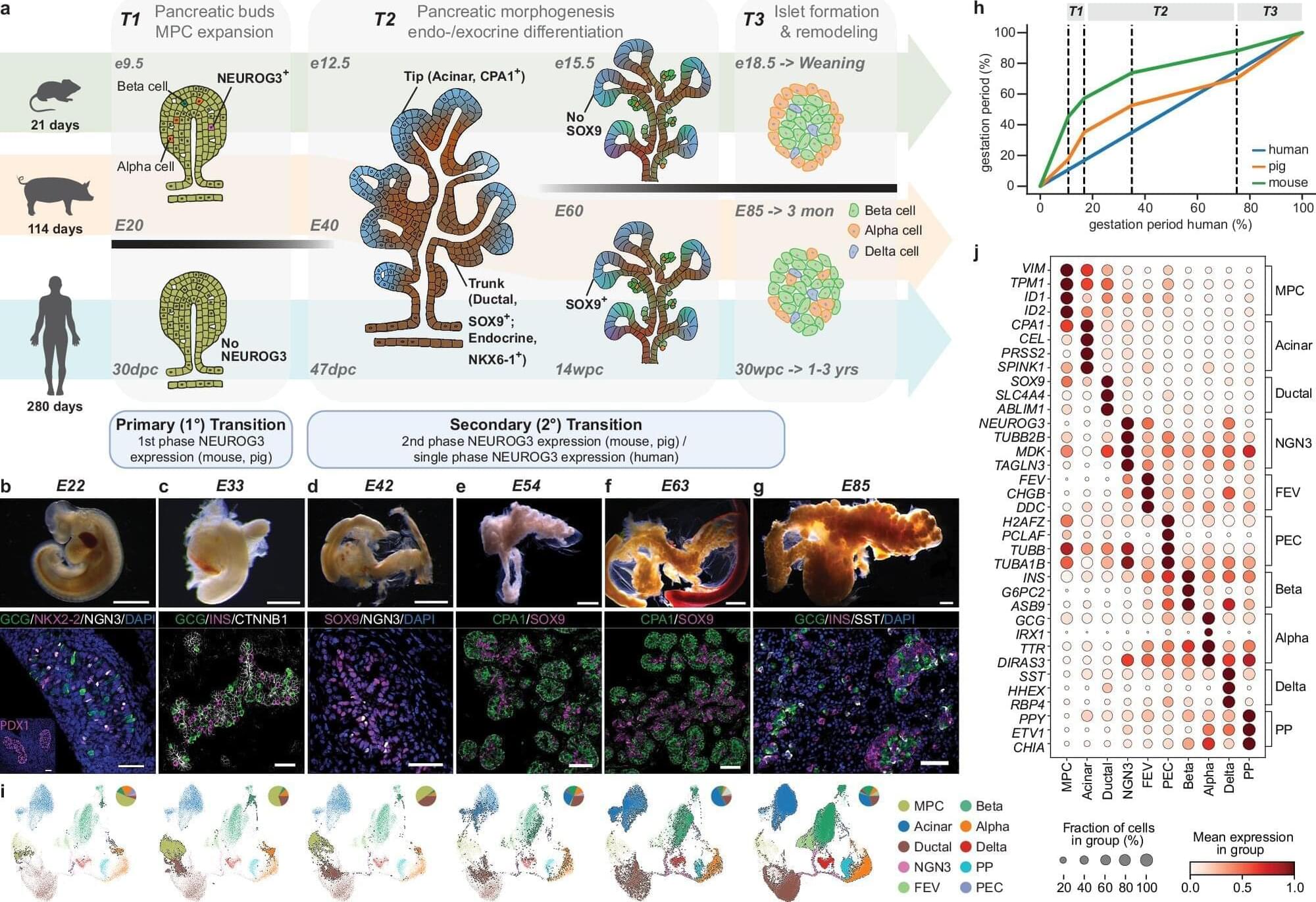
Pancreas development in pigs resembles humans much more closely than does the established mouse model. An international team headed by Helmholtz Munich and the German Center for Diabetes Research (DZD) has now produced a comprehensive evolutionary comparison of single-cell atlases of pancreas development. The results open up new prospects for regenerative therapies.
For decades, the pancreas and its development have been a major focus of diabetes and cancer research. Until now, the science was almost exclusively based on mouse models. However, mice differ from humans in many respects—from developmental duration to metabolism and gene regulation.
“Particularly for complex diseases such as diabetes mellitus, we need models that truly resemble humans,” therefore emphasizes Prof. Heiko Lickert. The DZD researcher is the director of the Institute of Diabetes and Regeneration Research at Helmholtz Munich and professor at the Technical University of Munich (TUM).