new study reveals that a stress response inside certain immune cells can trigger this debilitating side effect. This discovery could open the door to new ways to prevent or treat nerve damage in cancer patients.
The study appears in Science Translational Medicine.
Chemotherapy-induced peripheral neuropathy is a common and often severe side effect of cancer treatment, especially with drugs like paclitaxel. It can cause tingling, numbness and pain in the hands and feet, sometimes forcing patients to stop life-saving treatment early. Up to half of all patients receiving chemotherapy may experience this condition, but until now, the exact cause has remained a mystery.
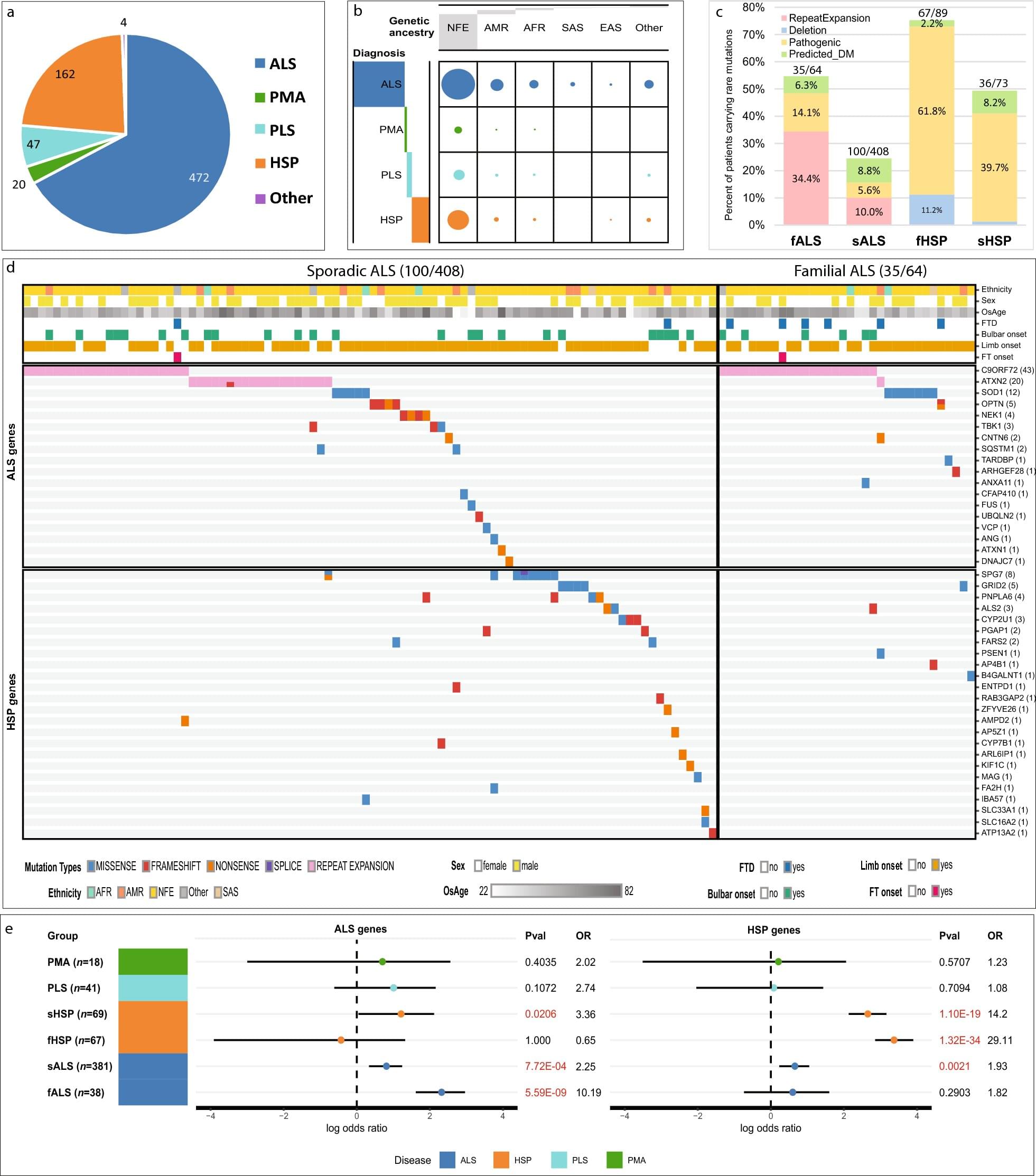
Motor neuron diseases, such as amyotrophic lateral sclerosis (ALS) and hereditary spastic paraplegia (HSP), share physical similarities but have been largely viewed as genetically distinct. However, an analysis led by investigators from St. Jude Children’s Research Hospital and the University of Miami Miller School of Medicine discovered that there are previously unknown ultrarare gene variants (genetic changes found in extremely few individuals) linked to the diseases, and significant overlap of contributing genes between the diseases among patients without family histories of a motor neuron disease.
This new appreciation of the shared genetic origins of different motor neuron diseases is critical to deciphering the origins of these disorders and ultimately developing meaningful therapeutics. The findings are published in Translational Neurodegeneration.
While both ALS and HSP cause progressive motor dysfunction, the two disorders also have distinct characteristics. Weakness in ALS may begin in the arms, legs, head or neck. HSP, by contrast, begins in the legs. The causative, or “canonical” genes for these diseases are also largely distinct.
An international team of scientists from three continents led by Dr. Petr Cígler of IOCB Prague has developed a method for creating light-emitting quantum centers in nanodiamonds in only a matter of minutes. In just one week, the process can yield as much material as conventional methods would produce in more than forty years.
Moreover, the resulting nanodiamonds show improved optical and quantum properties. The breakthrough brings us one step closer to the industrial production of higher-quality and more affordable quantum nanodiamonds, which have broad applications in research and technology. The article is published in Advanced Functional Materials.
The research team has introduced a new procedure called Pressure and Temperature Qubits (PTQ), which takes only four minutes. Diamond powder is placed in a press that generates extremely high pressure and temperature, reproducing the conditions found deep within Earth’s mantle. Under these conditions, quantum centers are formed inside the nanodiamonds.

ETH Zurich scientists have created “MetaGraph,” a revolutionary DNA search engine that functions like Google for genetic data. By compressing global genomic datasets by a factor of 300, it allows researchers to search trillions of DNA and RNA sequences in seconds instead of downloading massive data files. The tool could transform biomedical research and pandemic response.

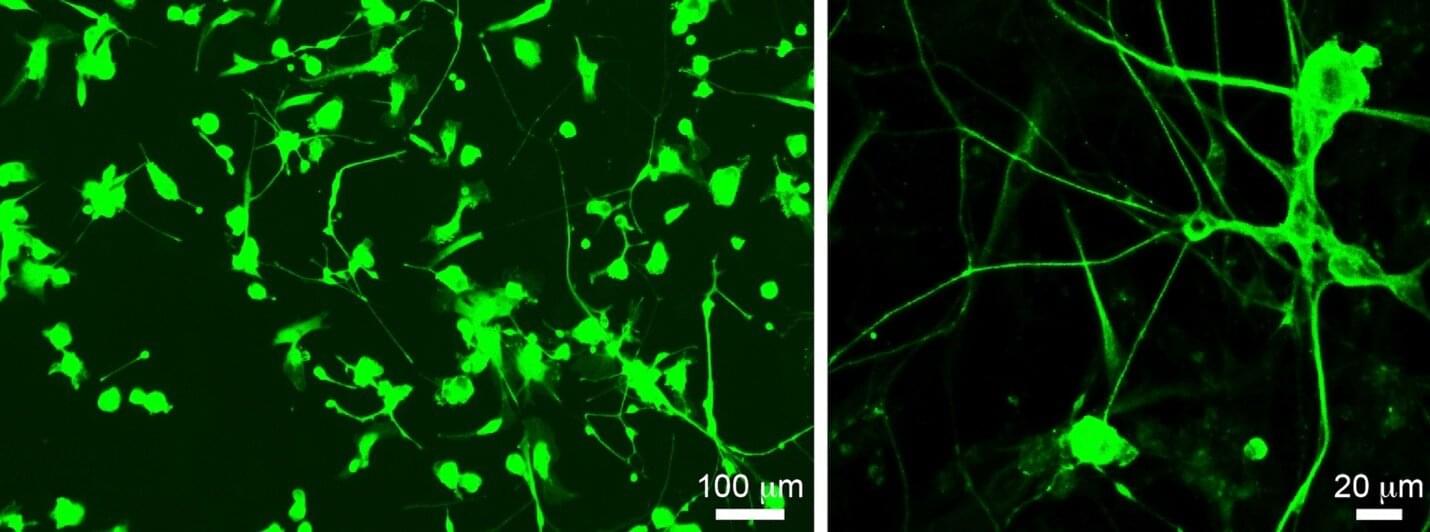
Spinal cord injury (SCI) remains a major unmet medical challenge, often resulting in permanent paralysis and disability with no effective treatments. Now, researchers at University of California San Diego School of Medicine have harnessed bioinformatics to fast-track the discovery of a promising new drug for SCI. The results will also make it easier for researchers around the world to translate their discoveries into treatments. The findings are published in the journal Nature.
One of the reasons SCI results in permanent disability is that the neurons that form our brain and spinal cord cannot effectively regenerate. Encouraging neurons to regenerate with drugs offers a promising possibility for treating these severe injuries.
The researchers found that under specific experimental conditions, some mouse neurons activate a specific pattern of genes related to neuronal growth and regeneration. To translate this fundamental discovery into a treatment, the researchers used data-driven bioinformatics approaches to compare their pattern to a vast database of compounds, looking for drugs that could activate these same genes and trigger neurons to regenerate.